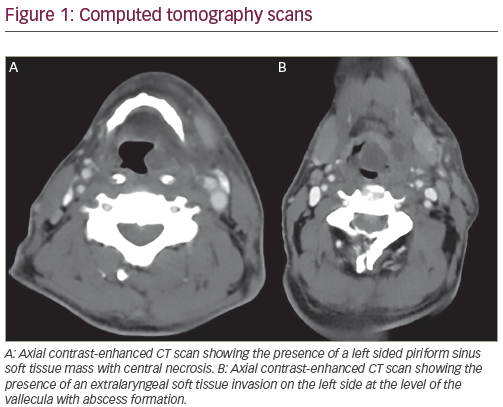
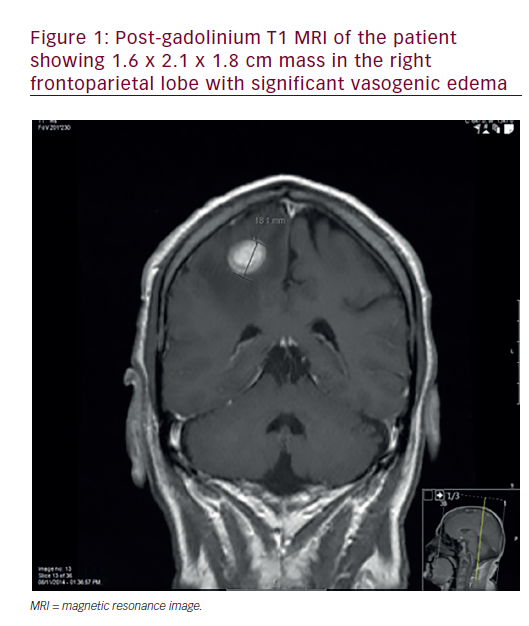
Over the past 25 years, systemic chemotherapy has been intensively investigated in the management of squamous cell head and neck cancer. Although modest palliative benefits have been identified for those patients with relapsed or refractory disease, it is when chemotherapy is coupled with definitive radiation in newly diagnosed patient that a significant improvement in the curative potential of non-surgical treatment has been demonstrated. Both sequential and concurrent schedules have been investigated.
The first success was seen in nasopharyngeal cancer, a disease that is generally not approached with definitive surgery. Intergroup 0099 demonstrated a highly significant improvement in survival for those patients treated with concurrent single-agent cisplatin and radiation followed by adjuvant 5-fluorouracil (5FU) and cisplatin compared with the results achieved after standard radiation therapy alone.1 Although this study was conducted in North America, similar results were also found in an endemic Asian population.2 Success was also achieved for other head and neck cancers subsites in Intergroup 0126, a study that randomized unresectable patients to radiation, radiation with concurrent cisplatin, and split-course radiation with concurrent cisplatin and 5FU.3 Concurrent cisplatin and radiation produced a three-year overall survival rate of 37%, which was statistically superior to the 23% survival achieved by radiation alone.
Larynx cancer was approached in a different fashion. A sequential, not concurrent treatment schedule was initially chosen for investigation with the goal of larynx preservation rather than improvement in survival. The first-generation larynx preservation trial from the Veterans Affairs Laryngeal Cancer Study Group (VALCSG) was reported in 1991, and demonstrated the ability of induction chemotherapy with 5FU and cisplatin followed by definitive radiation therapy in responders to preserve the larynx in 64% of patients without compromising their survival compared with patients undergoing laryngectomy followed by radiation.4 A second-generation larynx preservation trial, the Radiation Therapy Oncology Group (RTOG) 91-11 study, compared this induction chemotherapy schedule with definitive radiation alone and with radiation and concurrent cisplatin.5
The updated results of this study revealed a five-year locoregional control rate of 68.8% and larynx preservation rate of 83.6% in the concurrent chemotherapy and radiation arm—results that were statistically superior compared with the other two treatment groups.6 No survival advantage was observed for any treatment arm, which perhaps reflects the success of surgical salvage.7 Of note was a strong trend toward a reduced rate of distant metastases in the two treatment arms receiving chemotherapy. This work has established concurrent chemotherapy and radiation as the North American standard for organ preservation in locally advanced larynx cancer.8
Definitive non-operative treatment is not the optimal approach for all patients with this disease, and for many initial surgical resection is chosen. Radiation therapy has been considered a treatment standard for post-operative patients with high-risk pathological features such as positive surgical margins, extracapsular nodal invasion, angiolymphatic or perineural invasion, or multilevel lymph-node involvement. The benefit of chemotherapy has also been successfully demonstrated in these patients. Two very similar multi-institutional studies have been completed and have demonstrated the superiority of post-operative radiation and concurrent single-agent cisplatin compared with radiation alone.9,10 The similarity of these studies has allowed the results to be pooled.11 This analysis demonstrated a statistically significant improvement in survival with the addition of concurrent chemotherapy to post-operative radiation for those patients with either microscopically involved surgical margins or extracapsular nodal spread. Therefore, a consistent trend has emerged from a number of carefully conducted multi-institutional co-operative group trials, establishing concurrent chemotherapy (in general using single-agent high-dose cisplatin) and radiation as a standard of care in multiple clinical situations. A survival benefit has been demonstrated for patients with nasopharynx cancer, in patients with unresectable disease, and in the high-risk post-operative setting; furthermore, larynx preservation has been improved by this approach; a meta-analysis has confirmed this benefit: The Metaanalysis of Chemotherapy on Head and Neck Cancer (MACH-NC) collaborative group pooled 63 clinical trials performed between 1965 and 1993, which collectively enrolled more than 10,000 patients.12 In this analysis, the concomitant administration of chemotherapy with radiation yielded an 8% absolute survival benefit at five years.
Although an overall survival benefit was not observed in studies where chemotherapy was administered as induction prior to locoregional treatment, a marginally significant (p=0.05) benefit was found when only those induction regimens that employed a platinum and 5FU combination were examined. Similar treatment standards do not exist for the treatment of metastatic head and neck cancer, although a number of chemotherapeutic agents, including platinum compounds, fluoropyrimidines, folate antagonists, and taxanes, have demonstrated activity. Combinations of these chemotherapeutic agents appear to improve response rates; however, these come at the cost of increased toxicity with, historically, no demonstrated improvement in the six- to nine-month median survival of patients with metastatic disease.13–15 The landscape of head and neck cancer treatment is being transformed by advances in the understanding of the disease’s underlying molecular processes and patterns of treatment failure. The introduction of targeted agents and possible synergy with chemotherapy and radiation has made their integration into current treatment algorithms a subject of intensive scientific inquiry. The increasing recognition of distant metastasis as the most common pattern of failure in locally advanced disease treated with chemotherapy and radiation has led to the exploration of newer treatment approaches. This article summarizes some of the recent additions to the arsenal of chemotherapeutic options for head and neck cancer. Molecular Targeted Agents
The epidermal growth factor receptor (EGFR) is almost universally expressed in squamous cell cancers of the head and neck. Activation of this receptor results in downstream signaling, which mediates cellular proliferation, immortalization, and invasion. Its expression is an adverse prognostic indicator for survival and disease recurrence.16–18 When used as single agents, the oral EGFR inhibitors erlotinib and gefitinib and the monoclonal antibody cetuximab are known to have modest objective response rates of approximately 10% in chemotherapy refractory metastatic disease.19–22
The synergistic effect of EGFR inhibition and radiation or chemotherapy in head and neck cancer tumor cell lines is well recognized. This appears to be related to the restoration of the apoptotic response to these treatments.23,24 In a large phase III clinical trial, 424 patients with locally advanced head and neck cancer were randomized to either radiation alone or to radiation with concomitant cetuximab.25 Statistically superior three-year rates of locoregional control (47 versus 34%; p≤0.01) and overall survival (55 versus 45%; p=0.05) were observed in the experimental arm. One striking observation from this work was that unlike those trials testing the addition of chemotherapy to radiation, the improvements in outcome from adding cetuximab were not associated with an increase in treatment-related toxicity (with the exception of cutaneous and hypersensitivity reactions). However, the radiation therapy only control arm of this study was not the chemoradiation standard of care for locally advanced disease and, currently, no randomized comparison of radiation and cetuximab with concurrent chemoradiation has been performed.
The integration of EGFR inhibitors and concurrent chemoradiotherapy is being studied at several different venues. A phase II trial from Memorial Sloan-Kettering combining cetuximab with concurrent cisplatin and radiation reported encouraging results, although it was prematurely closed due to concern about adverse events.26 Two RTOG trials testing this combination have recently completed accrual and are awaiting results. RTOG 0522 is a randomized phase III study comparing cetuximab, cisplatin, and radiation versus cisplatin and radiation, and RTOG 0234 is a randomized phase II study comparing adjuvant cetuximab, cisplatin, and radiation versus cetuximab, docetaxel, and radiation after surgical resection. The Eastern Cooperative Oncology Group (ECOG) has also completed accrual to a phase II study (ECOG 3303) testing the addition of weekly cetuximab to concurrent cisplatin and radiation for unresectable locally advanced head and neck cancer.27 The regimen proved feasible and safe and the results were encouraging.
The use of EGFR inhibition with chemotherapy has also been explored in the setting of metastatic disease. The favorable side-effect profile and the relative ease of administration of these drugs make them an ideal agent for palliative therapy. An ECOG randomized phase III clinical trial compared cisplatin plus weekly cetuximab with cisplatin and placebo in 117 patients, and demonstrated a higher objective response rate with the combination (26 versus 10%; p=0.03) but no improvement in progression-free or overall survival.28 The recently published EXTREME (Cetuximab [Erbitox] in combination with Cisplatin or Carbo platin and S-Fluorouracil in the First Line Treatment of subjects with Recurrent and/or Metastatic Squamous Cell Carcinoma of the Head and Neck) trial randomized 442 patients with metastatic or recurrent head and neck cancer to either a combination of cisplatin (or carboplatin) and 5FU or to the same chemotherapy with cetuximab. An improvement in both the response rate and a survival advantage were noted with the chemotherapy and cetuximab combination (7.4 versus 10.2 months median survival; p=0.04), an unprecedented finding in randomized studies looking at chemotherapy combinations for metastatic head and neck cancer.29Sequential Therapy
Recently, the observation has been made that with the use of definitive concurrent chemotherapy and radiation, distant recurrence has replaced locoregional recurrence as the predominant pattern of treatment failure.30,31 Despite the lack of benefit for local control and the limited impact on overall survival, induction chemotherapy has repeatedly been demonstrated to decrease the rate of distant metastasis, suggesting that induction therapy may successfully influence this increasingly observed risk for distant failure. Furthermore, the delivery of active chemotherapy agents prior to definitive treatment allows for intensification of the treatment regimen without influencing the toxicity of definitive treatment. This has led to a resurgence of interest in induction chemotherapy prior to definitive concurrent chemoradiotherapy, or ‘sequential therapy.’ The introduction of taxanes and their activity in squamous cell head and neck cancers make these attractive agents for incorporation into induction chemotherapy regimens.
Three studies have demonstrated the superiority of taxane-containing chemotherapy combinations and their potential utility as induction regimens. The TAX324 study randomized 501 patients with locally advanced head and neck cancer to two arms: induction chemotherapy with cisplatin and 5FU or induction chemotherapy with cisplatin, 5FU, and docetaxel.32 Patients in both arms proceeded to definitive therapy using a non-standard radiation and concurrent weekly carboplatin schedule. Excellent overall and complete response rates were observed in both treatment groups. However, a clear survival advantage was demonstrated with the use of the taxane-based triplet combination as opposed to the more standard 5FU and cisplatin doublet (three-year overall survival 62 versus 48%; p=0.002). Locoregional control rates were also superior in the docetaxel-containing arm, although the rates of distant metastases were similar in the two arms.
Similar results were achieved by Vermorken et al. in TAX323, a study of 358 patients with locally advanced disease who were randomized to either induction chemotherapy with cisplatin and 5FU or cisplatin, 5FU, and doxetaxel prior to definitive radiation. Both progression-free survival and overall survival significantly favored the triplet induction combination.33 Hitt et al. compared the induction chemotherapy combination of paclitaxel, cisplatin, and 5FU with cisplatin and 5FU prior to concurrent cisplatin and radiation for patients with complete or major partial responses to induction therapy.34 The primary end-point of this study was complete response rate, and a result in favor of the taxane-containing induction regimen was observed (33 versus 14%; p<0.001). Overall survival was not statistically different between the two arms, but trended in favor of the taxane-containing induction regimen. The obvious conclusion from these trials is that a taxane, cisplatin, and 5FU induction chemotherapy regimen is better than the cisplatin and 5FU doublet; however, the choice of definitive management in these trials complicates the interpretation of the results. It is quite possible that the more aggressive and successful three-drug induction chemotherapy regimen merely compensated for the use of nonstandard or suboptimal locoregional treatment. The impressive partial and complete response rates to induction chemotherapy raise the question of how best to accomplish the post-induction radiation planning and delivery. Furthermore, a proportion of patients were not able to undergo definitive treatment, leading to concerns that the toxicity of induction chemotherapy may preclude definitive therapy in a subset of patients. The resolution of these matters and a true test of the value of these sequential approaches must await completion of the phase III clinical studies currently being conducted in the US and Europe (see Table 1). These studies compare three-drug induction chemotherapy followed by concurrent chemoradiotherapy with standard concurrent chemoradiotherapy alone. Human Papillomavirus-associated Disease
Perhaps the most important factor influencing current management strategies for this disease has been the recent recognition of the etiological and epidemiological importance of the human papillomavirus (HPV).35,36 Considerable evidence now exists that this virus is associated and likely etiological in the genesis of the majority of oropharynx cancer (but not other head and neck primary sites) that is diagnosed in the US. Patients with HPV-associated head and neck cancers tend to more commonly be younger, Caucasian, and non-smokers. Disease is more likely to be histologically poorly differentiated or basaloid in appearance and more advanced in clinical stage. There is a strong association with high-risk sexual behavior. Nonetheless, the prognosis of these HPVpositive patients after treatment is significantly better than those patients with HPV-negative oropharynx cancers or head and neck cancers of other primary sites.30,37 Therefore, it is apparent that the HPVpositive patient subset is a unique patient population that, if not properly accounted for, will favorably influence the impact of any treatment regimen being tested (see Table 2).
As such it is increasingly clear that this patient subset should be approached differently from the HPV-negative patients, that separate clinical questions should be asked, and that clinical trials should be conducted for the HPV-positive patients. Although their excellent prognosis after treatment is recognized, there is currently no information that would justify treating these patients any differently than their HPV-negative counterparts. This question will likely frame much of the future clinical investigations conducted for this disease.
Conclusion
Several intriguing new avenues for clinical exploration have opened in the treatment of squamous cell head and neck cancer. Molecular targets and their inhibitors are being actively studied in both the metastatic and definitive setting, both as single agents and in combination with more standard therapies. The EGFR inhibitors in combination with conventional treatments have already been demonstrated to improve survival in both previously untreated patients and patients with recurrent disease. The optimal integration of these agents into standard management strategies is being intensively tested. As newer targets are identified, newer targeted agents are entering clinical trials. Continued study of new conventional chemotherapeutics and combinations, as well as their sequencing and timing, is to be welcomed. Most importantly, a better understanding of disease behavior and etiology is allowing us to better stratify and study patient subsets and more appropriately individualize our management approach. ■