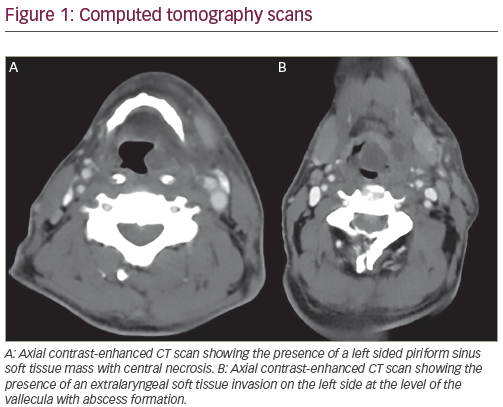
Squamous cell cancers of the head and neck (SCCHN) include a range of diseases consisting of cancers of the lip, gums, tongue, salivary glands, other oral cavity sites, nasal cavity, paranasal sinuses, pharynx and larynx. Locally advanced (LA) SCCHN tumours are at a stage at which they have locally spread from the primary site but have not yet advanced to a point of metastasis to other body sites. The poor recognition and low public awareness of SCCHN symptoms in its early stages results in about 501–70%2 of patients presenting with LA disease (stage III, IVb). Of these patients, approximately half subsequently develop locoregional or distant recurrences3 and the five-year survival rate in all patients with LA SCCHN is approximately 50%.1 Unlike most cancers, in which the primary cause of death is metastasis, the main reason for treatment failure and death in LA SCCHN is local recurrence. This article will focus on the treatment of LA SCCHN with cetuximab. A separate article reviewing targeted therapies in combination with chemotherapy for the treatment of recurrent and/or metastatic SCCHN by Pol Specenier and Jan Vermorken was published previously in European Oncology.4 Current Therapeutic Approaches for LA SCCHN
Patients presenting with LA SCCHN are usually treated with a combination of surgery, radiotherapy and chemotherapy. However, a minority of patients are suitable for potentially curative salvage surgery. Patients with LA SCCHN, especially where the tumour has not spread substantially from the primary site, are usually given treatment including either radiation therapy or surgery. Chemotherapy and irradiation can be given concomitantly (chemoradiotherapy) or sequentially, the chemotherapy often being given as the initial therapy (induction chemotherapy). Methotrexate was traditionally the first choice for chemotherapy, but it has now largely been superseded by platinum-based agents such as carboplatin or cisplatin, often administered with 5-fluorouracil (5-FU).
The suitability of induction chemotherapy for LA SCCHN therapy is controversial in terms of whether it is more effective than sequential therapy5 and treatment strategies for induction chemotherapy are continuing to evolve.6–8 The standard induction chemotherapy regimen consists of a platinum agent and 5-FU (often referred to as PF). More recently, the addition of a taxane such as docetaxel (or, less commonly, paclitaxel) to the PF regimen (a triple combination known as TPF) is emerging as a more effective and less toxic standard compared with PF for induction chemotherapy.9 Recent studies suggest that there is a lower risk of distant metastases after induction chemotherapy than after primary radiotherapy or chemoradiotherapy.10
Advances in the treatment of LA SCCHN over the past 30 years have provided some improvement in prognosis.11,12 Much of this improvement has been gained from the combination of different treatments rather than relying on a single treatment modality. However, there is a continuing requirement for more effective therapies to prevent both relapses and metastases and to improve overall survival. Improved understanding of the pathogenesis of SCCHN has led to the introduction of molecularly targeted strategies. The rationale behind these strategies is that by exploiting specific molecular modifications associated with cancer, transformed cells may be selectively targeted with less impact on healthy tissues. Such therapies are expected to have lower toxicity than chemotherapy, and may also have the potential to eliminate metastases that may not be eradicated with radiotherapy and/or surgery.13,14 Effective targeted therapy may specifically impede processes on which the cancer is dependent.
Epidermal growth factor receptor (EGFR) is a member of the human epidermal receptor (HER)/Erb-B family of receptor tyrosine kinases that transduce extracellular signals to intracellular responses. EGFR has been implicated in the growth, survival and invasive potential of tumour cells and studies in SCCHN have shown that 80–100% of tumours and many cells adjacent to tumour tissue have abnormal levels of EGFR.15 In one study, aberrant EGFR copy numbers were found in 32 of 134 tumours (24%) in patients with SCCHN and EGFR overexpression was associated with poor prognosis.16
Therefore, much current research has been directed towards targeting abnormal EGFR activity in SCCHN. The EGFR inhibitors currently being studied include monoclonal antibodies, such as cetuximab and panitumumab and low-molecular-weight tyrosine kinase inhibitors, such as gefitinib and erlotinib.1,17 To date, small-scale studies investigating the use of gefitinib or erlotinib in combination with chemoradiotherapy in the treatment of LA SCCHN have shown some encouraging efficacy and safety findings.18–21 Newer dual tyrosine kinase inhibitors are also undergoing clinical trials in SCCHN, of which the furthest developed is lapatinib.1
Cetuximab
Cetuximab (Erbitux®) is an immunoglobulin G1 (IgG1) monoclonal antibody that inhibits ligand binding to the EGFR,22 resulting in suppression of tumour growth, invasion, metastasis, DNA repair and angiogenesis.23,24 Moreover, cetuximab induces cell-mediated cytotoxicity.25 It also enhances the activity of several chemotherapeutic agents, including cisplatin and 5-FU, and sensitises cancer cells to the effects of radiation therapy.26–28Currently cetuximab is the only targeted therapy approved for use in combination with radiotherapy in patients with LA SCCHN. Cetuximab has been approved for this indication in over 70 countries worldwide including, the US, EU, Russia, Australia, South Africa and countries in Central and South America, the Middle East and South East Asia.
Clinical Development
A number of clinical trials have studied cetuximab in LA SCCHN. The use of cetuximab as an adjunct to radiotherapy was first investigated in 2001 in a phase I study29 in which patients were given an initial dose of 100–500mg/m2 followed by weekly doses of 100–250mg/m2. During a median 28-month follow-up period, 13 out of 15 patients showed a complete response and two had partial remissions. Fever, asthenia, transaminase elevation, nausea and skin toxicities (grade 1–2 in most patients) were the most frequent adverse events. It was concluded that cetuximab can be safely given with radiotherapy at an initial dose of 400–500mg/m2 and a weekly dose of 250mg/m2.
The subsequent phase II and III studies investigating cetuximab in LA SCCHN can be divided into two general types: firstly, those using cetuximab concurrently with either radiotherapy or chemoradiotherapy; and secondly, those using cetuximab as part of induction or post-induction therapy regimens. An overview of the subsequent phase II and III studies both completed and ongoing is given in Table 1.
Clinical Studies Including Cetuximab with Concurrent Radiotherapy or Chemoradiotherapy for LA SCCHN Cetuximab with Radiotherapy
To date, the largest trial completed using cetuximab in LA SCCHN was a phase III study in which patients with locoregionally advanced unresectable cancers of the oropharynx, hypopharynx or pharynx received either radiotherapy alone or radiotherapy plus cetuximab at 73 treatment centres.30 Patients were randomised to high-dose radiotherapy alone (≤76.8Gy total dose depending on site and regimen used) (213 patients) or high-dose radiotherapy plus weekly cetuximab (211 patients) at an initial dose of 400mg/m2, followed by weekly 250mg/m2 for the duration of radiotherapy. Three-year data from this study showed that in the radiotherapy plus cetuximab group compared with the group that received radiotherapy alone, there was a 32% reduction in the risk of locoregional progression (hazard ratio [HR] 0.68, 95% confidence interval [CI] 0.52–0.89; p=0.005) and a 26% reduction in the risk of death (HR 0.74, 95% CI 0.57–0.97; p=0.03). Median progression-free survival was 17.1 months in patients receiving radiotherapy with cetuximab and 12.4 months for radiotherapy alone (HR 0.70, 95% CI 0.54–0.90; p=0.006). Patient compliance with radiotherapy and the rates of distant metastases at one and two years were similar in the two groups.
Cetuximab did not increase the incidence of toxic effects normally associated with radiotherapy in SCCHN. After three years of treatment, in the groups receiving radiotherapy alone and radiotherapy plus cetuximab, the more common severe (grade ≥3) adverse events included mucositis (52 and 56%, respectively), acneiform rash (1 and 17%, respectively), radiation dermatitis (18 and 23%, respectively), weight loss (7 and 11%, respectively), xerostomia (3 and 5%, respectively) and dysphagia (30 and 26%, respectively). Compared with the group receiving radiotherapy alone, grade ≥3 adverse events that were reported at a significantly higher frequency in the group receiving cetuximab plus radiotherapy were acneiform rash (1 versus 17%; p<0.001) and infusion reaction (0 versus 3%; p<0.001). A summary of the main findings from this study is given in Table 2.
Recently, five-year data for the Bonner study were published and showed an overall survival of 45.6% in the combined treatment group compared with 36.4% for radiotherapy alone.31 After a median follow-up period of 60 months, the median survival time was 49 months for the combined therapy and 29.3 months for radiotherapy alone (HR for death 0.73, 95% CI 0.56–0.95; p=0.018). Indirect comparisons of the data from the Bonner trial with those of a meta-analysis (including 87 trials, mostly using concomitant chemotherapy, with a patient population of 16,665 and 5.5 years of follow-up) indicate that the survival advantage gained by adding cetuximab to radiotherapy is similar to that produced by adding chemotherapy to radiotherapy.32The pattern of adverse events after five years in the Bonner study was similar to the earlier findings.31 The most commonly reported grade 3–4 adverse events in the radiotherapy alone and radiotherapy plus cetuximab groups were skin reaction (21.2 and 35.1%, respectively), mucositis/stomatitis (51.9 and 55.8%, respectively), dysphagia (29.7 and 26%, respectively), xerostomia (2.8 and 4.8%, respectively), acneiform rash (1.4 and 16.8%, respectively) and infusion reaction (0 and 2.9%, respectively).
Cetuximab with Chemoradiotherapy
An ongoing single-arm phase II study in Philadelphia, US, using cetuximab concurrently with chemoradiotherapy, is investigating the use of cetuximab added to a standard chemoradiotherapy regimen.33 The study recruited 69 patients with LA SCCHN (mostly stage IV disease) who received initial cetuximab (400mg/m2) followed by weekly doses (250mg/m2) in combination with radiotherapy (70Gy) and cisplatin (75mg/m2). Preliminary results demonstrated that 23% of patients experienced a complete response, 25% had a partial response, 31% had stable disease and 5% showed disease progression. There was one case of grade 5 neutropenic fever that was attributed to treatment (although the publication did not specify which component of the treatment was considered to be the specific cause). Among 65 evaluable patients in this study, 97% showed some form of grade >3 toxicity, including neutropenia (26%), fatigue (23%), acneiform rash (28%) and radiation dermatitis (15%). Other grade 3 or higher events included mucositis (54%) and hyponatraemia (20%). Overall, these results indicated that weekly cetuximab with concurrent cisplatin and full-dose radiotherapy is a potential treatment for otherwise fit patients with unresectable LA SCCHN. The authors commented that longer follow-up is needed to determine locoregional control and survival end-points.
In another study,34 22 patients with LA SCCHN at a treatment centre in New York, US, received concomitant boost radiotherapy (1.8Gy/day in weeks one to six with a 1.6Gy/day boost, to a total dose of 70Gy) with cisplatin 100mg/m2 in weeks one and four and cetuximab 400mg/m2 in week one, followed by 250mg/m2 in weeks two to 10. The three-year overall survival rate, progression-free survival and locoregional control rates were 76, 56 and 71%, respectively. The toxicities were typical of those expected with definitive concurrent cisplatin and delayed, accelerated radiotherapy for SCCHN.34 Common grade 3 or 4 toxicities included mucositis, skin toxicity, nausea, vomiting, fever, dehydration, metabolic abnormalities and constipation. This study, however, was stopped due to two patient deaths (pneumonia and unknown cause), and three events from which the patients recovered (myocardial infarction, bacteraemia and atrial fibrillation). There was no indication that cetuximab was the cause of these events.
A small single-arm phase II study in Italy showed the benefit of combining weekly cetuximab with a regimen of alternating radiotherapy and chemotherapy (AlteRCC trial).35 Radiotherapy (2Gy/day to a total dose of 70Gy) was administered as a single daily fraction, from days one to five, in weeks two and three, five and six and eight to 10. Chemotherapy (cisplatin 20mg/m2/day and bolus 5-FU 200mg/m2/day) was delivered daily, on days one to five of weeks one, four and seven. In addition to chemoradiation, cetuximab was delivered weekly, on day one of each week, at an initial dose of 400mg/m2 (in week one) followed by a dose of 250mg/m2 (in weeks two to 10). After three months of treatment, in 16 evaluable patients, there was a complete response in 11 patients and a partial response in five patients. Two of the patients with partial responses received salvage surgery to make them disease-free. After a maximum follow-up of 15 months, 16/20 patients were alive and 16/20 were progression-free. Toxicity was evaluable in 20 patients. Grade 3 or 4 toxicities included diarrhoea (20%), febrile neutropenia (20%), hypomagnesaemia (10%) and mucositis (60%). Skin toxicity, starting as desquamating moist dermatitis and confined at the irradiated field, occurred in 18 patients.
Two large-scale phase III trials using concurrent cetuximab with chemoradiotherapy regimens for treating unresectable tumours are currently in progress. One of these, the Radiation Therapy Oncology Group (RTOG) 0522 study (NCT00265941),36 has completed the planned recruitment of 945 patients with LA SCCHN, at multiple centres in the US. Patients were randomised to receive concurrent cisplatin 100mg/m2 and radiotherapy (1.8Gy/day in weeks one to six with a boost of 1.5Gy/day to a total dose of 72Gy) with or without cetuximab (initial dose of 400mg/m2 and thereafter 250mg/m2/week). The study aims to compare the effect of the two regimens on disease-free survival and overall survival and to investigate any associations between outcome parameters and positron emission tomography (PET) scan findings of the tumour sites. In the other ongoing study, patients with LA SCCHN at treatment centres in France (planned recruitment of 406 patients) are being randomised to receive cetuximab (initial 400mg/m2 dose followed by 250mg/m2/week) plus radiotherapy (70Gy over seven weeks) with or without carboplatin (70mg/m2/day) and 5-FU (600mg/m2/day) (NCT00609284).37 This study aims to evaluate the effect of adding cetuximab to an existing chemoradiotherapy regimen compared with cetuximab and radiotherapy on progression-free survival and locoregional control over three years in patients with LA SCCHN (stages III–IV).Clinical Studies Using Cetuximab as Induction and/or Post-induction Therapy for LA SCCHN Cetuximab in Post-induction Therapy Only
The Radiotherapy With Cisplatin Versus Radiotherapy With Cetuximab After Induction Chemotherapy for Larynx Preservation (TREMPLIN) study was a randomised phase II trial that was conducted in France.38 Patients were required to be previously untreated and to have resectable LA laryngeal or hypopharyngeal tumours. Induction chemotherapy consisted of three cycles of docetaxel, cisplatin (both 75mg/m2) and 5-FU (750mg/m2/day). Patients showing ≥50% response were randomised to receive radiotherapy (70Gy) with either cisplatin (100mg/m2) (arm A) or cetuximab (400mg/m2 initial dose followed by 250mg/m2 weekly) (arm B). Preliminary results showed that out of 115 patients randomised, after three months of treatment there was no significant difference in larynx preservation (the primary end-point of the trial) between the two groups (93% in arm A and 96% in arm B). Induction therapy followed by radiotherapy with concurrent cetuximab appeared better tolerated than induction therapy followed by radiotherapy with concurrent cisplatin. It was noteworthy that among patients who received cisplatin and radiotherapy in this study (arm A), 16% had permanent renal failure, which would preclude them from receiving any further platinum-containing compounds as part of subsequent treatment regimens.38
Cetuximab in Induction Therapy or in Induction and Post-induction Therapy
A trial conducted in Sweden included 68 patients with LA SCCHN (56 with stage IV disease) who received two cycles of docetaxel and cisplatin (both 75mg/m2) and 5-FU (1,000mg/m2) followed by radiotherapy (68Gy/4.5 weeks) and cetuximab (initial dose of 400mg/m2 and 250mg/m2/week thereafter).39 Thirty patients were followed beyond six weeks and were evaluated for response and early toxicity. Preliminary results showed that after treatment with docetaxel, cisplatin and 5-FU there was a complete response in one patient, a partial response in 15 patients and stable disease in 14 patients. After radiotherapy with cetuximab there was a complete response in 10 patients, a partial response in 18 patients, and stable disease in one patient. Dermatitis in the irradiated neck (with accelerated fractionation) was problematic but did not interrupt treatment and healed rapidly. It was concluded that the TPF combination followed by radiotherapy concomitant with cetuximab is feasible, with manageable toxicities.39
Several trials have evaluated the use of cetuximab during both induction and post-induction therapy in combination with radiotherapy and chemotherapy medications. An example is a phase II single-arm study of 50 patients with unresectable LA SCCHN conducted in Spain.40 Induction treatment comprised docetaxel 75mg/m2 on day one, cisplatin 75mg/m2 on day one, 5-FU 750mg/m2 on days one to five and cetuximab 250mg/m2 on days one, eight and 15 (initial dose 400mg/m2 on cycle one, day one) and repeated every 21 days for four cycles. Patients then received accelerated radiotherapy with a concomitant boost (69.9Gy) and cetuximab 250mg/m2 weekly. The results showed that the addition of cetuximab to the induction regimen resulted in a good response rate (mainly a complete response). After two and four cycles of treatment, a complete response was seen in six (12%) and 10 (20%) patients, respectively, and a partial response was seen in 31 (62%) and 25 (50%) patients, respectively. Serious grade 3/4 adverse events included: neutropenia (24%), neutropenic fever (20%), diarrhoea (12%) and infection (6%). Grade 3 adverse events included mucositis (6%), renal failure (4%), asthenia (4%), rash (4%) and hypotension (4%). In addition, there were two adverse-event-related deaths (hepatic insufficiency, in a patient with previously unknown liver cirrhosis, and febrile neutropenia). The authors concluded that the cetuximab/chemotherapy combination should be given to LA SCCHN patients who have good performance status with specialised support.40
In another phase II study, 74 patients with resectable LA SCCHN at medical centres in the US received cetuximab as part of both induction and post-induction therapy.41 Patients received induction chemotherapy (weekly cetuximab 250mg/m2, paclitaxel 90mg/m2, carboplatin [area under the curve (AUC)] = 2). Patients showing a clinical response had a staging primary site biopsy at week eight; patients with positive biopsy (or persistent tumour) had a restaging biopsy at week 14 after chemoradiation (cetuximab 250mg/m2, paclitaxel 30mg/m2, carboplatin [AUC=1] and 50Gy). If the primary site biopsy was negative, patients completed their radiotherapy (68–72Gy) and continued chemotherapy (cetuximab 250mg/m2, paclitaxel 30mg/m2, carboplatin [AUC = 1]). If the primary site biopsy was positive at 14 weeks, salvage surgery was required. At week eight, 26 out of 40 patients (65%) who had a primary-site restaging biopsy had a complete pathological response. At week 14, 28 out of 28 patients (100%) who had a restaging biopsy had a complete pathological response. Grade 3 and 4 toxicities in this study included acneiform rash (12%), leukopenia/neutropenia (32/24%), dysphagia (29%) and stomatitis (82%).41 There was one grade 5 adverse event (death from encephalopathy). These results showed that the addition of cetuximab to both induction therapy and post-induction therapy is potentially effective and well tolerated. Long-term progression data or this study are yet to be published.Quality of Life
LA SCCHN has an extremely deleterious effect on quality of life (QoL). Symptoms occurring at this stage of disease can include difficulties swallowing and speaking, and surgical procedures to remove tumours in LA SCCHN can also be disfiguring.42,43 These symptoms often severely inhibit social interaction and diminish self-esteem. The treatments used for LA SCCHN can also negatively affect QoL since they can result in acute episodes of mucositis44 and xerostomia.45 Chemoradiotherapy has well-documented toxic effects including nausea and vomiting, diarrhoea, asthenia, hair loss and may necessitate the use of a feeding tube. These treatments therefore have the potential to substantially diminish QoL and may cause many patients to prematurely stop taking medication, resulting in treatment failure.46,47
A QoL analysis of a phase III trial comparing high-dose radiotherapy with or without concomitant cetuximab48 concluded that the addition of cetuximab to radiotherapy significantly improved locoregional control and increased overall survival without having any significant effects on QoL. In this study, compliance with completing QoL questionnaires was high and the data are therefore considered to be reliable. For both treatment groups, the scores for most of the QoL scales decreased during treatment, indicating a worsening of symptoms, but then returned to scores comparable with baseline levels by month 12. The European Organisation for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire C30 (EORTC QLQ-C30) and EORTC QLQ Head and Neck (H&N) Cancer-Specific Module were used in this study and would be expected to indicate any adverse effect of the acne-like rash on patient QoL. However, there was no difference between the treatment groups in QoL. The lack of difference between radiotherapy alone and radiotherapy plus cetuximab was particularly notable for global health status/QoL, social functioning, social eating and social contact. Pre-treatment global health status/QoL was identified as a significant prognostic variable in this study population. Parameters Cetuximab in the Treatment of Locally Advanced Head and Neck Cancer showing no difference between treatment groups for change from baseline to worst post-baseline scores were fatigue, nausea and vomiting, pain QLQ-C30, pain QLQ-H&N35, swallowing, sensory problems, speech problems, trouble with social eating, trouble with social contact and lower sexuality. The EORTC QLQ C30 global health status/QoL scores during the one-year follow-up period are plotted in Figure 1 and show the similarities in QoL status between the two treatment groups. The authors commented that the findings support the argument that adding cetuximab to radiotherapy is an attractive therapeutic option for patients with LA SCCHN.48
Cost-effectiveness
While the clinical evidence indicates a central role for the use of cetuximab in SCCHN, the cost of treatment needs to be assessed against the benefits. An analysis of the data from the Bonner phase III trial found that the additional cost per quality-adjusted life-year for patients receiving combined radiotherapy and cetuximab compared with radiotherapy alone was in the range of €7,538–10,836 and thus concluded that the combination of cetuximab and radiotherapy in SCCHN was cost-effective.49 Another analysis of this study indicated that the incremental cost-effectiveness ratio of cetuximab plus radiotherapy compared with radiotherapy alone in LA SCCHN was £6,390 (approximately €7,668 at an exchange rate of €1.20 per £1) per additional quality-adjusted life-year. This suggested that cetuximab plus radiotherapy is cost-effective compared with radiotherapy alone using assumptions based on a cost-effectiveness threshold of £20,000 (approximately €24,000).50Conclusions and Future Studies
Cetuximab is a significant advance in the treatment of LA SCCHN; its addition to radiotherapy confers significant benefits compared with radiotherapy alone. Based on the data from the Bonner phase III trial, the latest European Society for Medical Oncology (ESMO) guidelines recommend the use of cetuximab in combination with radiotherapy in LA SCCHN.51 The addition of cetuximab to chemotherapy regimens may also improve efficacy both when cetuximab is included in induction therapy and when it is included in post-induction therapy regimens. Completed clinical trials have shown that there is generally little difference in adverse event frequencies when cetuximab is added to chemotherapy or chemoradiotherapy regimens. The most commonly reported side effect of cetuximab in the treatment of various malignancies is an acne-like skin rash; however, this rash can be managed and may correlate with a good response to therapy.31,52,53 Ongoing phase III clinical studies will provide data on the efficacy and safety of the combination of cetuximab and concurrent chemoradiotherapy. Another emerging aspect of SCCHN is that certain tumour types have been shown to have an aetiology involving human papillomavirus (HPV); oropharyngeal tumours, in particular, are more often HPV-positive than other head and neck neoplasms.54–57 Patients with HPV-positive oropharyngeal tumours are significantly more likely to respond to chemoradiotherapy than those with HPV-negative tumours58–61 and this is also true of patients with tumours at other head and neck sites.62
The favourable prognosis of patients with HPV-positive SCCHN may mean that concurrent chemotherapy is too aggressive for such patients. Given the excellent efficacy and tolerability data for cetuximab plus radiotherapy in LA SCCHN,30,31 this combination may be a suitable option for the treatment of HPV-positive SCCHN, although this would need to be investigated in appropriately designed clinical studies. The results of the clinical trials published to date raise several additional areas for future research, including the most effective ways of using cetuximab (sequential or simultaneous radiotherapy with chemotherapy) and the optimum choice of other chemotherapy agents to be combined with cetuximab. The data will help to determine which regimen is most effective in improving prognosis in terms of survival and disease progression, and which regimens reduce or eliminate permanent toxicity and safety concerns and those which negatively impact on QoL. Future research should attempt to identify biomarkers of response to EFGR-targeted therapies in SCCHN. Such studies, combined with ongoing clinical trials, should further improve the therapeutic benefits for patients with LA SCCHN and help target treatments. ■