Adverse Event Management in Anaplastic Lymphoma Kinase-positive Non-small Cell Lung Cancer
Abstract
Overview
The development of oncogene-directed targeted therapies represents a new paradigm in the treatment of non-small cell lung cancer (NSCLC), offering improved outcomes compared with chemotherapy. Rearrangements of the anaplastic lymphoma kinase (ALK) gene are major oncogenic drivers in a subset of NSCLC patients. Since its launch in 2011, the ALK inhibitor crizotinib has become the standard of care in ALK-positive NSCLC, but resistance inevitably develops. Ceritinib and alectinib have received regulatory approval: the former in Europe, US and elsewhere in the world, the latter in Japan. ALK inhibitors target multiple pathways, and may therefore be associated with a wide range of adverse events (AEs), including gastrointestinal AEs, hepatotoxicity and, in the case of crizotinib and ceritinib, cardiac effects. While the majority of these AEs are reversible, manageable and not severe, it is important that both physician and patients are aware of toxicities to ensure prompt treatment. This article discusses the management of AEs in patients receiving currently approved ALK inhibitors, including treatment, regular monitoring, drug discontinuation or dose reduction and physician/patient education. Proactive management of AEs enhances patient quality of life and optimises the therapeutic index of these agents.
Keywords
Alectinib, ceritinib, crizotinib, ALK-positive non-small cell lung cancer
Article Information
Disclosure
Christian Rolfo has served on the Novartis International Speakers Bureau. Solange Peters and Ignacio Gil-Bazo have no conflicts of interest to declare.
Correspondence
Christian Rolfo, Associate Professor, University Hospital Antwerp Oncology, Head of Phase I Early Clinical Trials Unit, Antwerp, Belgium. E: Christian.Rolfo@uza.be
Support
The publication of this article was supported by Novartis, who were given the opportunity to review the article for scientific accuracy before submission. Any resulting changes were made at the author’s discretion.
Access
This article is published under the Creative Commons Attribution Noncommercial License, which permits any non-commercial use, distribution, adaptation and reproduction provided the original author(s) and source are given appropriate credit.
Acknowledgements
Medial writing assistance was provided by Katrina Mountfort at Touch Medical Media and funded by Novartis.
Received
2015-05-12T00:00:00
Further Resources

Trending Topic
It is with great pleasure that we present the latest edition of touchREVIEWS in Oncology & Haematology. This issue highlights the remarkable progress and innovation shaping the fields of oncology and haematology, featuring articles that delve into both emerging therapies and the evolving understanding of complex malignancies. We open with an editorial by Mohammad Ammad […]
Related Content in Lung Cancer
Highlights Immunotherapy, especially combinatory immunotherapy, has shown promise with prolonged survival for patients with advanced mesothelioma in the first-line setting (see the sections on ‘Systemic treatment and immunotherapy debut’ and ‘Randomized immunotherapy trials of mesothelioma’). Histology-based therapy is important to ...

Welcome to the latest issue of touchREVIEWS in Oncology & Haematology. We are honoured to present a series of compelling articles that reflect cutting-edge developments and diverse perspectives in this ever-evolving field. This issue includes a series of editorials and ...

Unmet clinical needs for patients with advanced epidermal growth factor receptor-mutated non–small-cell lung cancer Epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors (TKIs) have become the standard first-line therapy for patients with advanced non–small-cell lung cancer (NSCLC) harbouring ...

Accurately detecting lung tumours and their margins is important for disease outcomes.1,2 However, detection is challenging due to the use of minimally invasive surgery and current localization techniques, such as computed tomography (CT)-guided and endobronchial interventions, which add significantly ...
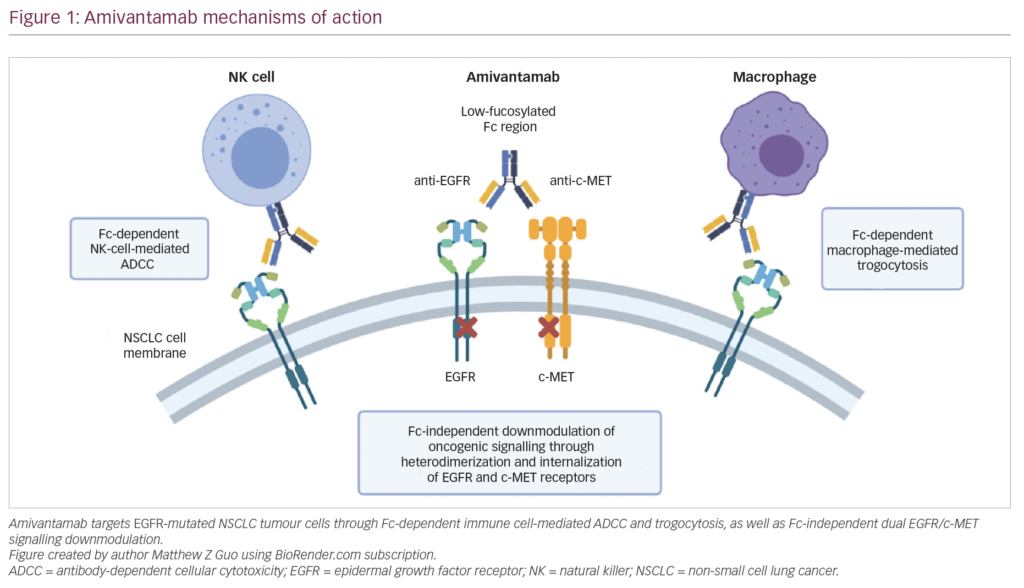
Cancer treatment has expanded rapidly in recent years as advancements in the fields of tumour biology and molecular diagnostics have informed the development of targeted therapies, improving survival in patients with oncogene-addicted cancers with therapeutically relevant molecular lesions. Osimertinib has ...
The treatment of patients with non-small cell lung cancer (NSCLC) has seen significant advances in the past decade, with the availability of multiple targeted therapy agents for oncogenic-driven non-squamous NSCLC and the advent of immunotherapy that has completely revolutionized the ...
Advanced non-small cell lung carcinoma (NSCLC) treatment paradigms have evolved during the past decade. Identification of tumor-specific molecular alteration in cancer driver genes has led to the development of targeted therapies.1–3 Most of the tumors harboring such alterations are sensitive ...
Radiation-induced esophagitis, caused by incidental damage to the mucosal lining of the esophagus during radiation therapy, is a common and clinically important toxicity in patients with lung cancer. Esophagitis generally develops 2–3 weeks after initiation of radiation therapy and presents as ...
Historically, chemotherapy represented the only available treatment for patients with advanced non-small-cell lung cancer (NSCLC) who did not show any target molecular driver, such as epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK) or the receptor tyrosine kinase ROS1. ...

At initial diagnosis of metastatic disease, central nervous system (CNS) metastases are present in 22–33% of patients with anaplastic lymphoma kinase (ALK)-rearranged (ALK+) non-small cell lung cancer (NSCLC). After crizotinib failure, the incidence can reach up to 70%, compared with 40% in ...
Non-small cell lung cancer (NSCLC) remains a world-wide health issue, accounting for 85% of all lung cancers, of which there were an estimated 2.1 million new cases and 1.76 million deaths in 2018; equivalent to 11.6% of the global cancer burden.1,2 This incidence is expected ...

Welcome to the fall edition of Oncology and Hematology Review (US), which features a wide variety of articles covering topics of interest to oncologists and hematologists, as well as the wider medical community. We begin with some expert interviews, which ...
Latest articles videos and clinical updates - straight to your inbox
Log into your Touch Account
Earn and track your CME credits on the go, save articles for later, and follow the latest congress coverage.
Register now for FREE Access
Register for free to hear about the latest expert-led education, peer-reviewed articles, conference highlights, and innovative CME activities.
Sign up with an Email
Or use a Social Account.
This Functionality is for
Members Only
Explore the latest in medical education and stay current in your field. Create a free account to track your learning.

