For most cancers, African-Americans have the highest mortality rates and shortest survival times of any racial or ethnic group in the US. Furthermore, compared with other racial/ethnic groups, African-Americans experience the highest rates of late-stage cancers and consequently the poorest cancer outcomes.1 In addition to later-stage cancer diagnoses, African-American cancer patients in the US frequently receive a different standard of care, even if diagnosed at the same stage of disease.1–5 Evidence further shows that when multiple treatment regimens are available, African-Americans are more likely to receive a less efficacious form of treatment, do not consistently receive information regarding all available treatment options, and are less likely to be referred to a specialist and/or to a chemoprevention clinical trial.2–6 Even though tremendous advances in cancer detection and treatment have benefited all populations, and despite targeted efforts aimed at addressing gaps in cancer outcomes in racial/ethnic minorities, African-Americans continue to suffer disproportionately from every major form of cancer (see Table 1).1
The widespread racial disparity in the early detection of cancer and in cancer morbidity, mortality, treatment, and survival are well-documented and reflect a national phenomenon. These persistent racial/ethnic disparities, which are often magnified in the major cancers such as cancers of the lung, colon and rectum, breast, and prostate, have prompted federal agencies to develop national cancer control initiatives and programs to assist states in eliminating cancer disparities in communities of color.7–8 Recommendations and initiatives targeting racial/ethnic minority populations in general and African-Americans specifically have been outlined in numerous reports such as ‘Healthy People 2010’ (HP 2010),8 ‘The Unequal Burden of Cancer,’9 and the landmark document ‘Unequal Treatment: Confronting Racial and Ethnic Disparities in Healthcare.’2 These documents, along with a host of data from research studies and clinical trial reports, have repeatedly confirmed that the disparity between racial/ethnic minority populations and Caucasians in the US persists.
However, those who are committed to reducing (and eventually eliminating) healthcare disparities in general and cancer disparities specifically concede that the time has come to move beyond merely documenting racial/ethnic disparities and to begin to focus our efforts and resources on strategies to address the seemingly intractable racial/ethnic disparities among social groups. We know that addressing the cancer disparity in African-Americans and other racial/ethnic minority groups requires targeted, multidisciplinary efforts that span the cancer continuum. Indeed, a number of national initiatives and community-based programs and interventions specifically targeting African-Americans and other racial/ethnic minority populations are under way.10–13 However, to date reducing the racial/ethnic disparity among social groups has proved to be an elusive goal.
It is the complex meaning of race/ethnicity and the manner in which it is used to assign value and life opportunities in society that poses the greatest challenge to reducing racial/ethnic disparities. Furthermore, the intricate manner in which race/ethnicity and social status are interwoven and the resultant health implications on minority populations in the US present an additional set of unique challenges. We believe that the distinct set of challenges embedded in issues of race/ethnicity and social status and their impact on cancer prevention and risk, early detection and screening, and diagnosis and treatment ultimately delay progress toward the goal of reducing cancer health disparities in African-Americans and other racial/ethnic minority populations. This brief report will highlight specific conceptual challenges that impede progress toward the goal of reducing the racial/ethnic gap in cancer health disparities and present recommendations for future research and intervention.
Reducing Cancer Disparities Among Racial/Ethnic Minority Populations
Clarifying the Root Causes of Racial/Ethnic Cancer Disparities The first task in addressing racial/ethnic cancer disparities requires that we better understand the root causes of inequities in disease among subpopulations and develop effective interventions and strategies to redress those inequities. However, the challenge lies in the complex, multifactorial nature of racial/ethnic disparities and our inability to point to a single etiological determinant of disease. Why do African-Americans suffer disproportionately from cancer? Is it a function of biology and genetics, behavior, and lifestyle, or disadvantaged social class status? These are critical questions that will have to be addressed to reduce the racial/ethnic gap.
How do we define the biological and genetic contributions to health disparities in African-Americans and other racial/ethnic minority groups in relation to their disadvantaged social status and the accompanying low socioeconomic positioning and decreased access to quality care? Disentangling these multiple effects is a daunting task.
Defining the Contribution of Biology and Genetics
What role do biology and genetics play in health disparities among African- Americans? This is a key controversy framing the scientific debate on the root causes of racial/ethnic health disparities. There are those in the field who are committed to exploring the biological and genetic basis of health disparities and firmly believe that biology and genes play a central role in their root causes. The proponents of biology and genetics point to the examples of breast and prostate cancers. African-Americans diagnosed with breast and prostate cancers appear to experience disproportionately increased rates of high-grade and aggressive tumor profiles, and often present at relatively younger ages with more advanced-stage disease. These findings appear to point to some biological/genetic component, and thus research in this area of cancer etiology is ongoing. Still, there are those who believe that while biology and genetics may play a role, the biological and genetic factors are inextricably bound to environmental factors (e.g. gene–environmental interactions).
Defining the Contribution of Lifestyle and Behaviors
Clarifying the role of behavioral, lifestyle, and cultural factors and their impact on racial/ethnic cancer disparities is an important challenge. To date, we do not fully understand what effect the personal health behaviors of African-Americans have on pervasive and systemic population-based racial/ethnic disparities. The fact remains that regardless of their personal health practices, African-Americans are at greater risk for developing and dying from cancer and have the lowest survival times among all racial/ethnic groups after a cancer diagnosis. And while lifestyle behaviors such as diet, physical activity, smoking, and cancer screening may appear to be a personal choice on the surface, in reality they often have much deeper historical, economic, and political roots.
Take smoking, for example. According to the Surgeon General’s report ‘Tobacco Use Among US Racial and Ethnic Minority Groups,’14 tobacco use among minorities is attributed to a number of factors. These factors include socioeconomic status (SES), acculturation, stress, biological factors, limited English proficiency, the price of tobacco products, and targeted advertising within racial/ethnic minority communities. Strategies to effectively reduce population rates of tobacco use in these communities must therefore include culturally relevant approaches, resources for smoking cessation, policy changes, rigorous prevention research, and additional resources to build infrastructures to facilitate tobacco control within racial/ethnic minority communities.15
Unfortunately, many African-American and Hispanic/Latino communities lack the necessary economic resources or political acumen to effectively mobilize against targeted tobacco campaigns in their communities.15 As a result, we have failed to see a substantial decline in tobacco use in minority populations, and consequently African-Americans experience higher lung cancer incidence and mortality rates and lower survival times compared with US Caucasians, due in part to their smoking behaviors. Hence, a seemingly personal decision to smoke tobacco is in reality rooted in wider social, economic, and political determinants of health and health disparities.
An example of the complexity of a seemingly personal health decision can also be seen in the case of cancer screening. Comparably high survival rates exist for all racial/ethnic groups diagnosed at a localized stage. However, African-Americans are more likely to be diagnosed with late/advanced-stage cancers. The increased likelihood of a late/advanced-stage cancer diagnosis suggests that racial differences in survival could be reduced if more cancers in African-Americans were diagnosed and treated at a clinically localized stage. Several studies have concluded that a later stage at diagnosis is the primary reason for the higher likelihood of cancer mortality among African-Americans compared with US Caucasians.16–20 Thus, efforts to identify factors that increase the likelihood of early-stage diagnoses in African-Americans are ongoing.
However, the under-utilization of screening services among racial/ethnic minority populations is not simply a matter of personal choice. As with the example of tobacco smoking, low screening rates have been attributed to factors such as the lack of access to state-of-the-art cancer screening facilities, lack of provider recommendation for cancer screening, cultural and language barriers, and being uninsured or underinsured.16–20 Each of these factors is beyond the level of the individual and points to more complex social, political, and economic determinants of health and health disparities.
While individual-level factors such as biological and genetic endowment, lifestyle choices, and screening behaviors are challenges that will have to be addressed to ultimately reduce cancer disparities in African-Americans, individual factors do not fully explain the persistent inequalities in population-based cancer mortality rates. For example, individual factors alone cannot account for the increased mortality risk among African-Americans without a high-risk genetic profile who engage in healthy lifestyle behaviors (including cancer screening). Rather, population-based cancer mortality consistently traces along a class gradient whereby the most vulnerable and impoverished populations experience the highest cancer mortality, while the most privileged and affluent populations have the lowest.21–24
Defining the Role of Social Determinants
The sociopolitical and economic conditions of resource-poor communities have long been implicated in premature mortality in a number of disease outcomes, including cancer. However, little is known about how the structural features of an entire community (including its historical, cultural, and social influences, social and physical institutions, and political activities) affect population health,22 but it is precisely these complex social, political, and economic issues that pose some of the greatest challenges for reducing cancer disparities in racial/ethnic minority populations.
Epidemiologists have noted that the relationship between SES and cancer incidence and mortality varies among racial/ethnic groups in the US.24 Persons of low SES often live in neighborhoods adjacent to toxins (i.e. city and county toxic waste landfills and commercial hazardous waste facilities).25 Low SES has been offered as a possible explanation for the low participation rates in early detection and screening of African-Americans and as a plausible explanation for the racial/ethnic disparity in advanced/late-stage cancer at presentation.26 While variations in cancer incidence may occur primarily through behavioral and environmental mechanisms, differences in mortality rates may represent the cumulative effects of health-risk behaviors, social and environmental factors, information and healthcare access, and appropriate medical care services.21–26
The Challenge of Targeting African-Americans and Other Racial/Ethnic Minorities to Reduce Cancer Health Disparities
A second major challenge in reducing cancer incidence and mortality among African-Americans is the equitable distribution of limited resources that are necessary for the reduction and eventual elimination of healthcare disparities among population groups. Of primary concern is the fact that an effective population-based strategy would consequently have universal utility, meaning that it would not be race- or ethnicity-specific; hence, a truly effective strategy to reduce disparities among African- Americans would also have health benefits among Caucasians and other racial/ethnic groups if applied equitably. What history has shown thus far is that the ‘best-off’ groups (i.e. US Caucasians and Asians/Pacific Islanders) would show the most impressive improvements and the ‘worstoff’ groups (i.e. African-Americans, American-Indians/Alaska Natives, and Hispanics) would show only modest improvements.27–28 Therefore, you would have positive improvements in all racial/ethnic groups if a public health strategy was applied equitably, but you would not necessarily close the gap between the racial/ethnic groups. This raises a key question: should effective public health strategies be targeted specifically to exclusive social groups such as African-Americans to reduce disparities but not applied equally to other racial/ethnic groups to improve cancer outcomes in all populations?
Targeting limited resources to racial/ethnic minorities is an approach that is vulnerable to criticism because even the ‘best’ performing subgroups also fall short of key targets (based on HP2010 goals) to improve key cancer outcomes.8 The critique of the targeting approach is that we should be directing cancer prevention and control resources to improve the cancer risk profiles of all populations and that targeting only African-Americans is an inherently unfair strategy.
The counter-argument is that targeting specific high-risk racial/ethnic minorities is simply a matter of extending quality care interventions to disenfranchised populations who would not receive care at all or would routinely receive a different (often lower) quality standard of care than majority populations. Take the example of cancer clinical trials. The Selenium and Vitamin E Cancer Prevention Trial (SELECT) is the largest-ever prostate cancer prevention trial. Previous studies suggest that selenium and vitamin E (alone or in combination) may reduce the risk for developing prostate cancer by 60 and 30%, respectively, but only a large clinical trial such as SELECT could confirm those initial findings.29
The SELECT study began enrolling patients in August 2001. The recruitment of Caucasians to the clinical trial was rapidly outpacing that of African-Americans. The rapid recruitment of Caucasians in proportion to African-Americans persisted despite literature that cited African-American prostate cancer incidence rates being on average 60% higher than in Caucasian men.1
The inclusion criteria were broadened to enhance the recruitment of high-risk African-American men into this important cancer prevention clinical trial. Researchers also elected to reduce the minimum age eligibility from 55 to 50 years for African-American men because these men have a higher incidence of prostate cancer at a younger age compared with all other groups. The SELECT eligibility criteria also allowed men with controlled comorbidities.
Lowering the minimum age requirement to 50 years for all African-Americans resulted in the accrual of 1,723 men (33% of the total African-American accrual), which would not have been possible if the 55-year age limit (for Caucasians) had been a uniform requirement.29 When enrollment closed in June 2004, approximately 15% of the participants were African-American. This targeted strategy was effective not only for enhancing enrollment of an under-represented at-risk minority subpopulation, but also for improving the generalizability of the cancer clinical trial results.29
To effectively reduce the cancer disparity gap among African-Americans, the challenge will be to focus our efforts on how to equitably distribute resources in such a way as to improve the cancer outcomes of African-Americans without impeding the improvement of Caucasians and other racial/ethnic groups. Williams30 provides five principles for reducing the disparity gap among African-Americans: provide insurance coverage and access to high-quality care for all; provide a diverse healthcare workforce to ensure culturally competent care; deliver patient-centered care; maintain accurate and complete race/ethnicity data to monitor disparities in care; and set measurable goals for improving quality and ensure that goals are achieved equitably for all racial and ethnic minorities.
These individual-level principles can be implemented throughout the US healthcare system, by federal and state governments, health plans, hospitals, medical groups, and healthcare professionals.30 However, to aggressively reduce the cancer gap, individual-level principles will have to be implemented alongside societal-level principles that address the social, economic, and political determinants of racial/ethnic cancer disparities. Societal-level principles include providing universal healthcare that does not deliver unequal treatment based on race/ethnicity and SES; investing in education and job opportunities for racial/ethnic minorities; and improving residential and social environments.
Conclusion
Despite the advances in cancer detection and treatment, racial/ethnic minority populations continue to experience poorer cancer-related outcomes. This article has provided a brief discussion of the challenges of reducing and ultimately eliminating cancer disparities among African-Americans and other racial/ethnic populations. The conceptual challenges of disentangling the multiple factors that contribute to our understanding of the root causes of health disparities and issues surrounding targeting racial/ethnic minorities will have to be addressed to close the cancer gap. ■
Bridging the Gap Between Populations—The Challenge of Reducing Cancer Disparities Among African-Americans and Other Ethnic Minority Populations
Article
References
- American Cancer Society, Cancer Facts and Figures for African Americans 2007–2008, American Cancer Society, 2007.
- Institute of Medicine, Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care, Washington, DC: National Academy Press, 2002.
- Shavers V, Brown M, Racial and ethnic disparities in receipt of cancer treatment, JNCI J Nat Cancer Institute, 2002;94:334–57.
- Bach PB, Schrag D, Brawley OW, et al., Survival of blacks and whites after a cancer diagnosis, JAMA, 2002;287(16):2106–13.
- Underwood W, DeMonner S, Ubel P, et al., Racial/ethnic disparities in the treatment of localized/regional prostate cancer, J Urology, 2004;171:1504–7.
- Morris AM, Billingsley KG, Hayanga AJ, et al., Residual treatment disparities after oncology referral for rectal cancer, JNCI J Nat Cancer Institute, 2008;100(10):738–44.
- National Cancer Institute (US), The National Cancer Institute strategic plan to reduce health disparities, Bethesda, MD: National Cancer Institute, 2002.
- Healthy People 2010, 2008. Available at: www.healthypeople.gov/
- Institute of Medicine, The Unequal Burden of Cancer: An Assessment of NIH Research and Program for Ethnic Minorities and the Medically Underserved, Washington, DC: National Academy Press, 1999.
- Lisovicz N, Johnson RE, Higginbotham J, et al., The Deep South Network for cancer control. Building a community infrastructure to reduce cancer health disparities, Cancer, 2006;107(Suppl. 8): 1971–9.
- Jandorf L, Fatone A, Borker PV, et al., Creating alliances to improve cancer prevention and detection among urban medically underserved minority groups. The East Harlem Partnership for Cancer Awareness, Cancer, 2006;107(Suppl. 8): 2043–51.
- Adams EK, Breen N, Joski PJ, Impact of the National Breast and Cervical Cancer Early Detection Program on mammography and Pap test utilization among white, Hispanic, and African American women: 1996–2000, Cancer, 2007;109(Suppl. 2): 348–58.
- Freeman HP, Patient navigation: a community based strategy to reduce cancer disparities, J Urban Health, 2006;83(2):139–41.
- US Department of Health and Human Services, Tobacco Use Among US Racial and Ethnic Minority Groups—Africans, American-Indians, and Alaska Natives, Asian-Americans and Pacific Islanders, and Hispanics: A Report of the Surgeon General. Atlanta, US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 1998.
- Fagan P, Moolchan ET, Lawrence D, et al., Identifying health disparities across the tobacco continuum, Addiction, 2007;102 (Suppl. 2):5–29.
- Gullate M, The influence of spirituality and religiosity on breast cancer screening delay in African-American women: application of the Theory of Reasoned Action and Planned Behavior, ABNF J, 2006;17(2):89–94.
- Lantz PM, Mujahid M, Schwartz K, et al., The influence of race, ethnicity, and individual socioeconomic factors on breast cancer stage at diagnosis, Am J Public Health, 2006;96(12):2173–8.
- Haas JS, Earle CC, Orav JE, et al., Racial segregation and disparities in cancer stage for seniors, J Gen Intern Med, 2008;23(5):699–705.
- Lee CT, Dunn RL, Williams C, Underwood W 3rd, Racial disparity in bladder cancer: trends in tumor presentation at diagnosis, J Urology, 2006;176(3):927–33.
- Gadgeel SM, Severson RK, Kau Y, et al., Impact of race in lung cancer: analysis of temporal trends from a surveillance, epidemiology, and end results database, Chest, 2001;120(1): 55–63.
- Braveman PA, Cubbin C, Egerter S, et al., Socioeconomic status in health research. One size does not fit all, JAMA, 2005;294(22):2879–88.
- LaVeist TA, Disentangling race and socioeconomic status: a key to understanding health inequalities, J Urban Health, 2005;82(Suppl. 3):iii26–34.
- Fiscella K, Williams DR, Health inequalities based on socioeconomic inequities: Implications for urban healthcare, Academic Medicine, 2004;79:1139–47.
- Singh GK, Miller BA, Hankey BF, Edwards BK, Area socioeconomic variations in US cancer incidence, mortality, stage, treatment, and survival, 1975–1999. NCI Cancer
Surveillance, National Cancer Institute, 2003;03–5417. - Evans GW, Kantrowitz E, Socioeconomic status and health: The potential role of environmental risk exposure, Ann Review of Pub Health, 2002;23:303–31.
- Byers TE, Wolf HJ, Bauer KR, et al., for the Patterns of Care Study Group, The impact of socioeconomic status on survival after cancer in the US: findings from the National Program of Cancer Registries Patterns of Care Study, Cancer, 2008;113(3):582–91.
- Pearcy JN, Keppel KG, A summary measure of health disparity, Public Health Reports, 2002;117(3):273–80.
- Harper S, Lynch J, Meersman SC, et al., An overview of methods for monitoring social disparities in cancer with an example using trends in lung cancer incidence by area-socioeconomic position and race-ethnicity, 1992–2004, Am J Epidemiol, 2008;167(8):889–99.
- Cook ED, Moody-Thomas S, Anderson KB, et al., Minority recruitment to the Selenium and Vitamin E Cancer Prevention Trial (SELECT), Clinical Trials, 2005;2(5):436–42.
- Williams RA, Eliminating Healthcare Disparities in America: Beyond the IOM Report, 2007;378.
Further Resources
Trending Topic
Cancers of the oesophagus and oesophagogastric junction (OGJ), termed upper gastrointestinal (UGI) cancers, are lethal and constitute a significant public health problem. In 2020, 604,000 cases and 544,000 UGI cancer-related deaths were estimated worldwide.1 UGI cancers are the seventh most frequently diagnosed cancer and the sixth most common cause of cancer-related death.1 In the USA, there were […]
Related Content in Diagnostics and Screening

Cancers of the oesophagus and oesophagogastric junction (OGJ), termed upper gastrointestinal (UGI) cancers, are lethal and constitute a significant public health problem. In 2020, 604,000 cases and 544,000 UGI cancer-related deaths were estimated worldwide.1 UGI cancers are the seventh most frequently diagnosed cancer ...

Somatic DNA mutational analysis has transformed how we characterize existing cancer and select treatments. For example, circulating tumour cell-free DNA (cfDNA) can be used to select targeted therapies, monitor treatment response and detect disease recurrence.1 However, early attempts with targeted ...
Molecular diagnosis and screening of cancer can be approached either in a broad, high-volume manner, or through using targeted, informed searches for specific genes. Technique selection will be critical in balancing patient benefit against healthcare resource cost, and physicians will ...
Over the last decade, genomics has become an increasingly important part of cancer care. In some tumor types, such as lung cancer and melanoma, genetic profiling for actionable mutations has become standard clinical practice.1,2 The development of novel targeted molecular ...
It has been clearly established that non-small cell lung cancers (NSCLCs) associated with known molecular drivers (e.g., rearrangement of the ALK gene, or mutations activating the epidermal growth factor receptor [EGFR] gene) respond favorably to targeted, first-line therapies compared ...

Q. What are the limitations of current methods of detecting biochemical recurrence of prostate cancer? The recommended imaging techniques for the diagnosis of metastases in prostate cancer are 99m-Tc bone scintigraphy for bone metastases and computed tomography (CT) or magnetic ...
Adjuvant systemic treatment of breast cancer is moving away from the limited portfolio of traditional hormonal drugs and chemotherapy, towards a gamma of novel anti-hormonal and chemotherapeutic drugs, immunotherapeutic approaches and small molecules. All these therapeutic approaches are unfortunately effective ...

Over the past several years, there has been a profound paradigm shift for those at high risk for lung cancer due to the development and release of scientific validation and national guidelines and recommendations for lung cancer screening. The lung ...

The rate of childhood cancer caused by a genetic predisposition is now estimated around 8.5%.1 While proposing a genetic counselling has been classically considered as part of good clinical practice for few malignancies (retinoblastoma, adrenocortical tumours, choroid plexus carcinoma), its actual ...
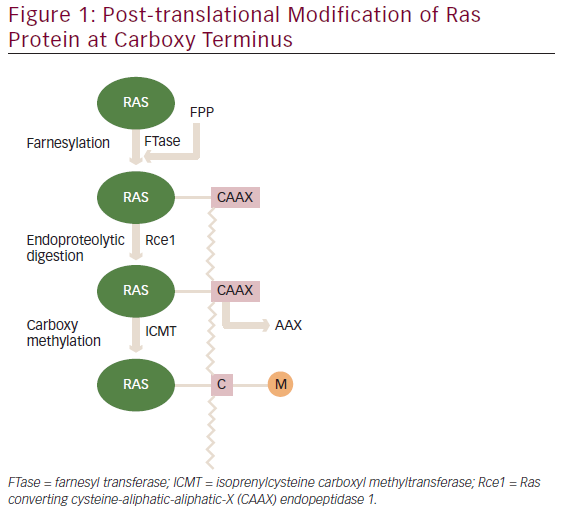
H-Ras, K-Ras, and N-Ras are the main members of Ras superfamily, which bind small molecules GTP and GDP interchangeably and can hydrolyze GTP to GDP. The Ras superfamily consists of more than 150 proteins and these can be classified under at ...

Advancing technology has led to the successful utilization of nextgeneration sequencing (NGS) for assessing cancer susceptibility, diagnosis, prognosis, and treatment.1,2 Currently, matching therapies that target single-nucleotide variations, gene fusions, or copy number variations are approved for clinical use across various ...

Oncologists are facing challenging times. Cancer is forecast to become the leading cause of death in the US fairly soon, and is already the leading cause of disability-adjusted life years lost.1 As the workload of the practicing oncologist inevitably increases, ...
Latest articles videos and clinical updates - straight to your inbox
Log into your Touch Account
Keep track of your clinical interests and newsletter subscriptions.
Register now for FREE Access
Register for free to hear about the latest expert-led education, peer-reviewed articles, conference highlights, and innovative CME activities.
Sign up with an Email
Or use a Social Account.
Register now for FREE access
Already registered? Login below.

