For decades, the standard treatment has been whole brain radiotherapy (WBRT), resulting in symptomatic improvement in the majority of patients 6 but longterm local control has been poor (0% to 14% at one year).7,8 A review of survival following WBRT in a selected large series of patients reported over the last 30 years indicates little change in the results.6,9,10 Approximately 25% to 50% of patients treated with brain irradiation will eventually die from intracranial progression.9,11 The incidence of brain metastases has increased over time, probably as a result of advances in neuroimaging procedures and improvements in the treatment of primary tumour and systemic disease, which has led to an increase of survival. Treatment for brain metastases patients includes corticosteroids, anticonvulsants to control seizures, surgery, radiotherapy, radiosurgery and chemotherapy. The appropriate aim of treatment is improvement or maintenance of quality of life. Surgical resection followed by WBRT may yield longer survival but is generally reserved for a minority of patients with a single brain lesion, well controlled primary disease and good prognosis.
Despite the recent advances in the diagnosis and treatment of brain metastases, it remains difficult to document an important change in overall outcome, neurological function and quality of life in this cohort of patients.
Diagnosis
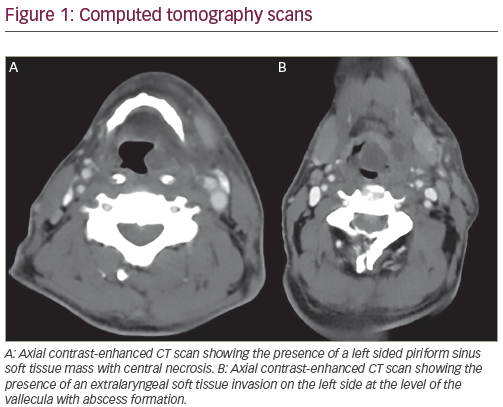
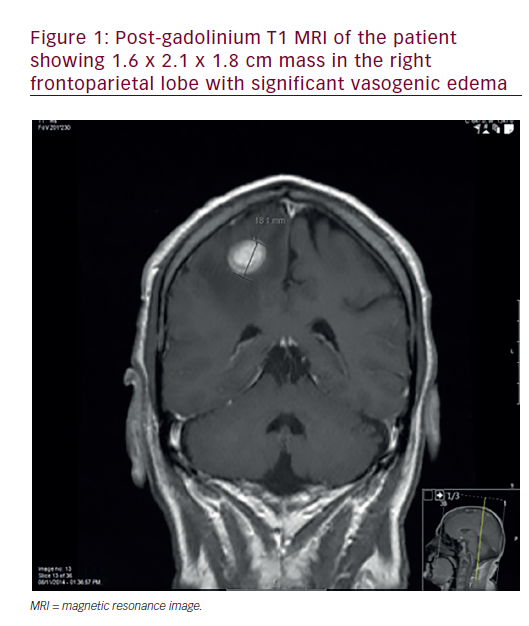
The clinical presentation of brain metastases is similar to that of patients with any intracranial mass lesion. Most patients experience debilitating neurologic symptoms, including headaches, focal weakness, cognitive dysfunction or altered mental status; 20% or more develop seizures. Severe neurologic dysfunctions frequently develop in patients, and up to 50% will die as a direct consequence of cerebral metastases. For many patients, brain metastases causes relatively mild symptoms and some may die as a result of progressive systemic, extracranial disease. One-third of the patients developing symptomatic brain metastases do not have any previous cancer history.12,13 The reports in the literature refer to patients in whom brain metastases were the first symptom of tumour discovered at diagnosis and to patients in which the primary site remained undetected after a thorough investigation. Contrastenhanced MRI is more sensitive than enhanced CT scanning in detecting brain metastases, particularly small lesions or metastases situated in the posterior fossa.14,15 MRI is particularly recommended for patients with an apparently single metastasis on a CT or for patients with limited disease (i.e lung tumours) in whom the detection of asymptomatic brain metastases would alter the therapeutic management. Radiographically, metastases are ring-enhancing lesions, most often located at the grey-white matter junction followed usually by significant oedema. About half of brain metastases are single lesions, the remainder being multiple. In the majority (80%) of patients, brain metastases develop after diagnosis of systemic cancer (metachronous presentation). When a brain metastasis from an undetected primary site is discovered at the first investigation on CT scan or MRI, it should be considered carefully. In most cases, the primary tumour is located in the lung and a chest CT scan is always recommended.16.
Surgery
In the past, few patients with brain metastases were considered for surgery. The role of surgical resection has increased with the improvements in anaesthesia, surgical technique and the recognition of benefit in selected patients mainly with single brain metastases. The goal of surgery is to provide immediate relief of symptoms, to establish histologic diagnosis and to improve local control. A series of trials 17,18,19 comparing patients who received surgical resection, with or without radiotherapy, have shown that combined treatment increases the median survival time to nine and 10 months versus three to six months for patients undergoing radiation treatment alone. Patchell reported in a randomised trial that resection was superior to WBRT in well selected patients. A critical prognostic factor was the degree of control of the primary tumour.17 The role of postoperative WBRT in single brain metastases was addressed in a randomised study by Patchell,20 where he demonstrated that recurrence of tumour in the brain was less frequent in the group of patients treated with surgery and WBRT than the group who received surgery alone. Patients treated with surgery had a lower incidence of local relapse (20% versus 52%) and a longer time of functional independence. Despite these encouraging results, few patients can benefit from surgical resection due to tumour location in a surgically accessible region, size, age and extensive systemic disease.
Complete surgical resection offers an immediate relief of symptoms of intracranial oedema and of seizures and a reduction of focal neurological deficits.
Radiotherapy
Whole brain radiotherapy has traditionally been the standard treatment for patients with brain metastases since 1950. WBRT has been shown to effectively improve neurologic symptoms and function for patients with minimum co-morbidity. This palliative approach does not offer survival benefit and the median survival ranges from three to six months. The published overall response rate is symptomdependent but ranges from 64% to 85%.21,22 Complications of modern WBRT are relatively uncommon and acute toxicity is unusual. Hypofractionated treatments as 30Gy in 10 fractions for two weeks are generally employed because of short life-expectancy. Several recent trials failed to demonstrate improvement of both local control and survival by dose escalation.23,24 The reason might be either suboptimal patient selection or failure to achieve local control due to application of insufficient total doses. The optimal dose fractionation schedules for patients with brain metastases have been evaluated in randomised trials conducted by the Radiation Therapy Oncology Group (RTOG).25 The median response and median survival were equivalent in all arms of these studies. Median survival ranged between 15 and 18 weeks and brain metastasis was the cause of death in 40% of the patients. While the RTOG trials failed to identify the best fractionation schedule, they enabled the identification for clinical factors associated with better survival.26 The RTOG developed three prognostic classes for brain metastases using recursive partitioning analysis (RPA) of a large database. These classes were based on Karnofsky performance status (KPS), primary tumour status, presence of extracranial systemic metastases and age. RPA class I included patients with KPS >70, less than 65 years of age with controlled primary and no extracranial metastases. Patients with KPS <70 were classified as RPA class III and all others were class II. This classification made the comparison between different trials possible and enabled the selection of aggressive treatments in specific patient populations. In patients with good prognosis who are likely to survive more than one year, a more protracted radiation schedule (eg, 40Gy in 2Gy fraction) may offer better response and better progression-free survival. Stereotactic Radiosurgery
Stereotactic radiosurgery is a technique of external irradiation that utilises multiple convergent beams to deliver a high single dose of radiation to a radiographically discrete treatment volume.27 Radiosurgery has several advantages over surgery. It can be used to treat metastases in surgically inaccessible areas of the brain, such as the brainstem. Because radiosurgery is a non-invasive procedure it is associated with less morbidity than surgery. In a multi-institutional trial 28 involving 116 patients treated with radiosurgery for a single brain metastasis, local tumour control was obtained in 99 patients (85%). Multivariate analysis showed that better local control was obtained in patients who received (WBRT) in addition to radiosurgery and in patients with radioresistant histologies (melanoma and renal carcinoma).
In a recently reported RTOG 95-08 trial the benefits of radiosurgery for patients with single brain metastasis were demonstrated.29 In this trial, 333 patients from 55 institutions with single brain metastasis were entered and were randomised to receive whole brain irradiation (37.5Gy in 2.5 fractions) with or without radiosurgery. Improved survival was found in 186 patients with single brain metastasis receiving radiosurgery (6.5 months versus 4.9 months, p=0.04). There was an important finding that patients receiving radiosurgery were more likely to improve and maintain their KPS. Radiosurgery combined with WBRT is recommended in patients with small lesions up to three in patients with good performance status and well controlled primary disease. Radiosurgery also has an important role in patients who experience recurrence of brain metastases following WBRT.
Chemotherapy
The role of chemotherapy in the treatment of patients with brain metastases has not been clearly defined. Traditionally, it has been assumed that the blood–brain barrier prevented chemotherapeutic agents from entering the central nervous system (CNS).30 However, there is evidence that the blood– brain barrier is partially disrupted within the brain tumours, which is easily recognised by MRI or CT with an intravenous contrast; if the barrier was intact it would prevent the accumulation of contrast inside the lesion. This suggests that other factors also contribute to the generally disappointing results of chemotherapy for brain metastases. These may include the intrinsic resistance to chemotherapy of many tumours that metastasize to the brain and the tendency for brain metastases to develop following the failure of primary chemotherapeutic agents to control the primary disease. Although the results of chemotherapy for brain metastases have generally been disappointing, a number of uncontrolled studies have demonstrated favourable response rates of brain metastases from chemosensitive tumours, such as breast cancer, small cell lung cancer and germ cell tumours.
The development of new chemotherapeutic agents with better penetration (i.e. topotecan, temozolomide (TMZ)) has renewed the interest in using chemotherapy to treat brain metastases.31 TMZ is a novel oral alkylating agent with demonstrated activity in primary and recurrent gliomas,32,33 it crosses the blood–brain barrier 34 and is highly bioavailable after oral administration,35 achieving effective concentrations in the CNS. The adverse event is mainly myelosuppression; however, the incidence of grade 3/4 neutropaenia and thrombocytopaenia is generally less than 10%.36In a phase II study reported from the Memorial Sloan Kettering Cancer Center, TMZ was administered in patients with recurrent or progressive brain metastases from a variety of primary cancers including non-small cell lung cancer and breast cancer.37 In this trial, 34 evaluable patients received temozolomide. In two patients the brain metastases had a partial response, while in 15 patients the brain lesions remained stable. The median overall survival was 6.6 months. A second study of 28 patients (12 with non-small cell lung cancers), where most patients had received both chemotherapy and whole brain irradiation, found a partial remission in one patient with brain metastases from a non-small cell lung cancer.38
Concurrent administration of TMZ with conventional whole brain irradiation (RT) was investigated in a phase II study from the Metaxas Cancer Hospital, Greece. In this trial, 52 patients with brain metastases from a variety of primary tumours (among whom 31 had non-small cell lung cancer, nine small cell lung cancer and five had breast cancer) were randomised to whole brain irradiation and a total dose of 40Gy with a daily fraction of 2Gy with or without concurrent administration of TMZ.39 TMZ was administered orally at a dosage of 75mg/m2 during radiation treatment and 200mg/m2/d X five days every 28 days for a maximum of six additional cycles. Treatment response was assessed on the basis of CT scanning or MRI two months after completion of radiation treatment according to the World Health Organization (WHO) criteria for response. Fortyfive patients were assessable for response. The objective response rate in the group receiving TMZ (96%) was significantly superior (p=0.017) to that achieved with RT alone (67%). Another measure of treatment efficacy is the requirement for medication to palliate neurologic symptoms. The proportion of patients who required corticosteroids in the TMZ+RT group, two months after the completion of RT, decreased from 100% to 67% compared with a decrease from 100% to 91% in the RT group. Patients treated with TMZ plus RT had a slight improvement in overall survival (8.6 months) compared with RT alone (7.0 months).The addition of TMZ to RT was generally well tolerated. There was no grade 3 or 4 myelosuppression.
In a phase III study 40 concurrent administration of temozolomide with whole brain radiotherapy was investigated further. In this trial, 123 patients with brain metastases were randomised to TMZ and RT or RT alone. The total radiation dose was 30Gy, with daily fraction of 3Gy. TMZ was administered with the same schedule as in the previous trial. Treatment response was assessed with CT scanning or MRI three months after completion of the radiation treatment. One hundred and twenty-three patients were assessable for response, among them 103 lung cancer patients. The objective response rate in the group receiving TMZ (50%) was significantly higher (p=0.028) to that achieved with RT alone (31%). The evaluation of lung cancer patients gave similar results, the response rate in the TMZ group (48%) was significantly superior (p=0.031) to that of the RT alone group (27%). The hazard ratio for death from any cause in the temodal group compared with the control group was 0.69 (95%CI: 0.46 -1.02, p=0.06). The median survival in the TMZ+RT group was 7.87 months while in the RT group it was 4.93 months. In the subgroup analysis for the lung cancer patients, median time to progression in the brain was 4.8 months (95% CI: 1.71–9.18) for the RT group, while more than 50% of the patients in the TMZ+RT group were disease-free. There was a significant difference between the two groups (p<0.001).
In summary, metastases in the CNS are common and new treatment modalities are needed. The combination of TMZ with radiation therapy needs to be confirmed in a large phase III international trial.