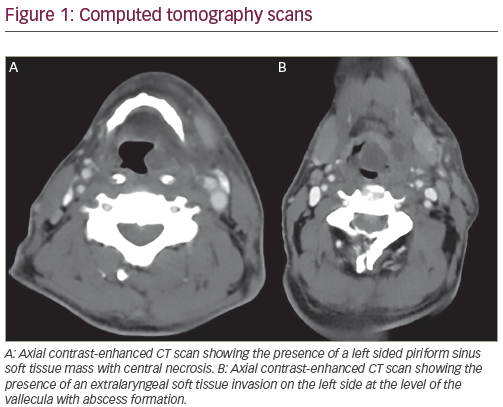
Squamous cell cancer of the head and neck (SCCHN) accounts for approximately 3% of adult malignancies in the US and is the seventh leading cause of cancer death worldwide.1,2 SCCHN has been classified into four stages:
• stages I/II are classified as early disease;
• stages III/IV M0 are divided into resectable and non-resectable
disease; and
• stage IV M1 defines recurrent or metastatic disease.
Squamous cell cancer of the head and neck (SCCHN) accounts for approximately 3% of adult malignancies in the US and is the seventh leading cause of cancer death worldwide.1,2 SCCHN has been classified into four stages:
• stages I/II are classified as early disease;
• stages III/IV M0 are divided into resectable and non-resectable
disease; and
• stage IV M1 defines recurrent or metastatic disease.
Early-stage disease is generally cured through radiotherapy courses and/or surgery. However, there have been an increasing number of newly diagnosed cases of advanced-stage SCCHN.3 The main goal of treatment for locoregional SCCHN at stage III/IV M0 is organ preservation and cure. Locoregional failure is the most common pattern of disease progression, as distant metastases are rarely identified at initial diagnosis.
Over the past 10 years, research has focused on the treatment of locoregional SCCHN with various chemotherapy regimens, altered radiotherapy and the addition of newly targeted agents. Standard therapy for advanced SCCHN is the use of chemotherapy with a platinum compound, usually cisplatin. SCCHN is chemosensitive and has a response rate of approximately 60–90%.4 However, chemotherapy commonly results in severe mucositis, which is a debilitating condition that frequently requires the insertion of a feeding tube to maintain a desirable nutritional profile. In addition, the introduction of chemotherapy has not been linked to a prolonged survival rate.5
Epidermal Growth Factor Receptor
Epidermal growth factor receptor (EGFR) is overexpressed in approximately 90% of SCCHN tumours.6 An increase in the expression of EGFR has also been noted as an early event in the progression of SCCHN and has been seen in pre-malignant lesions.7 A lower level of EGFR has been associated with an improved disease-free interval after treatment with radiation and induction chemotherapy in patients with laryngeal and hypopharyngeal cancers.8 Hitt and colleagues examined biopsies in SCCHN and reported that a higher level of EGFR was linked with a decrease in disease-free interval and a shorter time to treatment failure.9 Moreover, the expression of EGFR has been identified as an independent predictor of locoregional relapse, but showed no correlation to the risk of distant metastasis.7
Tyrosine Kinase Inhibitors
Gefitinib was the first tyrosine kinase inhibitor (TKI) to be approved by the US Food and Drug Administration (FDA) for use in non-small-cell lung cancer (NSCLC). Gefitinib is a selective inhibitor for the EGFR tyrosine kinase domain, which is responsible for activating antiapoptotic pathways.10 A phase II trial demonstrated that gefitinib was well tolerated and had positive activity in patients with recurrent or metastatic SCCHN.11 Gefitinib was administered in a once-daily dose of 500mg, which gave a median time to progression of 3.4 months and a median survival time of 8.1 months. Diarrhoea was reported in three patients treated with gefitinib, and skin toxicity was seen as an indication of patient response and improved survival.
A second phase II trial examined gefitinib integration in a concurrent chemoradiation regimen in recurrent SCCHN patients followed by a 500mg/day dose of gefitinib alone in an adjuvant setting. The authors of the study concluded that the drug was effective at prolonging time to progression and survival time in these patients.12 An alternative dose regimen of 250mg/day in patients with advanced SCCHN showed partial or no activity. This suggests a dose-dependent activity of gefitinib in SCCHN.
A phase I/II trial studied the effect of induction chemotherapy alongside gefitinib followed by concurrent chemotherapy or radiation therapy in patients with locally advanced SCCHN.13 Eleven of the 34 patients had a complete response to gefitinib treatment and 18 patients had a partial response; an overall response rate of 85% was reported. One year after the treatment regimen the survival rate was 86%, with a progression-free survival of 68%. No additional toxicity was found with the use of gefitinib. The study concluded that TKI could be added to chemotherapy to improve the activity of the treatment, with an acceptable toxicity profile.
A second TKI, erlotinib, has also recently been approved by the FDA for use in NSCLC, as well as in pancreatic cancer. A dose of 150mg/day was established in a phase I trial using erlotinib in combination with chemotherapy with cisplatin and radiotherapy for locally advanced SCCHN.14 In the phase II part of the study, 11 of the 13 participants achieved complete response to the treatment regimen, and the authors concluded that the protocol was well tolerated and active in SCCHN. Phase III trials are needed to compare standard chemotherapy regimens in combination with TKIs or with a placebo for locally advanced SCCHN.
Monoclonal Antibodies
The second strategy to decrease expression of EGFR involves the use of monoclonal antibodies that target the extracellular domain of the receptor. Binding of a monoclonal antibody to the receptor prevents activation by ligand binding through competitive inhibition; this also results in the degradation and internalisation of the EGFR, downregulating its expression. The most well characterised and well tested monoclonal antibody used in SCCHN is cetuximab. In 2000, cetuximab used concurrently with radiation was demonstrated to reduce cell proliferation in cell lines regardless of high expression of EGFR in the line.15 These promising pre-clinical results led to further clinical investigation with cetuximab.
A preliminary study of cetuximab in addition to radiotherapy in patients with locoregional advanced SCCHN demonstrated a complete or partial regression.16 Several phase II trials have been conducted with cetuximab, evaluating its potential alongside different treatment regimens. A phase II multicentre study of cetuximab in combination with chemotherapy with a platinum-based product in metastatic and recurrent SCCHN patients demonstrated a recovery rate of 10% and a disease control rate of 53%.17 The most common adverse event was a skin rash; the drug was otherwise well tolerated.
A second phase II trial evaluated cetuximab in combination with cisplatin in patients with metastatic and recurrent SCCHN.18 Patients were initially started on a three-week course of cisplatin and then treated again with cisplatin if the first cycle achieved partial or complete response. Patients who remained stable or whose cancer progressed were transferred to treatment with cisplatin in combination with cetuximab. Cetuximab treatment resulted in a skin rash, although it did not exacerbate cisplatin toxicity. However, response to the combined treatment was minimal and the study concluded that the relevance of cetuximab would be better defined in a single-agent trial.
In 2005, a retrospective study was conducted to compare cetuximab in combination with platinum-based chemotherapy versus cetuximab monotherapy, using historical controls in patients with metastatic or recurrent SCCHN whose initial chemotherapy course had failed.19 The median survival time for cetuximab monotherapy was 5.9 months, which represents a large increase compared with historical controls, with a median survival time of 2.5 months. An acne-like rash appeared in 69 and 72% of patients on combined therapy and monotherapy with cetuximab, respectively.
To date, several phase III trials have been carried out to evaluate the potential of cetuximab in SCCHN. An international phase III trial compared radiation therapy with or without administration of cetuximab for locoregional advanced SCCHN.20 Patients receiving radiation therapy alongside cetuximab showed more favourable responses to treatment and had a median two-year survival rate of 62% and a three-year survival rate of 57%; this compared with 55 and 44%, respectively, with radiation therapy alone. However, the monotherapy increased the risk of skin conditions, although cetuximab monotherapy had no effect on the incidence of severe mucositis. This result was confirmed by another phase III trial comparing monotherapy and combination therapy with cetuximab, which also concluded that the addition of cetuximab to radiotherapy improved locoregional control and reduced mortality without increasing the common toxic effects of radiotherapy.21
The addition of cetuximab to radiotherapy provides an additional option for patients with locoregional advanced SCCHN and represents a new foundation for the future development of treatment for the disease. Future trials need to compare this new combination with the older, more traditional regimen of chemotherapy and radiation therapy. In the absence of a direct comparison, physicians should enter patients into regimens after an examination of the benefits and side effects of each on an individual basis.
Quality of Life Benefits of Cetuximab
Quality of life is paramount in advanced locoregional SCCHN patients. Quality of life assessments were performed in a study comparing treatment with radiotherapy alone versus radiotherapy in combination with cetuximab.22 The study used two questionnaires – the European Organisation for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire C30 and the Quality of Life Questionnaire, Head and Neck Module – to address the symptom burden and its effect on physical and functional wellbeing and emotional and social interactions. The two treatment regimens provided similar results over a 12-month period and both showed a decrease in quality of life as treatment progressed. The greatest impact on treatment was seen in loss of appetite, fatigue, swallowing difficulties and sensory problems; these symptoms improved after eight to 12 months.
A significant difference in locoregional control has been noted between treated and untreated patients, and it can be postulated that the symptoms in uncontrolled disease would result in a lower quality of life compared with that of patients receiving treatment.21 The conclusion of the study was that patients receiving radiotherapy plus cetuximab had no worse quality of life than those administered radiotherapy alone. Advances in multimodality therapies should seek to improve quality of life in parallel with improvements in survival.
Epidermal Growth Factor Receptor Expression and Response to Treatment
A study comparing treatment for SCCHN with cisplatin alone versus cisplatin in combination with cetuximab demonstrated that the treatment effect of cetuximab was greater in patients presenting with moderate EGFR expression rather than in those with high EGFR expression.23 This may indicate that patients with high EGFR expression are underdosed with current regimens of cetuximab or that ligand-independent signalling is more important in this subset of patients, who might be more effectively inhibited using a quinazoline.
The difference between responses to treatment in the two groups may reflect differences in EGFR expression and function between different tumour types.24 Future studies will need to investigate the relevance of a high EGFR expression to pinpoint mechanisms of resistance and thus establish whether a higher dose of cetuximab, a specific combination of TKIs or the combination of a TKI and another agent would be more effective in this patient population.
Summary
Over the past 10 years, research has focused on the treatment of locoregional SCCHN with various regimens of chemotherapy, altered radiotherapy and the addition of new targeted agents. EGFR is overexpressed in approximately 90% of SCCHN tumours and therefore is considered to be an important target for therapy. Currently, two strategies have been explored to target EGFR: TKIs and monoclonal antibodies. Gefitinib and erlotinib are both TKIs that have been demonstrated to improve chemotherapy when used in combination with existing regimens. Cetuximab is a monoclonal antibody that has been evaluated in several trials as monotherapy and in combination with radiotherapy and chemotherapy for locoregional SCCHN. Cetuximab has performed well in these trials, improving time to progression and survival. The main adverse effect is skin acne. The cost-effectiveness and optimal dose regimen for cetuximab have yet to be determined. Ongoing and future trials with cetuximab will provide important data and establish how it will benefit future treatment for SCCHN. ■