Approximately 10 % of patients with localised Hodgkin’s lymphoma (HL) and 20 % with disseminated HL fail to respond to or relapse after a first-line treatment.
The standard of care indicated by a randomised study1 is salvage chemotherapy followed by consolidation with autologous stem cell transplant (ASCT). Salvage chemotherapy followed by ASCT induces long-term remission in ~50 % of patients.2 However, there is a limited impact on survival.1,3 The increase in the cumulative incidence of second malignancies in the transplanted patients compared with conventionally treated HL patients is still a major issue.4 The main point is to achieve a major response with complete remission (CR) prior to transplantation, because a suboptimal response to pretransplant salvage chemotherapy is the major determinant of ASCT failure.5 The introduction of the pretransplantation positron emission tomography (PET) scan has changed the paradigm of treatment. Standardisation of criteria of responses with PET scan was based on the Deauville criteria or the Five-Point Scale 6. In a series of 111 consecutive patients, a 5-year progression-free survival (PFS) of 23 % and 79 % (p<0.001) and a 5-year overall survival (OS) of 55 % and 90 % (p=0.001) were documented in patients with or without 18-Fluoro-deoxyglucose (FDG)- avid lymphoma lesions at ASCT, respectively.7
Patients with a positive PET after a second-line salvage therapy have a worse event-free survival (EFS) and require new approaches.
The European intergroup study Hodgkin disease relapse 2 (HDR2) showed that adding sequential high-dose chemotherapy after salvage chemotherapy and before ASCT does not improve a prognosis and is associated with additional adverse effects.8
Transplantation Strategy – Auto or Allo or Tandem?
A single ASCT is appropriate for intermediate-risk patients. Various conditioning regimens have been used as chemotherapy prior to ASCT. However, there have been no prospective comparison trials. BEAM (carmustine, etoposide, cytarabine and melphalan) has been one of the most popular regimens since its introduction in the 1980s. More data regarding newer regimens, including active agents such as bendamustine, busulfan and fotemustine, are required.
To improve the outcomes of patients with high-risk disease, tandem transplantation could be proposed provided they display chemosensitivity and that there is no progression between the two transplants. The LYmphoma Study Association (LYSA) reported a prospective multicentre trial that evaluated a risk-adapted salvage treatment without a PET scan with a single or tandem ASCT for 245 relapse or refractory patients.9
Of the poor-risk patients, 105 (70 %) received a tandem ASCT, whereas 92 intermediate-risk patients (97 %) received a single ASCT. The 5-year PFS and OS estimates were 73 % and 85 %, respectively, for the intermediate-risk group and 46 % and 57 %, respectively, for the poor-risk group. The outcomes for the primary refractory and poor-risk/ relapsed HL groups were similar.
As improvements in OS after an allogeneic haematopoietic cell transplantation (allo-SCT) has been reported (following the introduction of reduced intensity conditioning [RIC] regimens, improvements in human leukocyte antigen [HLA] typing, and more careful patient selection), this strategy was also tested in HL. A large, prospective phase II study conducted by the Grupo Español de Linfomas/Trasplante de Médula Osea (GEL/TAMO) and the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation (EBMT) enrolled 78 relapsed or refractory HL patients.10 Fourteen patients (18 %) were unable to be transplanted due to inadequate disease control. They died after a median time of 10 months. After transplantation, the PFS was 48 % at 1 year and 24 % at 4 years. The patients who were allografted in CR had a significantly better outcome with a PFS of 70 % and 50 % at 1 and 4 years, respectively. Within the tandem transplantation framework, an ASCT followed by a RIC allo-SCT is also of particular interest for young patients, with a reported 10-year PFS of 60 %.11 However, high-dose chemotherapy and radiotherapy cause significant acute and long-term side effects. Patients who relapse after an ASCT have a poor prognosis. New drug development in HL is particularly important not only to improve the prognosis of patients who fail first-line standard-dose chemotherapy but also to decrease the shortand long-term toxic effects that are correlated with the burden of chemo-radiotherapy.
New Drugs Strategy
New drugs should aim to inhibit specific receptor interactions on the surface of tumour cells and specific driving pathways in malignant HL cells to avoid non-specific side effects from chemotherapy. Moreover, targeted therapies with tolerable side-effect profiles are needed in patients who are not suitable for a high-dose chemotherapy (HDCT) or ASCT, particularly frail patients or patients in developing countries.
Brentuximab Vedotin – More than a Bridge to Transplantation?
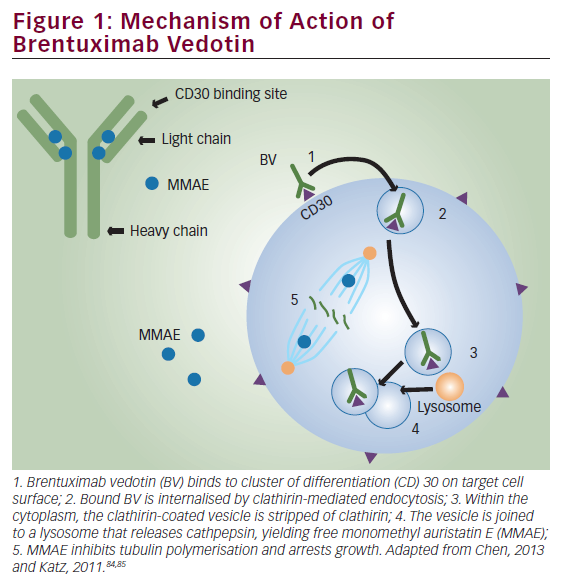
Brentuximab vedotin (BV) is an anti-CD30 antibody conjugated to monomethyl auristatin E, an anti-microtubule. A member of the tumour necrosis factor (TNF) receptor superfamily, CD30 is a transmembrane glycoprotein receptor expressed on the surface of Hodgkin Reed Sternberg (HRS) cells. It was granted accelerated approval by the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for patients with relapsing HL with ASCT failure or who are ineligible for an ASCT. The most common adverse events are peripheral neuropathy, cytopenias and fatigue. Approval for HL was based on the results from a multicentre phase II trial in which 102 patients with relapsed HL after an ASCT achieved an overall response rate (ORR) of 73 %, CR rate of 32 % and partial emission rate (PR) of 40 % with median response durations of 6.7, 20.5 and 3.5 months, respectively.12 With an updated median follow-up of 53 months, 12 patients remained in CR without a consolidative allo-stem cell transplant.
Current trials are evaluating BV as a first-or second-line therapy in combination with standard chemotherapy (BV-adriamycin, vinblastine and dacarbazin [AVD], BV-dexamethasone, high-dose ara cytarabine, platinum [DHAP] and BV-ifosfamide, carboplatinum, etoposide [ICE]). Interestingly, the introduction of BV seems to increase the quality of response as a bridge to allow transplantation with minimal toxicity. Maintenance therapy post-auto-transplantation was reported in the phase III AETHERA study, which evaluated BV after an ASCT in highrisk relapse/refractory HL.13 The patients included had to satisfy one of the following criteria: chemo resistance, relapse after CR <1 year or extranodal disease. They were randomised after the ASCT between BV 1.8 mg/kg q3wk versus placebo. The main end point was the PFS by an independent review. The PFS was extended by 18.8 months. The median PFS was 42.9 months in the 165 treated HL patients versus 24.1 months in the 164 control patients. No overall survival difference was observed at an initial interim analysis. However, a cross-over from placebo to BV occurred in 85 % of these cases.
Programmed Death 1 Blockade in Hodgkin’s Lymphoma
The therapeutic blockade of immune checkpoint pathways, in particular cytotoxic T-lymphocytes associated programmed-death (PD-1), has been tested in haematological malignancies. Two blocking monoclonal anti-PD-1 therapies have been evaluated in two phase I studies with remarkable efficacy. The first drug, nivolumab14, included 23 relapsed HL patients after an ASCT and BV in 78 % of the cases. The response rate was 70 %, including 17 % with CR and acceptable safety. The rate of PFS at 24 weeks was 86 %. These data led to an accelerated approval in both the US and Europe. The same outstanding results were observed in 29 HL patients with a 66 % response rate and 21 % CR.15
Conclusion
Increased knowledge of the molecular pathogenesis of HL might lead to the development of targeted drugs that achieve long-term responses with good tolerability. Numerous questions remain about their optimal role and use. However, for the first time new drugs have been developed for HL with promising results.
In first-line treatment, interim assessments with PET scan will permit the monitoring of treatment efficacy and the rapid adaptation of the development strategy with new agents.
For relapse/refractory patients, BV and checkpoint blockade agents will increase the quality of remission and act as a bridge before proceeding to transplant or preventing relapse post-transplantation.