Increased expression of intracellular annexin I is seen in bronchial epithelial cells grown in the presence of dexamethasone,9 and secreted annexin I appears to be proteolytically degraded by the human neutrophil elastase to an inactive form.10,11
Annexin I is a critical mediator of apoptosis.12 15 While over-expression of annexin I has been observed in pancreatic,16 breast, and gastric cancers,17 reduced or no expression of annexin I has been reported in prostate and esophageal cancers.18 21 Thus, differential regulation of annexin I in a tissue-specific manner may be associated with the development of cancers in these sites.
Absence of annexin II expression has been reported in two B-cell lymphoma cell lines, Raji and OMABL- 1.22 While annexin II is closely related to annexin I in amino acid identity, its cellular function is clearly different.9 Both annexins I and II are upregulated in pancreatic carcinoma,16 and recent reports have shown absence of both annexins I and II in prostate carcinoma.20 24 Thus, it appears that both annexins I and II may be coordinately regulated. In view of these observations, the expression of annexin I in human B-cell lymphomas and cell lines was investigated in this study.
Methods Cell Culture, Drug Treatment, and Reagents
The human B-cell lymphoma cell lines used in this study are: progenitor B-cell lines (Nalm-6, REH, HPB-Null, PBE-1), B-lymphoblast cell lines (WI-L2,TK-6,DW-10, DHL-16), and Burkitt s lymphoma cell lines (Raji, Ramos, OMA-BL-1, Namalwa). TK-6 is a lymphoblast cell line that is heterozygous for thymidine kinase.TK-6 is a derivative of the WI-L2, a lymphoblast cell line.DW- 10 and WI-L2 are EBV transformed mature B-cell lines. PBE-1 and NALM-6 are both precursor B-cell acute lymphoblastic leukemia cell lines. NALM-6 is an established cell line and PBE1 is a line established shortterm from a patient with acute lymphoblastic leukemia (ALL) at the University of Nebraska Medical Center (please note that a DNA fingerprint analysis25 of over 500 lymphoma-leukemia cell lines indicated that PBE-1 and NALM6 may be identical). DHL-16 is a follicular B-cell lymphoma cell line.26 Human adenoids were used as a source of normal B-cells, and contained >80% B-cells as determined by cell sorting and flow cytometric analysis. In other experiments, normal B-cells were isolated from PBL of a healthy volunteer using the human B-cell isolation kit (Miltenyi Biotec Inc., Auburn, CA) as per manufacturer s guidelines. SW1116, HeLa and 293T cells were used as positive controls in the indicated experiments. Cells were grown in a growth medium consisting of Eagle s minimum essential medium (GIBCO-BRL, Grand Island, NY) supplemented with 10% heat-inactivated fetal bovine serum, L-glutamine (2mM), penicillin (100U/ml) and streptomycin (100µg/ml). All cell lines were determined to be mycoplasma-free by a polymerase chain reaction (PCR)- based mycoplasma detection assay. 5-aza-2 – deoxycytidine (deoxyC, Sigma Chemical Co., St Louis, MO) was freshly prepared in distilled water. Raji and OMA BL-1 cells, growing in T25 flasks, were incubated in one of the following: 3µM or 6µM deoxyC for three days or six days; 2.5 to 10µg/ml lipopolysaccharide from E. coli (Sigma) for 24 hours; or 100µM H2O2 for two to 24 hours. Genomic DNA, total cellular ribonucleic acid (RNA), and protein were extracted from cells using previously published procedures.22,27 Absence of Annexin I Expression in B-cell Non-Hodgkin s Lymphomas and Cell Lines
Tissue Sections
Formalin-fixed, paraffin-embedded tissues representing normal tonsil (n=2), diffuse large cell non-cleaved Bcell lymphoma (n=2), small lymphocytic B-cell lymphoma (n=2), and follicular mixed B-cell lymphoma (n=2) were generously made available for these studies by Dr Dennis Weisenburger, from the Department of Pathology and Microbiology, University of Nebraska Medical Center, Omaha, NE. Sequential serial sections prepared from these specimens were used in immunohistochemical analysis.
Antibodies
Polyclonal rabbit anti-human annexin I antisera (a646) was a gift from Dr Blake Pepinsky (Biogen, Boston,MA). Polyclonal anti-human phosphoglycerate kinase (PGK) antiserum (a35) was prepared as described previously.28 The mouse monoclonal anti-CD20 antibody was from Beckman/Coulter Inc. (Westbrook, MA).
Gel Electrophoresis and Immunoblotting
Protein extracts were prepared from cells using protein lysis buffer containing 50mM Tris-HCl pH7.5, 2.0mM PMSF, 5.0mM iodoacetamide, 5.0mM EDTA, 150mM NaCl, 0.5% NP-40, and 0.5% Mega- 9. Protease inhibitors were added just prior to use (leupeptin at 1?g/ml and pepstatin at 2?g/ml).Total amounts of protein extracts were quantitated using Bio-Rad protein assay (Pierce, Rockford, IL). A total of 50?g of protein from each extract was separated on a 12% SDS-PAGE. After electrophoresis, proteins were transferred to polyvinylidene fluoride (PVDF) membrane (Millipore, Bedford, MA).The membranes were blocked in 1x TTBS with 7% powdered milk overnight at 4ºC and then probed for one hour with rabbit polyclonal anti-human annexin I (a646) at 1:500 dilution and rabbit polyclonal anti-human PGK (a35) at 1:1000 dilution. Anti-rabbit horseradish peroxidase (HRP) (Promega, Madison, WI) at a dilution of 1:5000 was used for one hour at room temperature to detect antigen–antibody complexes. Membranes were developed using ECL+ (Amersham Pharmacia Biotech, Arlington Heights, IL). For quantitation of the immunoreactive band, the blot was scanned on a laser densitometer (Molecular Dynamics, Sunnyvale, CA).
RNA Isolationand Polymerase Chain Reaction Analysis
Total RNA was isolated from the cell monolayers or tissue samples using RNeasy mini kit (Qiagen Inc., Valencia, CA) or by TRIzol method (GIBCO/BRL). Annexin I message was characterized by PCR using the enhanced Avian RT-PCR kit (Sigma).Total RNA (5?g) was added to each RT reaction using random nonamer primers. The cDNA products were amplified using annexin I specific primers to give a 522 bp product.The primers designed for amplification were:
Annexin I forward: 5’-GCAAGAAGGTAGAGATAAAG-3’
Annexin I reverse: 5’-ATCTCTCTTCAGTTCCTCTC-3’
For verification of the integrity of the RNA samples, and as a control in all the RT-PCR analyses, the expression of the a-tubulin gene was examined. The primers A-tu1 (5’-AAG AAA TCC AAG CTG GAG TTC-3’) and A-tu2 (5’-GTT GGT CTG GAA TTC TGT CAG-3’), specific for the a-tubulin gene, generated a 300bp PCR product corresponding to the a-tubulin gene. Southern Blot Analysis
Genomic DNA was isolated from Raji, HPB-null, Nalm-6 and HeLa cell lines, and subjected to restriction endonuclease digestion. Southern blot analysis29 of genomic DNA was performed using radiolabeled 522bp annexin I cDNA product from the PCR reaction as described above.
Immunohistochemistry
Standard methods were used to prepare sequential 8?M sections of paraffin-embedded tissues, which were mounted on polylysine-coated glass slides.Tissues were cleared and rehydrated with two 10-minute rinses in a ready-to-use tissue deparaffinization solution (Biogenex Inc. San Ramon, CA). Sections were then stained by the immunoperoxidase method using the ready-to-use Vectastain Quick kit (Vector Labs, Burlingame, CA). Primary antibodies used included rabbit polyclonal anti-human annexin I antibody and mouse monoclonal anti-human CD20 antibody.
Results
Expression of Annexin I in Human B – cell Lymphoma Cell Lines
Annexin I protein levels were measured by immunoblot analysis in various B-cell lymphoma cells along with other cell lines known to express annexin I. Extracts made from adenoid cells (containing >80% B-cells) were used as control normal cells, and HeLa cell extracts were used as positive controls.As expected HeLa cells (lane 1) expressed high levels of annexin I, and adenoids (lane 13) expressed annexin I but at a much reduced level when compared with HeLa cells. Annexin I protein was undetectable in any of the progenitor B-cell lines (Nalm-6, REH, HPB-Null, PBE-1) and Burkitt s lymphoma cell lines (Raji, Ramos, OMA-BL-1, Namalwa). Among the Blymphoblast cell lines (WI-L2, TK-6, DW-10), only WI-L2 expressed annexin I, while its derivative TK-6 did not express annexin I. Using a different source of normal B-cells (obtained from PBL of a healthy volunteer), annexin I expression in normal B-cells was once again confirmed. In addition, significantly reduced expression of annexin I in the Burkitt s lymphoma cell line Raji and the follicular lymphoma cell line DHL-16 was found, when compared with the normal B-cells as evidenced by the levels of PGK in the same blot.The expression of 3-PGK included as an internal control on the blots was seen in all the cell lines except the adenoids. Expression of proliferating cell nuclear antigen and the 70 and 90kDa subunits of Ku autoantigen were unaffected in these cell lines (data not shown), indicating a specific lack of expression of annexin I.
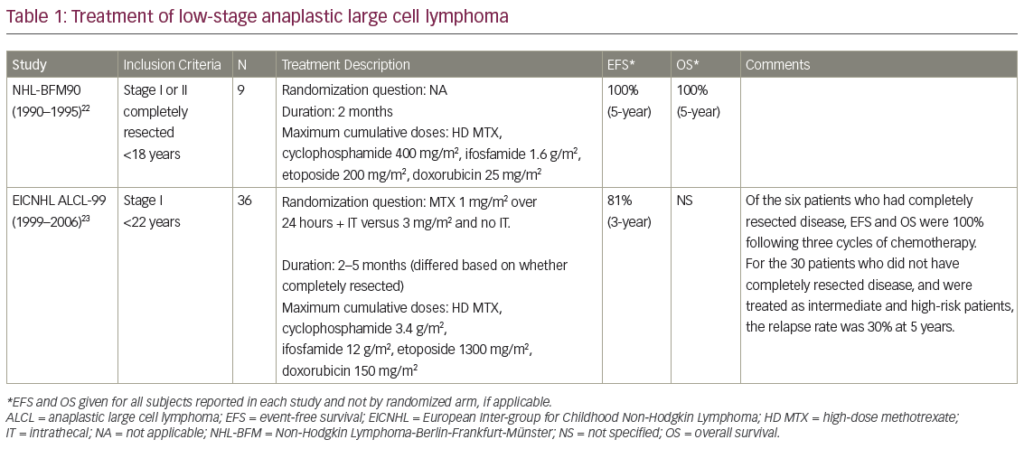
Annexin I is Transcriptionally Regulated in B – cell Lymphoma Cell Lines
In view of the lack of annexin I protein expression in the B-cell lymphoma cell lines, annexin I message expression was examined by RT-PCR analysis. RTPCR analysis showed that annexin I message was present in adenoids and absent in all of the B-cell lymphoma cell lines. As expected, HeLa, 293T and SW1116 cells showed annexin I message.
Annexin I gene was examined in selected cell lines by Southern blot analysis. There was no alteration in the annexin II genomic DNA as seen by no change in the hybridization pattern after cleavage by restriction enzymes, Xba I, Bam H1, and Apa I. These enzymes generated specific fragments that were identical in the Raji, HPB-null and Nalm-6 cell lines when compared with HeLa cells. Therefore, the down-regulation of annexin I expression in B-cell lymphoma cells is at the transcriptional level. Effect of Gene Demethylation on Annexin I Expression
Methylation of the CpG dinucleotide has been shown to directly inhibit transcription or stabilize structural changes in chromatin that prevent transcription. The nucleotide analog, deoxyC was used to inhibit DNA methylation in Raji cells to measure re-expression of annexin I protein. Exposure of Raji cells to either low (1?M) or high (10?M) deoxyC did not result in reexpression of annexin I protein after three days (lanes three and four). However, after six days of treatment with deoxyC, a weak protein band (lanes five and six), which appeared at the position of annexin I, was observed. There was no difference in annexin I reexpression at 1?M or 10?M deoxyC concentrations.
Raji Cell Line Response to LPS and H 2O2
To determine whether inflammatory stimulus provided by exposure of cells to bacterial lipopolysaccharides (LPS) will result in expression of annexin I, Raji cells were treated with various concentrations of LPS, and annexin I protein expression was measured by immunoblot analysis. Data has shown that exposure of Raji cells to LPS does not result in expression of annexin I in these cells. The effect of producing reactive oxygen species (ROS) in Raji cells on annexin I expression was also investigated. Data has shown that exposure of Raji cells to H2O2 did not cause annexin I expression.
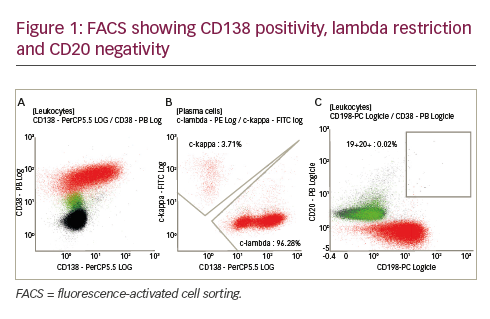
Immunohistochemical Analysis of Annexin I Expession in B-cell Lymphoma
Annexin I expression was studied by immunohistochemical analysis on archival specimens representing various B-cell lymphomas. Tonsil tissue sections were included as representatives of normal tissue. Positive identification of B-cells was performed by immunodetection of CD20 antigen. As expected, CD20 positivity was observed in the germinal centers and a few CD20 positive cells were observed in the epithelial layer. Annexin I expression was predominantly in the epithelial layer.Weakly positive annexin I expressing cells were found within the germinal center. At higher magnifications, distinct cytoplasmic localization of annexin I was observed in a subset of cells within the germinal center.These results are consistent with the immunoblot data, indicating a low-level of annexin I expression in the adenoids.
Annexin I expression was absent in the B-cell lymphomas. Distinct B-cell clusters were observed by their CD20 positivity in diffuse large cell non-cleaved B-cell lymphoma, small lymphocytic B-cell lymphoma, and follicular mixed B-cell lymphoma. However, annexin I expression was undetectable in each of these B-cell lymphomas, even at higher magnifications. Strong epithelial cell staining of annexin I was observed in these specimens.These results are consistent with the absence of annexin I expression in B-cell lymphoma cell lines.
Discussion
In this study, the expression of annexin I in B-cell lymphoma was examined. Annexin I is a pleotrophic calcium- and phospholipid-binding protein whose proposed functions include anti-inflammatory activity, mediation of apoptosis, regulation of cell differentiation, and membrane trafficking.8,15,30 Annexin I is expressed in the secretory bronchial epithelial cells,9 and its antiinflammatory N-terminus is lost in the bronchoalveolar lavage fluids from healthy smokers,11 indicating the importance of this protein in human health and disease. Annexin I is frequently over-expressed in human cancers including pancreatic16 and breast17 cancers. However, recent reports indicate that annexin I expression is down-regulated in other human tumors, particularly esophageal and prostate tumors.19–21 Annexin I is similar to the closely related protein annexin II, even though the two proteins are proposed to carry out distinct physiological functions. Curiously, both annexins I and II are lost in prostate cancers.20–24 Altered expression and loss of annexin II in B-cell lymphoma cell lines has been reported previously.22 In view of this, the expression of annexin I in B-cell lymphomas and cell lines were examined in the present study. The data presented in this manuscript indicate that annexin I is present in normal B-cells. Immunohistochemical examination of normal tonsil sections indicates that a subset of cells within the germinal centers distinctly express annexin I. The germinal center normally harbors highly proliferative B-cells;31 however, it also represents a highly dynamic environment that creates intense genomic instability among B-cells.32 It is unclear whether the subset of Bcells that show annexin I expression represent proliferative cells or cells that are undergoing differentiation. Association of annexin I expression and cellular differentiation has been shown previously.33
In contrast, most cells lines and neoplasms of pregerminal B-cells did not show detectable annexin I expression. Data are consistent with the loss of annexin I observed in prostate and esophageal cancers.21 The etiology of reduced annexin I expression was studied by examining the possible mechanisms. Southern blot analysis indicated that annexin I gene was intact in the Raji and OMA-BL- 1 cells, indicating that genomic deletion of annexin I is not the cause for loss of annexin I. Gene silencing by hypermethylation of annexin I promoter was examined by culturing cells in the presence of a demethylating agent, and the data indicated reexpression of annexin I protein after prolonged exposure to 10?M concentration of deoxyC. Thus, methylation of annexin I promoter could be one of the mechanisms for annexin I silencing in these cells. These results are similar to the re-expression of annexin II observed in a prostate cancer cell line after treatment with deoxyC,23 indicating methylation as a general mechanism for silencing annexins I and II in cancer tissues. Treatment of cells with a proinflammatory agent or an apoptosis-inducing agent did not result in expression of annexin I.
Conclusion
The data presented in this paper show that the antiinflammatory protein annexin I is expressed in normal B-cells, but not expressed in B-cell lymphomas and cell lines, similar to the absence of annexin I in prostate and esophageal cancers. Thus, annexin I may be an endogenous suppressor of cancer development, and loss of annexin I may contribute to B-cell lymphoma development. The additional mechanism(s) by which annexin I expression is down-regulated and the physiological consequences of annexin I loss in B-cell lymphomas need further investigation.
Acknowledgements
The author wishes to thank Dr Dennis Weisenburger for providing tissue specimen, Dr Sam Pirucello for providing the cell lines, and Dr Abhijit Banerjee for consultation.
© 2004 Vishwanatha et al; licensee BioMed Central Ltd. This is an Open Access article: verbatim copying and redistribution of this article are permitted in all media for any purpose, provided this notice is preserved along with the article’s original URL.
The electronic version of this article is the complete one and can be found online at: http://www.biomedcentral.com/1471- 2407/4/8