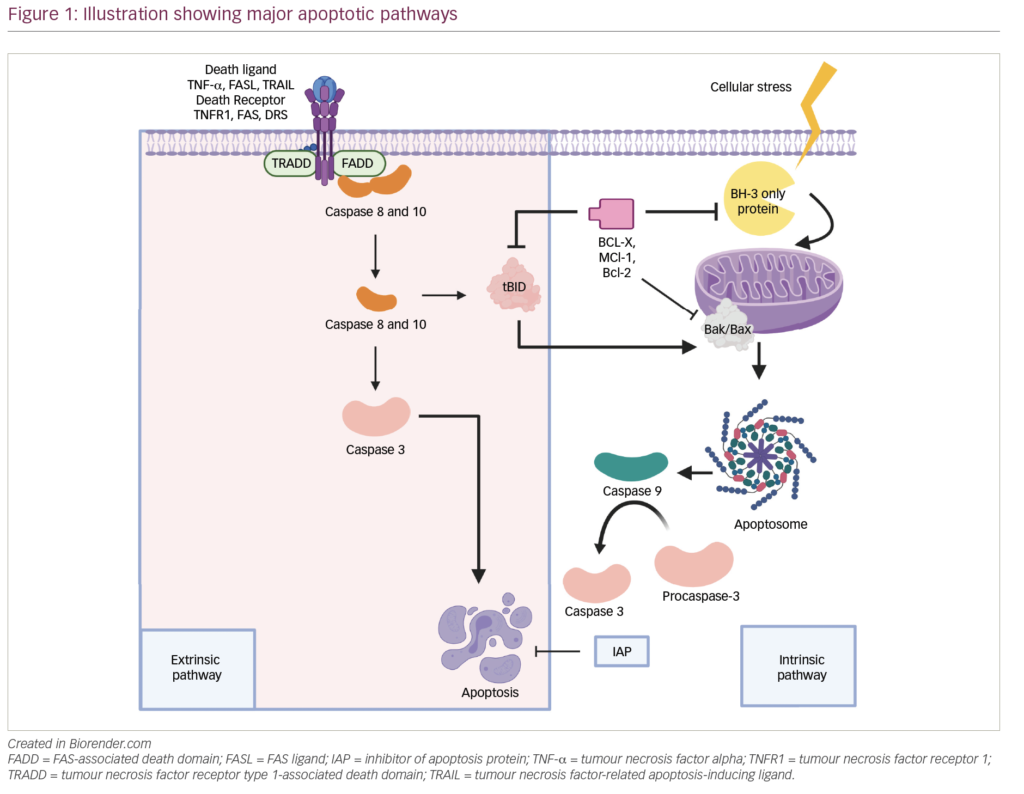
Multiple myeloma (MM) is a malignancy originating from a post-germinal centre lymphocyte, the antibody-secreting plasma cell. It remains incurable despite the fact that numerous pre-clinical and clinical investigations have been conducted. Therapeutic milestones allowed for the extension of survival times in affected subjects during a 35-year period, as reported recently.1 By nature, MM requires systemic therapy while local procedures (irradiation, surgical interventions) remain supportive treatment. Currently, presence of end-organ damage caused by the accumulation of malignant plasma cells (hypercalcaemia, renal insufficiency, anaemia and bone disease [CRAB]) is considered to be symptomatic myeloma and thus requires treatment.2 The initial attempts to bring cytotoxic chemotherapy to myeloma patients in the late 1960s consisted of drug combinations utilising the alkylator melphalan and corticosteroids.3,4
Melphalan and prednisone (MP) was considered the standard of care for patients with MM in the following years. Attempts to achieve disease control and survival rates beyond those seen with the use of MP by intensive chemotherapy combinations were not successful: an analysis of individual patient data combined with a meta-analysis of 27 trials revealed no difference in mortality rate for patients treated with MP compared with those receiving combination chemotherapy.5
High-dose Chemotherapy with Autologous Stem Cell Transplantation
Substantially increasing the dose of melphalan provided the basis for the first major breakthrough in myeloma treatment: the introduction of high-dose chemotherapy with subsequent retransfusion of autologous peripheral blood progenitor cells (APBPCs). Initial reports of dose-intensified melphalan (still without APBPC transplant) appeared in the early 1980s. Response to therapy in the first small series on nine subjects was remarkable, especially in those few who had received no prior treatment.6
However, a major obstacle was the high rate of infectious complications associated with a number of treatment-related deaths due to profound and long-lasting neutropenia. The administration of granulocyte-macrophage colony-stimulating factor (GM-CSF) shortened the duration of neutropenia without affecting the mortality rate.7 Following reports of successful transplantation of APBPCs in two previously untreated subjects with myeloma, this transplant modality replaced autologous bone marrow. Engraftment after APBPC transplantation was reliable, sustained and faster compared with bone marrow grafting, with APBPCs being more easily accessible.8–10
The first randomised trial documented high-dose therapy with autologous stem cell transplantation to be superior to conventional treatment in terms of response, progression-free survival (PFS) and overall survival (OS) in 1996.11 Since then, this modality has been widely adopted as a standard of care in up-front management for subjects with newly diagnosed disease up to 65 years of age. As malignant myeloma plasma cells are inherently responsive to irradiation and a broad range of cytostatic drugs, several approaches to achieving myeloablation before APBSC transplant have been investigated. High doses of the alkylator melphalan have become a backbone in the setting of autologous transplant alone or in conjunction with total body irradiation (TBI). However, in a randomised comparison no superiority for the melphalan/TBI combination was noted over an increased melphalan dose (200mg/m2 of body surface area, MEL200).12 This was due mainly to higher incidences of acute toxicity in the irradiation-based approach. Relatively few data have become available since then focusing on further improvement of conditioning. Only recently, the Intergroupe Francophone du Myélome (IFM) group reported on their experience incorporating the first-in-class proteasome inhibitor bortezomib into a single high-dose melphalan-based conditioning protocol (Bor–Mel). Fifty-four subjects <65 years of age with symptomatic myeloma received four doses of bortezomib at 1mg/m2 with two doses each before and after melphalan 200mg/m2.13 The primary objective was the cumulative complete remission (CR) plus very good partial remission (VGPR) rate three months following autologous transplant. At the time of response assessment, 70% of patients had achieved at least VGPR, including 32% CR. Interestingly, Bor–Mel seemed to be equally effective irrespective of whether patients had received standard vincristin/ adriamycin/dexamethasone (D) or bortezomib–dexamethasone prior to stem cell transplantation. A case–control study suggests Bor–Mel to induce slightly more CR than melphalan 200mg/m2 alone; however, no prospective comparison has been carried out.
Single versus Double Transplant
A further intensification of treatment was pioneered by the Arkansas group over 10 years ago. Patients with newly diagnosed disease received two sequenced autotransplants. The first transplant was delivered after uniform conditioning with MEL200, while three options were possible prior to the second APBPC transplantation.14 Randomised comparisons of single versus tandem transplants have been published by French and Italian groups.15,16 While both studies showed an increase in response quality (higher proportion of subjects achieving CR/near CR [nCR] and VGPR) and PFS after tandem transplant, OS following two cycles of high-dose treatment was superior only in the French trial. Interestingly, tandem transplants seemed beneficial in both studies mainly for patients with less than VGPR achieved following the first transplantation. Those results should be interpreted with caution as they are based on retrospective analyses.
Autologous Transplantation in Elderly Subjects
Whether high-dose chemotherapy and APBPC transplantation also improves treatment outcomes in subjects >60 years of age has been a matter of debate. In an Italian protocol, previously untreated patients between 55 and 75 years of age (median 64 years) were to receive three cycles of melphalan 100mg/m2 each followed by transplantation of APBPCs. Their outcomes were compared with pair mates having received standard MP: the dose-reduced ‘high-dose’ protocol was feasible and no toxic deaths occurred. Significantly, more complete responses were seen following autologous transplant, event-free survival (EFS) was doubled and even OS was significantly increased compared with standard treatment.17 However, advances in myeloma treatment may challenge standards of care in relatively short intervals of time; this was true for a three-arm randomised trial by the French IFM group in subjects between 65 and 75 years of age. Patients were to receive – apart from standard MP and one cycle of high-dose melphalan 100mg/m2 – MP plus thalidomide.18 Thalidomide, a compound with anti-angiogenic and anti-inflammatory properties, is known to be effective in MM and has been extensively studied in both newly diagnosed and relapsed disease.19–21 In this three-arm trial, MP plus thalidomide (MPT regimen) yielded the best results in terms of response rate, PFS and OS. Therefore, the authors concluded that MPT should be the new standard of care in elderly myeloma patients.
A slightly longer time to event (EFS) was observed in a German multicentre trial utilising tandem transplants after conditioning with melphalan 140mg/m2. The authors randomly compared four cycles of anthracycline/dexamethasone-based induction followed by stem cell mobilisation versus immediate apheresis. The EFS was 20 months without versus 22 months with induction treatment.22 This small difference was not significant. Considering event and treatment-free interval, the German approach was (in a historic comparison) superior to several other trials in this age group, allowing for a 16-month period off treatment.
Novel Compounds in Patients Not Eligible for Transplantation
Given the convincing results with the ‘novel drugs’ thalidomide, bortezomib and lenalidomide in patients with relapsed or relapsed/ refractory disease,19,20,23,24 it seems logical to translate their activity into first-line treatment. Mainly, combinations of novel drug + MP have been tested in phase III trials. Besides the previously described French IFM 99-06 study,18 four other studies have investigated MPT in elderly non-transplant subjects. As a comparator, all four had utilised standard MP.25–28 While all five trials yielded superior results in terms of EFS and PFS, prolongation of OS was reported from the two French studies only.18,26 This may be due to different age limits and treatment schedules. The Dutch and the Nordic groups in particular utilised relatively high thalidomide doses (400mg daily), which in conjunction with significant toxicity may have led to relatively low treatment intensity. However, those studies have not yet been published as full papers.
On average, PFS/EFS extension with MPT versus MP was five to seven months, with the exception of the Nordic protocol (two months). The range of PFS in the trials was 10–19 months (MP) and 13–28 months (MPT), respectively. A median PFS of 16.7 months was achieved with the combination of thalidomide/dexamethasone (TD) versus 20.7 months with MP in a randomised trial. Despite high-quality response being in favour of TD, OS for this arm was significantly shorter compared with MP.28 One explanation for these results was the higher toxicity seen with TD in frail elderly subjects; however, one might speculate on the impact of an alkylator (melphalan) for longevity of remission achieved. Based on the results of a phase I/II trial, the combination of the first-in-class proteasome inhibitor bortezomib with MP (VMP) versus MP alone was subsequently tested in a randomised phase III study: patients allocated to VMP had better outcomes for response rate and all survival parameters and also achieved responses more rapidly.30
A subgroup analysis suggested that VMP is also active in unfavourable cytogenetic subgroups. Median time to progression was prolonged by approximately seven months following VMP, with patients achieving CR having duration of that remission of two years. Recently, an Italian group reported on their experience with the MPR regimen31 (melphalan, prednisone and lenalidomide). Subjects having received the maximum tolerated dose of MPR achieved a median PFS of 28.5 months and a two-year OS of 91%32 (see Table 1).
Novel Compounds in Patients Eligible for Transplantation
TD given to patients with newly diagnosed MM resulted in significantly more severe (grade >3) toxicity compared with high-dose dexamethasone alone: 45% for TD versus 21% for dexamethasone. However, the response rate was significantly higher in the combination arm (63 versus 41%).33 The difference in response favouring TD over dexamethasone was retained in a larger phase III trial, while the difference in grade 4 toxicity was less pronounced (30% TD versus 23% dexamethasone).34 Since it is currently not known whether a favourable response to novel drug combinations can abrogate high-dose chemotherapy and PBPC transplantation, numerous clinical protocols use such combinations to improve disease control in the early stages while continuing to use the consolidatory high-dose effect. Comparing standard vincristine/doxorubicin/dexamethasone (VAD) induction with thalidomide/doxorubicin/dexamethasone (TAD) followed by stem cell mobilisation and single or tandem transplant, a Dutch–German co-operative group found a higher VGPR rate both before and after transplant, while CRs were not significantly different.35 Data for PFS/EFS and OS are still awaited. The addition of thalidomide to standard therapy in a tandem-transplant setting yielded a higher rate and a longer duration of VGPR or better, with a trend for better OS compared with pair mates treated without thalidomide.36 The Italian group reported PFS at four years of 51% with versus only 31% without TD. Post-relapse survival was comparable (24 months with versus 25 months without previous thalidomide). The Arkansas group incorporated thalidomide in a randomised fashion throughout all treatment phases into their ‘TotalTherapy2’ protocol.37 Considering the primary end-point (five-year EFS), the subjects allocated to thalidomide fared significantly better: 56 versus 43% EFS without thalidomide. Of note, post-relapse survival was significantly shorter for patients having received thalidomide in their first-line therapy: 1.1 with versus 2.7 years without prior thalidomide exposure. This contributed to an identical OS, leading to speculation that thalidomide may induce myeloma drug resistance if (in comparison with the Italian schedule) it is administered for a long time.
Bortezomib/dexamethasone (VelDex) given to patients <65 years of age with untreated myeloma resulted in significantly higher overall response rate (ORR) and higher VGPR when randomly compared with standard VAD in a French trial.37 Those results were valid for assessment before and after high-dose chemotherapy. At a relatively short follow-up, two-year PFS was significantly extended following VelDex, but did not translate into superior OS at two years from randomisation. Recently, the German DSMM group reported an interim analysis on an ongoing up-front trial. Bortezomib, cyclophosphamide and dexamethasone (VelCD) was administered to patients with newly diagnosed disease before stem cell transplant.38 The maximum tolerated dose of cyclophosphamide had previously been determined to be 900mg/m2 on cycle day one.39 The ORR to VelCD was 84%, and in the interim analysis no statistically significant differences of response in relation to presence of adverse cytogenetic factors including del(17p) were detected. The thalidomide analogue lenalidomide (Revlimid®) is known to be effective in relapsed MM. A study investigated this immunomodulatory drug with dexamethasone in treatment-naïve patients randomly comparing conventional-dose (RD) with dose-reduced dex (Rd).40 Not only was toxicity significantly higher in the RD group, but also the one-year OS favoured reduced-dose dexamethasone. A landmark analysis of this trial compared response and survival data at two years: 102 patients had gone off continuous RD/Rd treatment and received autotransplants as a consolidation, while 91 subjects still received RD and an additional 120 Rd. OS at two years clearly favoured both autologous SCT and Rd over RD. Notably, assignment to transplant was sanctioned by investigators or patients probably harbouring bias; therefore, data beyond protocol-driven end-points should be interpreted with caution. Lenalidomide, dexamethasone in a modified dose and clarithromycin (Biaxin), known as the BiRD regimen, was used by a US group for induction in newly diagnosed myeloma patients.41 BiRD treatment (repeated every three weeks) was delivered on a continuous basis without foreseeing any means of consolidation. However, patients <65 years of age with acceptable performance status were allowed to go off protocol for high-dose treatment. Eighteen patients successfully underwent chemotherapy/G-CSF-based administration of APBPC one year from introduction of BiRD. For the vast majority, the course of SCT was uneventful. Response to BiRD was favourable, with 70% CR + VGPR for 52 subjects not proceeding to high-dose therapy receiving a mean number of 14 treatment cycles without having reached median EFS at the time of the report.
The German Myeloma Study Group (DSMM) group is currently evaluating lenalidomide/ adriamycin/ dexamethasone (RAD) in a phase II trial. Following stem cell harvest, all patients are to undergo one cycle of high-dose melphalan 200mg/m2. If risk factors are present – serological and cytogenetic (fluorescence in situ hybridisation [FISH] features) – the second transplant is a dose-reduced allogeneic stem cell transplantation (allo-SCT), while subjects without any of those variables (‘very favourable risk’) are to undergo a second autograft.
Stem Cell Mobilisation in Combinations of Novel Drugs
In contrast to the previous trial, significant impairment of stem cell mobilisation utilising R in conjunction with cyclophosphamide and dexamethasone was reported by a group from the Mayo Clinic.42 A trend towards a lower yield of CD34+ APBPCs was also reported from the bortezomib/dexamethasone arm of the French IFM 2005/01 trial,37 and two groups from the US observed difficulties in mobilisation after initial lenalidomide-based treatment.42,43 Recently, these unforeseen events prompted the International Myeloma Working Group to publish recommendations in terms of safe and adequate stem cell collection for subjects receiving at least one of the novel drugs for induction.44 The group’s summary was steady-state mobilisation using G-CSF alone to be adequate for thalidomide/dexamethasone and bortezomib/ dexamethasone, irrespective of further disease- and patient-related variables. For patients ≥65 years and/or receiving lenalidomide/ dexamethasone induction beyond four cycles, G-CSF alone is no longer recommended and should be replaced by cyclophosphamide + G-CSF. This approach should be used as first-line therapy in all cases when additional cytotoxic drugs were used in addition to lenalidomide/dexamethasone.
Allogeneic Stem Cell Transplantation as a First-line Approach
The efficacy of allo-SCT is mainly based on immune control of the malignant disease by donor-derived lymphocytes; however, direct cytotoxic effects from the conditioning regimen may add to response. Results from early trials employing myeloablative conditioning such as busulphan/cyclophosphamide were discouraging as they showed high transplant-related mortality (TRM) rates. Introduction of reduced-intensity conditioning (RIC) for myeloma resulted in less toxicity; however, ongoing disease relapse contributed to OSS rates of <50%.45,46 A retrospective registry study (the European Group for Blood & Marrow Transplantation [EBMT]) evaluating the impact of RIC compared with conventional ablative preparative regimens showed shorter PFS following RIC. OS was not different between the groups.47 Data on up-front allo-SCT are scarce compared with autologous transplantation. To date, results from only 129 subjects having undergone auto-/allo transplant in three randomised trials have been fully published. Inclusion criteria, design, preparative regimens and comparator arms were different between the French,48 the Italian49 and the Spanish50 studies. The IFM 99-0348 trial utilised melphalan 200 and auto-SCT followed by busulphan/fludarabine/antithymocyte globulin (ATG) and allo-SCT as conditioning regimens. The comparator arm (IFM 99-03) used an increased melphalan dose (220mg/m2 with or without an anti-interleukin 6 [IL-6] antibody), which at this time was a novel regimen. These studies included subjects with high-risk features (i.e. elevated β2-microglobulin and FISH del 13q). OS for auto-allo-SCT was 35 versus 41 months following double auto- SCT. Similarly, there was no difference between the treatment arms in terms of EFS. Of note, the CR rate was relatively low (33%) after auto-allo transplants.
An Italian protocol employed 2Gy TBI as a RIC conditioning for subjects following one uniform cycle of melphalan 200mg/m2. Eligibility for this trial was based on identification of a sibling donor, but was not confined to high-risk features. Fifty-eight out of 82 subjects for whom a donor had been identified eventually received their allografts. These patients fared better than the ones receiving tandem melphalan 200mg/m2 in that they achieved twice the tandem auto CR rate (55%) and had superior EFS as well as OS. OS was 80 months with allo-SCT versus 54 months following tandem-auto SCT. The Spanish Programa para el Estudio de la Terapeutica en Hemopatia Maligna (PETHEMA) allocated subjects responding with less than nCR to one autograft (conditioned with busulphan/ melphalan) to either allo-SCT (depending on availability of a donor) or a second auto transplant.50 Complete response to allogeneic transplant occurred in 40% of 25 subjects versus only 11% following tandem autografts. To date, at a short follow-up there was no difference in OS; however, a trend towards prolonged EFS emerged for the auto/allo arm.
Recently, a group from Seattle reported on their experience using 2Gy TBI + fludarabine as a conditioning regimen in 102 consecutively treated myeloma patients. With a considerable follow-up of more than six years, estimated five-year OS was 64% and estimated five-year PFS was 36%. The authors claimed patients receiving their allografts no later than 10 months from initial treatment and being in good performance status to have a particular good prognosis. Unfortunately, there was no plateau but an ongoing relapse risk following allogeneic transplant. Finally, the German DSMM group investigated tandem high dose melphalan versus high-dose melphalan followed by a fludarabine/melphalan-based allo-RIC regimen in subjects with FISH-del 13q. Between 2002 and 2007, 199 subjects were recruited, 126 of whom received allografts from either sibling or unrelated donors, making it the largest series to date. An interim analysis was submitted to the 2009 annual meeting of the American Society of Hematology (Knop and Einselle, unpublished data). In all, optimal timing, conditioning regimen and patient- and disease-specific characteristics need to be determined to render allo- SCT a suitable first-line regimen for larger patient cohorts.
Conclusion
During the past two decades, significant progress has been made in first-line therapy for MM by both transplantation- and pharmacotherapy-based approaches. Currently, high-dose melphalan 200mg/m2 with support of APBPCs following cytoreduction with novel agent + dexamethasone (it is unclear whether the additional cytostatic agent contributes to enhanced outcome) can be considered a standard of care for patients up to 65–70 years of age. It is still a matter of debate whether immediate autotransplant needs to be performed in subjects with excellent remission following induction treatment. Deferred high-dose therapy (i.e. at first disease progression) may be an option that does not worsen a given patient’s prognosis. For transplant-eligible subjects, allogeneic-RIC transplant may offer extension of treatment-free intervals and even cure; however, optimal timing and patient selection should be further elucidated. As relapse occurs late after allo- SCT, drugs preferably with immunomodulatory properties may add to more durable disease control. In elderly subjects, melphalan/prednisone plus a novel agent represents a breakthrough after decades of stagnation. Whether melphalan/prednisone given in conjunction with a novel drug may be substituted for dexamethasone seems unclear given that TD was not superior to MP in a randomised comparison. ■