Just over a decade ago, regional nodal basins were assessed by complete lymph node dissections, either in the axilla, the groin, or in the neck, depending on the location of the primary tumor.4 The problem with this approach was that many patients did not ultimately have lymph node metastases, and did not, therefore, benefit from more accurate staging or better locoregional control, although all patients suffered the morbidity of extensive surgery.5–6 In 1992, a new technique involving intra-operative lymphatic mapping was reported for the first time.7 This technique revolutionized the assessment of regional lymph nodes and is now known as sentinel lymph node (SLN) mapping, performed in combination with a sentinel lymph node biopsy (SLNB).7
SLNB Principles and Techniques
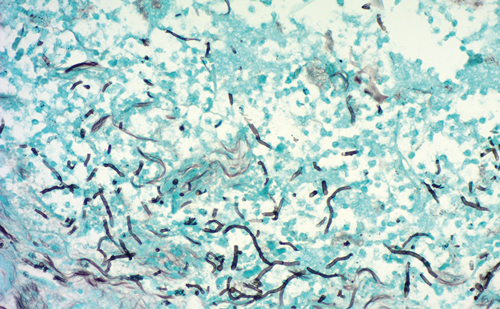
Morton and colleagues were the first to demonstrate that lymphatic drainage from a melanoma can be ‘mapped’ by injecting the skin around the tumor with blue dye.7 Injected blue dye was shown to travel through lymphatic channels to the first, or ‘sentinel’, lymph node that drains the tumor (see Figure 1). After intense histologic analysis of the SLN, these investigators were able to show that the pathologic status of the SLN accurately reflected the pathologic status of the entire regional nodal basin. A positive SLN is associated with a higher chance of the remaining nodes containing metastases, while patients with negative SLNs infrequently have other nodes that contain tumor cells.7,8 SLN mapping therefore spares approximately 80–85% of patients with melanoma and 65–70% of patients with breast cancer from having a complete lymph node dissection for what will ultimately be a negative nodal basin.7–10
The technique of SLN mapping has been significantly refined over the past decade. The accuracy of detecting the SLN when using blue dye alone is approximately 80%.7–10 In order to further increase the accuracy of detecting the SLN, a radiolabeled tracer such as technetium-99m-labeled sulfur colloid is also injected at the site of the primary tumor.11 A lymphoscintogram is an X-ray obtained before surgery that localizes the radiolabeled SLN and visually directs the surgeon to the proper node(s) and basin for biopsy. The SLN is now considered ‘hot’ with radioactivity and can be measured quantitatively by a gamma counter probe.The incision is targeted directly over the SLN by using the gamma probe to detect the area of highest radioactivity. The surgeon then follows the blue-colored lymphatic channels to the SLN, in addition to using the gamma counter to find the SLN with the most radioactivity (see Figure 2). After excision, the blue and hot node is counted with the gamma probe ex vivo and the number is recorded. In addition to removing any blue nodes, any remaining nodes in the regional basin that are greater than 10% of the ‘hottest’ node are also removed. Using these combined modalities, the accuracy of detecting the SLN has increased to between 92% and 98%.7–12 Pathologic Assessment of the Sentinel Node
Another significant stride in SLN mapping involves targeted histologic analysis by the pathologist. The pathologist receives a single lymph node, or just a few nodes, which can be subjected to a much more intense and targeted analysis. In addition to performing many levels of tissue slicing with hematoxylin and eosin (H&E)-staining, immunohistochemistry (IHC) is performed as a complement to H&E. IHC involves the use of tumor-specific antibodies to detect micrometastases. IHC confirms the presence of tumor cells in cases where H&E is not definitive, and has also been shown to detect an additional 8–12% of tumor cells that were missed by H&E staining.13–14 The clinical significance of micrometastases detected by IHC only is not completely clear at the present time.
Indications for SLN Biopsyin Melanoma and Breast Cancer
Patients with melanoma most likely to benefit from SLNB have an intermediate thickness (1.0–4.0mm) lesion.7 The incidence of nodal metastases ranges from 6% for patients with a 1.0mm melanoma to 35% in patients with a 4.0mm melanoma.12 For patients with melanoma <1.0mm, other indications for SLNB include Clark IV depth, presence of tumor regression, and presence of ulceration.15 Some relative indications may include younger age, a high number of mitoses, male gender, and axial location.16 Patients with thick melanoma (>4.0mm) have a high incidence of systemic disease and should undergo a proper extent of disease evaluation. For patients without distant metastases or clinically positive nodes, SLNB should still be offered as it provides valuable staging and prognostic information.17
For patients with breast cancer, SLN mapping and biopsy is indicated for T1 and T2 lesions (<5cm), provided that there are no clinically palpable nodes.3 The incidence of finding a positive SLN is 30–35% for patients with earlystage breast cancer.3,9–11 The incidence of finding a positive SLN in patients with ductal carcinoma in situ (DCIS) is so small that SLN mapping is not recommended for patients undergoing lumpectomy.3 However, SLNB is acceptable for patients with DCIS when total mastectomy is to be performed, mainly because SLNB is impossible after all breast tissue is removed, and if invasive cancer is found in the specimen, the patient would be committed to axillary lymph node dissection (ALND).
Clinical Trials Involving SLN Mapping for Melanoma
Four major prospective, randomized clinical trials are evaluating lymphatic mapping and SLNB for melanoma. The first two are the Multicenter Selective Lymphadenectomy Trial-I (MSLT-I) and the MSLT-II. The MSLT-I randomized patients with intermediate thickness melanoma to receive wide-excision and observation or wide-excision plus SLNB. If the SLNB revealed a positive node, patients then had a completion lymph node dissection (CLND). The goal of this study was to determine if early detection and management of regional nodes would improve survival.Patients who had wide-excision alone had a therapeutic lymph node dissection (TLND) only if clinical nodal recurrence was detected during the observation period. Results from the MSLT-I were recently presented at the 2005 American Society of Clinical Oncology (ASCO) meeting and demonstrated no difference in five-year overall survival between the two groups.18 However, 18% of patients treated by wide-excision alone had regional nodal recurrence, indicating that SLNB followed by CLND provides excellent locoregional nodal control. The goal of the MSLT-II is to determine if SLNB alone is adequate therapy for patients with a positive SLN, or if patients should proceed to CLND.The MSLT-II enrolls patients with intermediate thickness melanoma to wide excision with an SLNB, and then randomizes patients with a positive SLN to either observation or CLND. Results from the MSLT-II are not yet available.The hypothesis of the Florida Melanoma Trial (FMT) is that patients with a positive SLN who undergo CLND have no disease-free survival or overall survival advantage when compared to patients who have an SLNB alone, provided that all patients are also treated post-operatively with adjuvant interferon alpha-2b (IFN α−2b).This trial further stratifies patients based on the method of detection of metastases in the SLN, as determined by H&E, IHC, and by quantitative polymerase chain reaction (PCR).
The Sunbelt Melanoma Trial (SBMT) has a complex study design, with a total of seven arms in the randomization process. All patients with melanoma >1.0mm had SLN mapping and biopsy, with histologic analysis for micrometastases determined by H&E and IHC.All patients with a histologically positive SLN had CLND, and then patients were further randomized to either observation or adjuvant IFN α-2b. This part of the trial will determine the role of adjuvant IFN α-2b in patients with minimal nodal disease. Patients who had a histologically negative SLN then had further molecular analysis of the node by PCR. Patients with a PCR negative node will be observed, but those with a PCR positive node were randomized to observation, CLND, or CLND with IFN α-2b.This part of the trial will determine the value of molecular staging by PCR, and will determine the optimal treatment for patients upstaged by PCR.
Clinical Trials Involving SLNMapping for Breast Cancer
Three prospective, randomized clinical trials are evaluating SLNB in women with breast cancer. The National Surgical Adjuvant Breast and Bowel Project (NSABP) B-32 opened in 1999 and randomized women with clinically node-negative early-stage breast cancer to undergo SLNB alone, or SLNB with mandatory ALND. For patients randomized to the SLNB-alone arm, if the node was positive then an ALND was offered.The goal of the NSABP B-32 trial is to determine the value of SLNB alone compared to SLNB followed by ALND for patients with a histologically negative SLN.
Conclusion
Sentinel lymph node mapping and biopsy is a relatively new technique for staging and treating patients with melanoma or breast cancer.With tremendous benefits and minimal risk, SLNB has become so widely adopted that elective lymph node dissections are practically antiquated. Although this procedure has a learning curve, most surgeons who treat breast cancer and melanoma have been able to incorporate this technique into their practice.
Results from the clinical trials presented in this article will answer many questions about SLN mapping and will likely change the management of these diseases significantly over the next few years.A multidisciplinary team approach will certainly help physicians evolve as novel therapies and treatment algorithms are developed.
ACRO Meeting Focuses on Successes and Challenges for Radiation Oncologists`
The American College of Radiation Oncology (ACRO) held its 16th annual meeting at Disney’s Contemporary Resort in Orlando from February 23–25, 2006. The meeting’s program reflected the state of the field, including recent advances and current challenges.
Educational Program
Brian Kavanagh, MD, from the Anschutz Cancer Pavilion in Aurora, CO, explained the historical development and basic concepts of stereotactic body radiation therapy. John Kresl, MD, PhD, from St Joseph’s Hospital in Phoenix, AZ, followed with a review of robotic radiosurgery, pointing out its ability to correct continuously for patient and target motion. Then Anjit Singh, PhD, from Siemens Medical Solutions, gave an overall perspective of adaptive radiotherapy, showing how radiation oncologists must approach the problem of treating a tumor that changes its location and shape.
Intensity-modulated radiation therapy (IMRT) was also a hot topic. Arno Mundt, MD, from the University of Chicago, IL, suggested that gynecologic IMRT could be an alternative to brachytherapy for cervical and endometrial cancers. Seymour Levitt, MD, from the University of Minnesota, extolled advances in targeting radiotherapy, but warned ACRO members to be aware of the limitations of IMRT. He and Clifford Chao, MD, from the M D Anderson Cancer Center in Houston, TX, reminded meeting attendees that they needed to have the proper expertise to handle the targeting. In addition, the University of Alabama’s Ruby Meredith, MD, PhD, described clinical trials to evaluate radiolabeled antibody therapy, especially as it relates to non-Hodgkin lymphoma. Roger Macklis, MD, from the Cleveland Clinic, OH, noted how radioimmunotherapy must involve cooperative efforts between radiation oncologists and those in nuclear medicine.Other procedures discussed were Y-90 miscrosphere brachytherapy for liver malignancies (by Andrew Kennedy,MD, of Wake Radiology Oncology in Cary, NC), the integration of antiangiogenic therapy (by Michael O’Reilly,MD, from the M D Anderson Cancer Center), and heat shock proteins as a model for new molecular targets (by David Gius,MD, PhD, of the National Cancer Institute, Bethesda, MD). The future of radiation oncology was previewed by Lajos Balogh,PhD, from the Roswell Park Cancer Institute in Buffalo, NY, who told the ACRO attendees about nanotechnology and its potential application for radiation therapy.
Economics and Regulations
Tim Williams, MD, from Boca Raton Community Hospital, FL, focused the meeting on economic issues when he described reimbursement as it pertains to targeted therapies. Further, Michael Kuettel, MD, PhD, MBA, also from the Roswell Park Cancer Institute, gave ACRO members a healthcare economics update. He also introduced Jennifer Dreyfus, MBA, a Washington consultant from Competitive Health Systems, who is being retained by ACRO to help members understand federal regulatory language. Andrew Woods, Esq., a long-time legal consultant for ACRO, from the Washington, DC, firm of King & Spalding, gave a sobering statement that Americans elect politicians who promise to cut taxes yet expect the highest quality healthcare. He emphasized that ACRO must work to convince government officials to continue funding for new technologies.
In addition, David Diamond, MD, from the Florida Oncology Network, talked about nuclear regulatory activities, and Robert Keenan, Esq., also from King & Spalding, talked about updates in the Stark antireferral laws.
Exhibitor Support
Nearly 50 technical exhibitors were in an adjacent hall to show off their latest wares and practice-management strategies.Vendors also funded breakfasts or lunches that featured speakers discussing subjects such as respiratory gating, radioprotection, and highdose- rate brachytherapy for cervical cancer.
The ACRO meeting is also a time to honor those in the field.The organization’s Gold Medal was given to former ACRO president and chairman A Robert Kagan, MD, who continues to serve as the editor-in-chief of the organization’s newsletter. Ralph Dobelbower, MD, PhD, who was instrumental in the development of the ACRO practice accreditation program, was given the prestigious Simon Kramer Award, named after one of the founders of ACRO.An additional three dozen physicians were inducted as new Fellows of the College.
ACRO Chairman James Rubenstein, MD, reminded meeting attendees of the many activities of ACRO, including a practice review and accreditation program, sponsorship of coding workshops, lobbying efforts, publication of a practice management guide, and the annual meeting. He added that ACRO would be featuring a new malpractice insurance program for radiation oncologists, and he announced that the American Journal of Clinical Oncology had recently been designated the organization’s official journal. The next annual ACRO meeting, he said, would be held at the Hotel del Coronado in San Diego, CA, from February 22–24, 2007.