Introduction
MM is a haematologic cancer arising from a common progenitor of the B-cell lineage. It is characterised by a clonal expansion of plasma cells in the bone marrow (BM) and is nearly always accompanied by the presence of a monoclonal immunoglobulin (Ig) or Ig fragment in the serum and/or urine of patients.1,2 It is the second most common haematologic malignancy, and is responsible for at least 2% of cancer-related deaths. In 2006, an estimated 16,570 new cases of MM will be diagnosed in the US, and 11,310 deaths will be attributed to this disease.3 The last several years have seen the introduction of a number of novel agents in the treatment of this otherwise incurable cancer. Important advances, including drugs such as thalidomide (Thal), lenalidomide, and bortezomib, have provided effective therapeutic options for patients with MM.4 These and other novel agents hold promise to translate MM into a chronic disease.
Identification and Validation of Novel MM Therapies
In order to overcome resistance to conventional therapies and improve patient outcome, novel treatment approaches that target mechanisms whereby MM cells grow and survive in both the BM and extramedullary microenvironment are needed.
Our in vitro and animal model studies have demonstrated the importance of the BM microenvironment in promoting MM cell growth, survival, drug resistance and migration. These model systems have allowed for the development of several promising biologically based therapies that can target the MM cell in its BM microenvironment and thereby overcome classic drug resistance in vitro, including Thal and its more potent immunomodulatory analog, lenalidomide, as well as the proteasome inhibitor bortezomib.4 Once in vitro promise of these novel agents was demonstrated, efficacy was tested in murine models. Importantly, these laboratory studies have been translated to phase I, II, and III trials to evaluate their clinical efficacy and toxicity. In this context, bortezomib was FDAapproved for the treatment of relapsed and/or refractory MM;5 and both Thal and lenalidomide are currently being considered for approval by the agency. Several other classes of novel biologics, including histone deacetylase inhibitors, heat shock protein inhibitors, mTOR inhibitors, cyclin d inhibitors, pAKT inhibitors and oral proteasome inhibitors, among others are undergoing preclinical and clinical evaluation.These preclinical studies have informed the use of rational combination treatments in ongoing clinical trials.
Multiple Myeloma: A New Paradigm in Cancer Therapy
Abstract
Overview
Multiple myeloma (MM) is an incurable heematologic cancer responsible for 2% of cancerrelated deaths. Preclinical studies have underlined the significance of the bone marrow milieu in promoting tumour cell growth, survival, progression and the development of drug resistance. MM has emerged as a novel paradigm for cancer therapy since targeting both the MM cell and its microenvironment has resulted in improvements in patient outcome. This article will focus on the treatment of MM, with an emphasis on novel biologically based therapies.
Article
Further Resources
2. Share This

Trending Topic
Multiple myeloma (MM) is the second most common haematological malignancy, with upwards of 35,000 diagnoses in the USA each year.1,2 It remains a leading cause of blood cancer–related mortality worldwide, and although therapeutic advances have allowed for significant improvements in the median overall survival,3,4 the majority of patients still experience cycles of relapse that are eventually fatal.5 While patients with MM are living longer, a subgroup with high-risk disease at diagnosis still does poorly, with a median […]
Related Content in Multiple Myeloma

The optimal maintenance approach postautologous stem cell transplantation (ASCT) for patients with multiple myeloma is still an area of ongoing research. Lenalidomide has demonstrated clear superiority in clinical trials compared to placebo, and continuous post-transplant lenalidomide until progression is considered ...

Treatment options for myeloma have rapidly expanded in the past decade and, as with many cancers, the number of oral drug options has increased considerably. Given the more limited number of cancer hospitals, patients often must travel considerable distance to ...

Multiple myeloma (MM) remains largely an incurable disease with only a small percentage of patients achieving long-term remission.1 Here, we highlight some of the major studies on MM presented at the American Society of Hematology meeting in December 2021. Monoclonal gammopathy ...

Background on anti-BCMA CAR-T therapy Despite notable advances in the development of new drugs and the improvement of survival rates over the last 20 years, multiple myeloma (MM) persists as an incurable disease and nearly 35,000 new cases are expected in the ...

Multiple myeloma (MM) is the second most common haematological malignancy, affecting an estimated 450,600 patients worldwide, with an estimated 34,920 new diagnoses and 12,410 patient deaths in the USA in 2021.1,2 Although established curative therapies for MM have not yet been defined, we have ...
Multiple myeloma is a plasma cell malignancy that typically develops in individuals in their late 60s with an average survival time of ~8 years.1 Despite recent advances in treatment, multiple myeloma remains largely incurable due to development of drug resistance in ...
Multiple myeloma (MM) is a malignant clonal disorder of plasma cells in the bone marrow, and more than 32,000 new cases are expected in the USA each year.1 Many clinical advances, including novel drugs and continuous therapy with treatment sequencing, have ...

Multiple myeloma (MM) is the second most common hematological malignancy after non-Hodgkin’s lymphoma.1 Although recent decades have seen considerable advances and improvements in clinical outcomes for patients with MM,2 MM remains incurable, with a high disease burden.3 Clonal evolution ...

Amyloidosis is a group of diseases resulting from intracellular and extracellular deposition of insoluble abnormal amyloid fibrils, which alters the normal function of tissues. Glycosaminoglycans, apolipoprotein-E and serum amyloid P component comprise 10% of deposits, while amyloid fibrils formed by misfolded ...
An ever-expanding understanding of the biological basis for multiple myeloma (MM) combined with a widening array of effective therapies has dramatically improved outcomes for patients and has provided new opportunities for disease segmentation and targeted therapeutics.1 While this progress is ...
Smoldering myeloma is an asymptomatic clonal plasma cell disorder that is characterized by the presence of ≥3 g/dl serum M-protein and/or 10–59% bone marrow plasma cell infiltration, and is a precursor stage to multiple myeloma.1 Historically, patients with smoldering myeloma ...
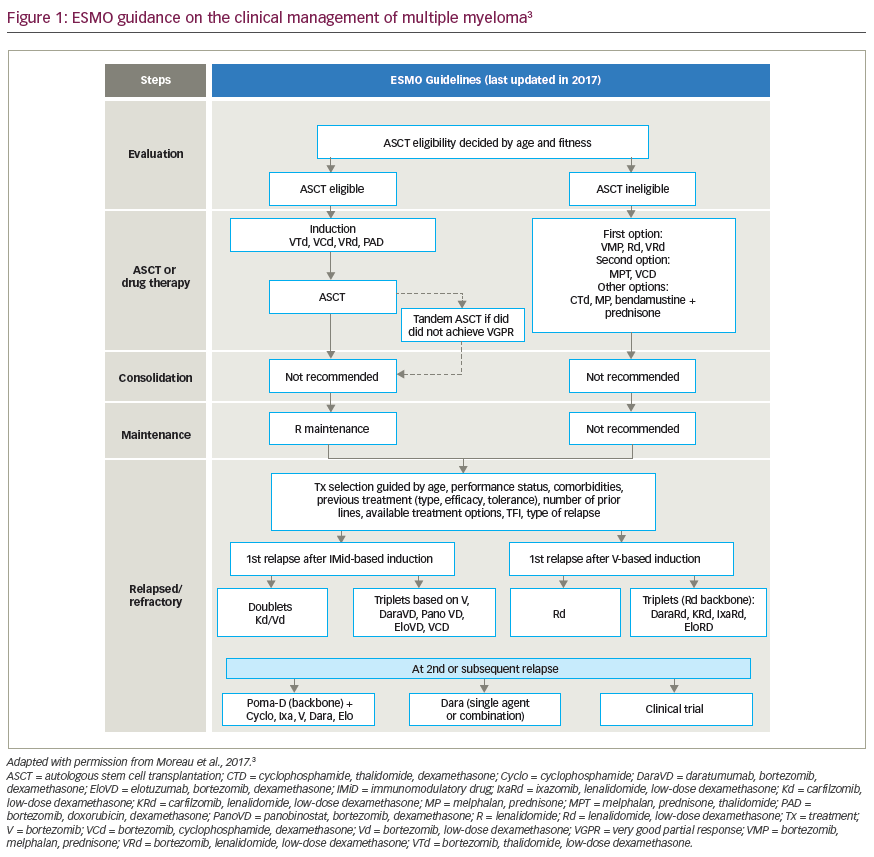
The past decade has experienced a shift in the treatment landscape for multiple myeloma (MM), with the introduction of novel agents and new combination therapies which have improved survival outcomes for patients across all disease settings.1 These advances in treatment ...
Latest articles videos and clinical updates - straight to your inbox
Log into your Touch Account
Earn and track your CME credits on the go, save articles for later, and follow the latest congress coverage.
Register now for FREE Access
Register for free to hear about the latest expert-led education, peer-reviewed articles, conference highlights, and innovative CME activities.
Sign up with an Email
Or use a Social Account.
This Functionality is for
Members Only
Explore the latest in medical education and stay current in your field. Create a free account to track your learning.

