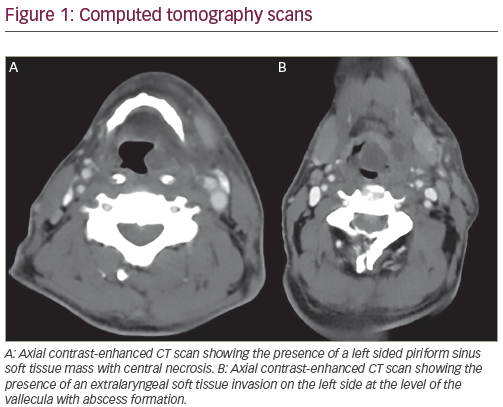
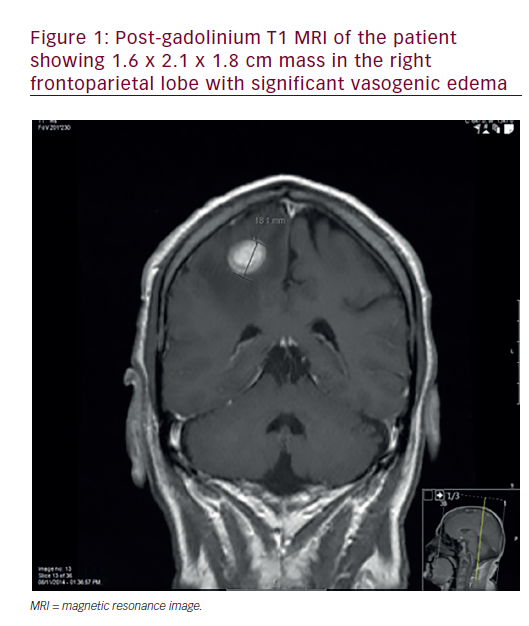
Despite advances in multimodality treatment, the 5-year survival rate of patients with locally advanced head and neck squamous cell carcinoma (HNSCC) does not exceed 40–50% and survival rates for recurrent or metastatic disease (R/M) remain dismal.1,2 Low survival rates in combination with significant toxicities caused by current treatment strategies used in HNSCC emphasise the necessity for new treatment options.
For patients with recurrent or metastatic disease, recommended treatment is cisplatin-based chemotherapy ± cetuximab for fit patients and single agent chemotherapy (methotrexate) or cetuximab for patients with poor performance status (PS), albeit with limited efficacy.3 In this setting, strategies to improve outcome include the introduction of novel therapeutic strategies and molecular targets, as well as intensification of treatment. The epidermal growth factor receptor (EGFR), a transmembrane tyrosine kinase receptor belonging to the HER/ErbB family, is a long-standing challenging target in HNSCC, as it is overexpressed in up to 90 % of cases;4 overexpression of EGFR also correlates with poor clinical outcomes.5 Cetuximab, a chimeric immunoglobulin G1– human monoclonal antibody against the extracellular domain of EGFR, has emerged as a powerful tool in the treatment of R/M head and neck cancer. In the Erbitux in First Line Treatment of Recurrent or Metastatic Head and Neck Cancer (EXTREME) study, the addition of cetixumab to platinum-based chemotherapy with fluorouracil was shown to improve overall survival (OS), progression-free survival (PFS) and response rates.6 However, cetuximab has demonstrated modest response rates when used as monotherapy.7 Afatinib, an oral irreversible ErbB family blocker that inhibits all kinase-active members (EGFR, HER2 and HER4), which is currently approved for the treatment of EGFR mutated non-small-cell lung cancer,8 has been shown similar antitumour effects compared with cetuximab in R/M HNSCC in a phase II trial.9
Based on these results, the phase III LUX-Head and Neck 1 clinical trial presented at the European Society for Medical Oncology (ESMO) Congress 2014 assessed the efficacy of afatinib as monotherapy compared with single agent methotrexate as second-line treatment in HNSCC.10 Patients were stratified according to PS and prior use of cetuximab. The study met its primary endpoint showing an increase in PFS of 0.9 months with afatinib compared with methotrexate (2.6 versus 1.7 months, hazard ratio (HR) 0.8; p=0.03). In addition, it showed improvement in tumour shrinkage and response rate in favour of afatinib.10 The difference in PFS of 0.9 months is of unclear significance and unlikely to lead to drug approval. However, this study is important, as it is the second study since the EXTREME trial that showed benefit of a novel agent in HNSCC and the first study to demonstrate an active oral targeted agent in HNSCC. Of note, subgroup analysis showed a benefit primary seen in EGFR-naïve patients, suggesting a degree of cross-resistance that was not seen in phase II studies.9 Ongoing studies evaluating adjuvant afatinib in locally advanced HNSCC after chemoradiotherapy (LUX-Head and Neck-2) will hopefully clarify the effectiveness of afatinib;11 until then, active search of predictive biomarkers might lead to identification of specific groups of patients with greater benefit.
Another rationale for improvement of treatment strategies in HNSCC is to overcome resistance to cetuximab, which might lead to tumour progression after initial clinical response. One proposed mechanism of de novo or acquired resistance in HNSCC is HER3 activation.12 MEHD7945A is a monoclonal antibody that simultaneously blocks ligand binding to EGFR and HER3 and inhibits signalling by all major liganddependent HER family members.13 It has shown superior preclinical activity compared with mono-specific HER antibodies, particularly in HNSCC and has displayed activity in cetuximab-resistant models.13
Furthermore, HER3 ligand neuregulin 1 (NRG1) is overexpressed in HNSCC and may select for benefit in dual inhibition of EGFR/HER3.14 In a phase II clinical trial presented in ESMO 2014, MEHD7945A was compared with single agent cetuximab as second- or further-line therapy in patients with R/M HNSCC not suitable for local therapy.15 The study demonstrated a comparable, albeit not superior, efficacy of MEHD7945A compared with cetuximab, with similar response rates and PFS. Unfortunately, NRG1 was not confirmed as a biomarker, as high NRG1 expression did not correlate with MEHD7945A efficacy. Toxicity was similar between the two groups, with higher gastrointestinal (GI) toxicity observed in the MEHD7945A group, consistent with other regimens targeting multiple HER receptors.15
On the other hand, there is a great body of evidence supporting that the immune system plays an important role in cancer development, as tumour cells exploit inhibitory checkpoint pathways that suppress antitumour T-cell responses.16 In this setting, the programmed cell death (PD-1) pathway blocks the immune response by downregulating T-cell effector functions. Programmed cell death ligand 1 (PDL-1) is overexpressed in tumour cells, including HNSCC, and activates the PD-1 checkpoint pathway by binding to the PD-1 receptor, resulting in inhibition of antitumour T-cell responses.17 Two anti-PD-L1 monoclonal antibodies, MEDI4736 and MK3475, have been evaluated in recent phase I trials.18,19 MEDI4736 has been shown to have an overall response rate (ORR) in approximately 11 % of patients, with a manageable safety profile (grade ≥3 adverse events [AEs] in 7 % of patients).18 MK3475 has demonstrated response rates as high as 20 % albeit with greater toxicity (grade ≥3 AEs in 17 % of patients).19 Of note, some responses are durable, but specific biomarkers are lacking.
It is important to emphasise that HNSCC is currently divided into two distinct entities in terms of aetiology and clinical behaviour. Tobacco/ alcohol consumption-associated HNSCC is described as human papilloma virus (HPV) negative (HPV-) subset and is usually p16 negative. Infection with high-risk HPVs is linked to a growing proportion of oropharyngeal cancers (HPV positive [HPV+] HNSCC). HPV+ HNSCC is associated with a better prognosis and might require different treatments.20 In addition, HPV status has prognostic significance in recurrent/metastatic setting as well.21,22 As a result, current clinical trials attempt to assess benefit of investigational agents separately in HPV- and HPV+ HNSCC. In the LUX-Head and Neck 1 study, HPV- patients were found to have greater benefit from afatinib therapy.10 The Study of Panitumumab Efficacy in Patients With Recurrent and/or Metastatic Head and Neck Cancer (SPECTRUM) also showed that the benefit from the addition of the fully human anti-EGFR antibody panitumumab to cisplatin/5-fluorouracil (5-FU) chemotherapy was restricted to p16-negative patients23 while the analysis of the EXTREME study showed that both HPV+ and HPVpatients derive benefit from the addition of cetuximab to platinum/5- FU chemotherapy.6 Anti-PDL-1 antibodies had conflicting results, with MEDI4736 showing greater benefit in HPV- tumours18 while MK3475 demonstrated equal efficacy in both.19
Finally, intensification of chemotherapy treatment protocols is another rationale for potential improvement of outcomes in HNSCC. However, treatment-related toxicity in HNSCC remains a matter of major importance. Final analysis of the AIO trial has shown that addition of docetaxel to EXTREME regimen of cisplatin, 5-FU and cetuximab not only does not improve outcome compared with EXTREME regimen but also increases GI toxicity and infections.6,24
In conclusion, the prognosis of patients with R/M HNSCC remains dismal. As intensification of chemotherapy protocols carries the risk of unacceptable toxicity: the future lies on the introduction of novel molecular-targeted agents. Identification of predictive biomarkers for patient selection for targeted therapies including immunotherapy is essential. Importantly, future studies should focus on assessing effectiveness of investigational treatments according to HPV status.