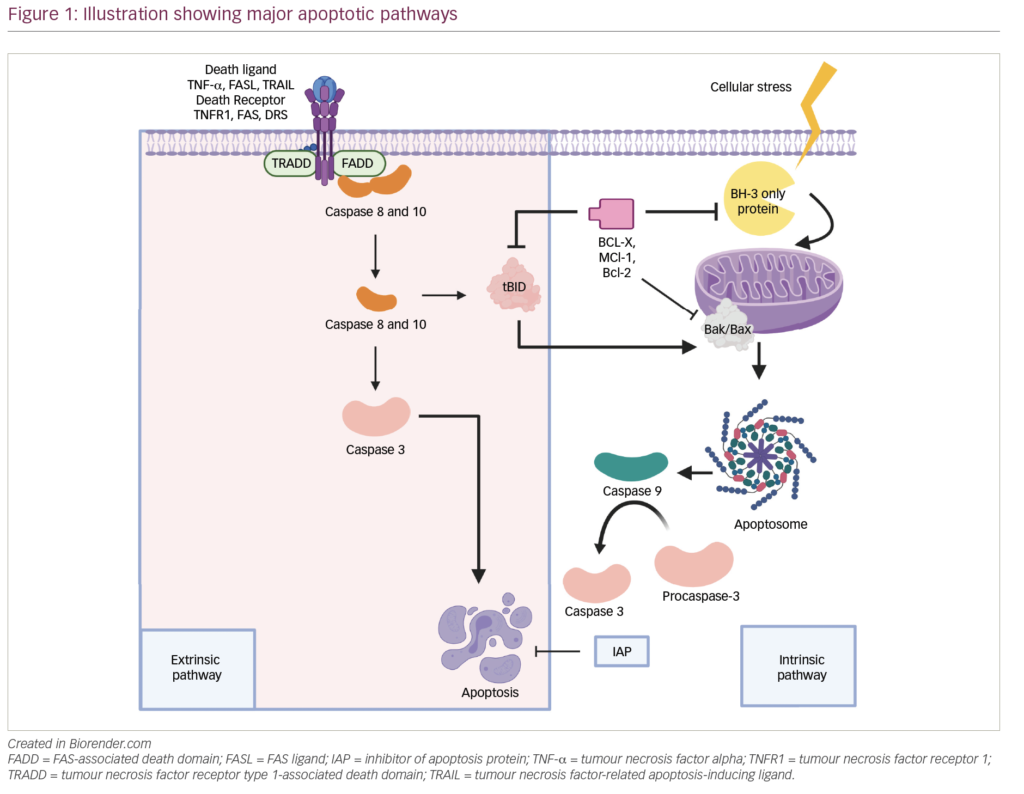
The optimal maintenance approach postautologous stem cell transplantation (ASCT) for patients with multiple myeloma is still an area of ongoing research. Lenalidomide has demonstrated clear superiority in clinical trials compared to placebo, and continuous post-transplant lenalidomide until progression is considered standard of care. All patients, however, do not derive equal benefit from single-agent lenalidomide, and intensified maintenance strategies are under investigation.1 Our group initially reported improved outcomes with risk-adapted, triplet maintenance compared with the historically short-lived progression-free survival (PFS) noted with either no maintenance or lenalidomide alone.2 More recently, the FORTE study, a randomized phase II trial, assessed the use of carfilzomib and lenalidomide, which noted a significant benefit compared to lenalidomide alone.3
In this context, the ATLAS study (ClinicalTrials.gov Identifier: NCT02659293) was designed to further address the question of optimal post-ASCT management.4 ATLAS is an international, randomized, phase III trial comparing two post-ASCT maintenance strategies: carfilzomib, lenalidomide and dexamethasone (KRd) for 36 cycles followed by lenalidomide until progression, versus lenalidomide monotherapy until progression. This simple approach was further modulated by adding a measurable residual disease (MRD)-based de-escalation. Among all patients who received KRd, standard-risk subjects who achieved MRD negativity after cycle 6 transitioned to a “risk-adapted” de-escalation to lenalidomide monotherapy after cycle 8. Patients were randomized after ASCT and stratified based on the post-ASCT response (≥ = very good partial response or < = very good partial response) and cytogenetic status (standard or high-risk). At a median follow up of 33.8 months, median PFS in the KRd group was 59.0 months, compared with 41.1 months in the lenalidomide group. The authors concluded that KRd and MRD-directed de-escalation in standard-risk patients should be considered as a new option for post-ASCT maintenance.4
While ATLAS met its primary endpoint, it is unclear how its results fit into the larger context of available and recent information. The primary analysis of the DETERMINATION study, as well as the parallel long-term follow up of the IFM2009 trial were recently reported.5,6 Both trials compared triplet induction with lenalidomide, bortezomib and dexamethasone (RVD), followed by consolidative ASCT and lenalidomide maintenance with RVD induction, followed by lenalidomide maintenance, without consolidative ASCT. The key difference in the trial designs was the duration of lenalidomide maintenance: IFM2009 treated patients for 1 year, whilst DETERMINATION used continuous lenalidomide as tolerated until progression. Both are large, phase III trials that represent modern-day treatment paradigms and serve as benchmarks for future studies.5,6
While designed to answer a different question, the scheme of ATLAS, specifically the control arm, is similar to the ASCT arms of IFM2009 and DETERMINATION: triplet induction, ASCT and lenalidomide maintenance. Despite similar treatment schemes, each of the three trials reported a dramatically different frontline median PFS of 67.5, 47.2 and 41.1 months in DETERMINATION, IFM2009 and ATLAS, respectively.4,5,6 Acknowledging the limitations of cross-trial comparisons, one would still anticipate similar durations of remission for similar therapy.
The roughly 25-month difference (67.5 months versus 41.1 months) in PFS between DETERMINATION and ATLAS is most striking. Both trials used ASCT followed by lenalidomide maintenance until progression. The induction regimens, however, varied significantly between the two studies. Patients in DETERMINATION (and IFM2009) uniformly received RVD, whilst in ATLAS, 60% of subjects received bortezomib, thalidomide and dexamethasone and 20% received bortezomib, cyclophosphamide and dexamethasone, with only 13% of the population receiving lenalidomide as part of induction.4,5,6 Proportions of patients with high-risk cytogenetics between DETERMINATION and ATLAS were similar (19.1% versus 21.0%, respectively).
Head-to-head comparisons of thalidomide and lenalidomide, as well as lenalidomide versus cyclophosphamide-based regimens, have demonstrated the superiority of lenalidomide.3,7 Decades of clinical trial data have also confirmed the impact of depth of response on PFS, with deeper responses correlating with longer progression-free intervals. Thus, sub-optimal induction is one logical explanation of the 25-month PFS difference observed between ATLAS (control arm) and DETERMINATION (ASCT arm). Notably, complete response, stringent complete response and MRD negative rates after induction in ATLAS have not yet been reported and may provide important insight into why this large difference in PFS was observed.
If sub-optimal induction is contributing to inferior PFS, this raises the question of whether more intensive post-ASCT maintenance (KRd versus lenalidomide) can fully restore outcomes, or whether response to induction (and ASCT) remain a major driver of long-term PFS that cannot be rescued with maintenance therapy. Thus, is ATLAS a positive study because the KRd arm was superior, or because the control arm underperformed? Randomized trials have yet to describe if intensified maintenance (doublet or triplet) is superior to lenalidomide after lenalidomide-based triplet induction. The FORTE trial may help to answer this question, however one out of three of the patients also received induction with cyclophosphamide, and one out of three did not undergo ASCT, both of which will likely contaminate such analysis. It is also important to scrutinize the endpoint of PFS in a maintenance trial, particularly when lenalidomide confers an overall survival advantage in standard-risk patients, and we know that PFS is an imperfect surrogate. Before a new treatment can be adopted, its impact on overall survival should be clear. This becomes increasingly important when the treatment burden, cost and overall quality of life with KRd versus lenalidomide is considered, especially if a similar or superior outcome could be achieved with 4–6 cycles of optimal induction therapy.
An additional observation with regards to the de-escalation of therapy in the standard-risk patients who achieved MRD negativity after 6 cycles of KRd, also warrants attention. Whilst it appears that the outcomes of such patients are similar to the standard-risk patients who completed KRd (PFS not reached in both groups), it should be highlighted that this was a prespecified, non-randomized intervention. Thus, it is unknown if such patients would continue to benefit from additional KRd compared with de-escalation. A second randomization with treatment assignment would be required at the time of achievement of MRD to answer this question. The difference in PFS between the standard-risk, MRD-negative patients in both treatment arms (KRd lenalidomide, and lenalidomide) should also be noted. Despite both groups achieving MRD negativity, the PFS curves are dramatically different. This highlights the importance of sustained MRD negativity, which is recognized as a better surrogate of PFS compared to one isolated MRD timepoint.8
Overall, we do not question the internal validity of ATLAS. It is a well-executed, randomized trial and one of the first to be conducted in Eastern Europe. We believe it is critical, however, to acknowledge that both induction regimens and MRD-directed discontinuation may represent confounders when comparing with other contemporaneous trials. We caution the extrapolation of its results to patients who receive RVD (or KRd) induction, as well as those receiving continuous maintenance in the context of standard-risk disease.