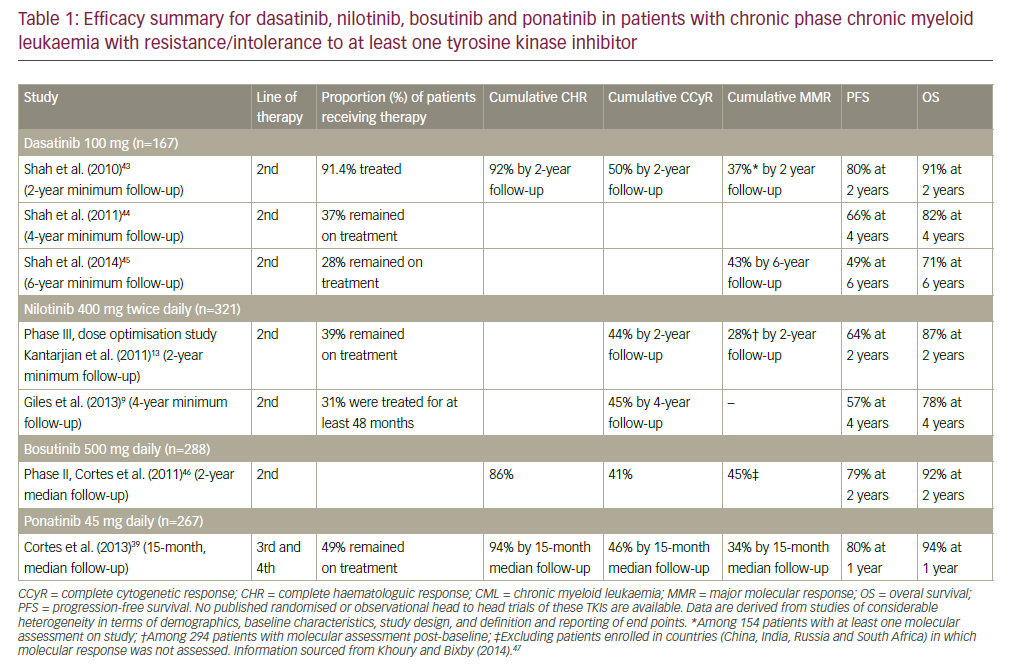
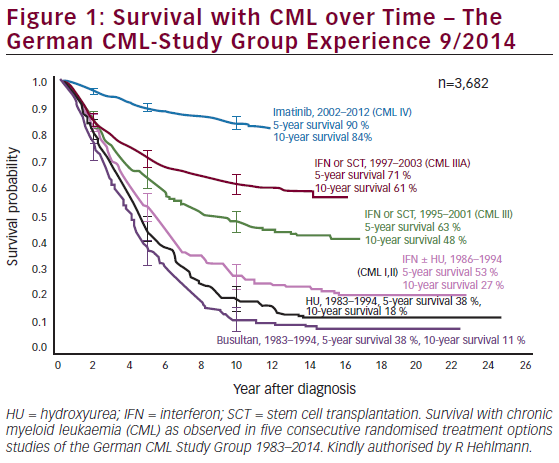
Chronic lymphocytic leukaemia (CLL) is the most common form of leukaemia in adults. It is caused by antigen-experienced B lymphocytes that either do not die and accumulate because of external survival signals or go into apoptosis and are replenished by proliferating precursors. The worldwide incidence of CLL is in the range of 0.5–5.5 cases per 100,000 people per year,1 with men twice as likely to develop it than women. In the Western hemisphere it is the most frequent form of leukaemia. The median age at presentation is approximately 65 years, but 20% of patients present before the age of 55 years.2,3 Although the overall median survival is approximately 10 years, the clinical course and prognosis of CLL are extremely heterogeneous: some patients never require treatment and have a survival similar to that of healthy age-matched individuals, whereas others have a poor prognosis and an early treatment requirement.
Today, advances in technology and understanding permit prognostic stratification of CLL patients leading to tailored therapies that have the potential of eradicating the leukaemic clone.
CLL Treatment Options
Until the early 1980s, chlorambucil was the primary treatment for CLL. However, this allowed only palliation of the disease rather than a cure. Subsequently, fludarabine (flu), a purine analogue, was introduced into the array of therapies for CLL. This drug, active in previously treated and untreated patients, showed a better complete remission rate and an advantage in terms of progression-free survival, although there was little effect on overall survival.4–8 Flu has also been investigated in combination with cyclophosphamide and some anthracyclines.
Monoclonal Antibodies and CLL
In the last decade a new class of therapies – humanised monoclonal antibodies (MAbs) – became available for the treatment of CLL. First came rituximab in 1997 for the treatment of patients with relapsed or refractory, low-grade or follicular, CD20-positive B-cell non-Hodgkin’s lymphoma, subsequently approved for CLL. Then, in the early part of the new century, another treatment option became available.
Alemtuzumab is a recombinant DNA-derived humanised MAb, and is the first to be targeted against the cell surface glycoprotein CD52. It was originally known as Campath-1 and was derived from mouse antibodies raised against human lymphocyte proteins.
Initially, it was not a good candidate for therapy because of the non-human components of the antibody. Therefore, Greg Winter and his colleagues humanised Campath-1 by extracting the hypervariable loops that had specificity for CD52 and grafting them onto a human antibody framework, creating Campath-1H.
Alemtuzumab was first approved by the US Food and Drug Administration (FDA) in May 2001. This was followed a few months later by approval in Europe. It is indicated for patients with B-cell CLL who have been treated with alkylating agents and have failed fludarabine therapy; it is a second-line therapy for CLL.
It is also used under clinical trial protocols for treatment of some autoimmune diseases, such as multiple sclerosis, and in some conditioning regimens for bone marrow transplantation and kidney transplantation.
Although rituximab (used alone either at standard or at high doses) has proved highly effective in different forms of non-Hodgkin’s lymphoma (NHL), the results in CLL have not been satisfactory.9–12 Alemtuzumab, used in monotherapy, seems to be more active: good results have been achieved both in poor-risk patients (flu-refractory patients and patients with p53+ cells) and in untreated patients.13–15 It is currently being investigated for use as a first-line treatment for B-cell CLL.
Given that alemtuzumab’s target, CD52, is expressed by normal B-cells, T-cells and natural killer (NK) cells, it should be noted that the administration of this MAb causes an immunodepression and an increased risk of infection, particularly with cytomegalovirus. With regard to the correlation between response to therapy and biologic features, it has been reported that p53+ patients show an unsatisfactory response to alkylating agents, flu and rituximab, whereas alemtuzumab may be effective.
Monoclonal Antibodies in Combination Therapy
Better results have been reported with schedules that combine chemotherapy with MAbs. Data from the Cancer and Leukemia Group B 9712 (CALGB 9712) study have shown that the simultaneous administration of flu and rituximab is superior in terms of response than a schedule in which rituximab is given as consolidation treatment after flu.16
A retrospective comparative analysis of CALGB 9712 and CALGB 9011 reported that the addition of rituximab in untreated CLL patients (simultaneously or concurrently) to flu may prolong overall survival.17 The efficacy of rituximab in combination therapy has been supported by encouraging results with a schedule that combined flu and rituximab with cyclophosphamate.18,19
In untreated patients the overall response rate (ORR) was 95% and the complete response rate (CRR) 70%; within the latter, 78% of patients achieved a flow-cytometric complete remission.18 In previously treated patients the ORR was 73% (CRR 25%), with 32% of molecular remissions in the 37 evaluated patients (all in complete remission).19
The combination of flu with alemtuzumab, when administered in patients with relapsed or refractory CLL, has also proved effective.20,21 Nevertheless, this conclusion is based on small series of patients; larger studies are necessary for better definition of the efficacy of this combination.
In many patients, however, even combination therapy results in persistent, minimal, residual disease as has been shown by both polymerase chain reaction (PCR) and flow cytometry.22 Given that remission duration and better outcome correlate with the quality of response, the efficacy of a consolidation treatment, administered with the intent of eradicating minimal residual disease, has been evaluated.23
Conclusion
After decades during which conservative management was the normal approach to CLL, the attitude to this disease has changed remarkably during the past few years. Advances in diagnosis and prognosis and in the comprehension of the pathogenesis of the disease and biology of CLL lymphocytes, and the broadening of the array of therapies, which include newer and potentially more efficacious therapies and stem cell therapy procedures capable of eradicating the leukaemic clone have challenged the conservative management that for many years has been the normal approach to CLL patients. In fact, through complete biologic and clinical examination it is both possible and mandatory to perform a correct diagnostic and prognostic characterisation aimed at identifying patients with a good or poor prognosis. In this last category it needs to be clarified whether early and aggressive treatment may affect life expectancy. All these issues indicate that the scene is set for all CLL patients to be offered a more targeted treatment (or non-treatment) algorithm based on the clinical and biologic characteristics of the disease.