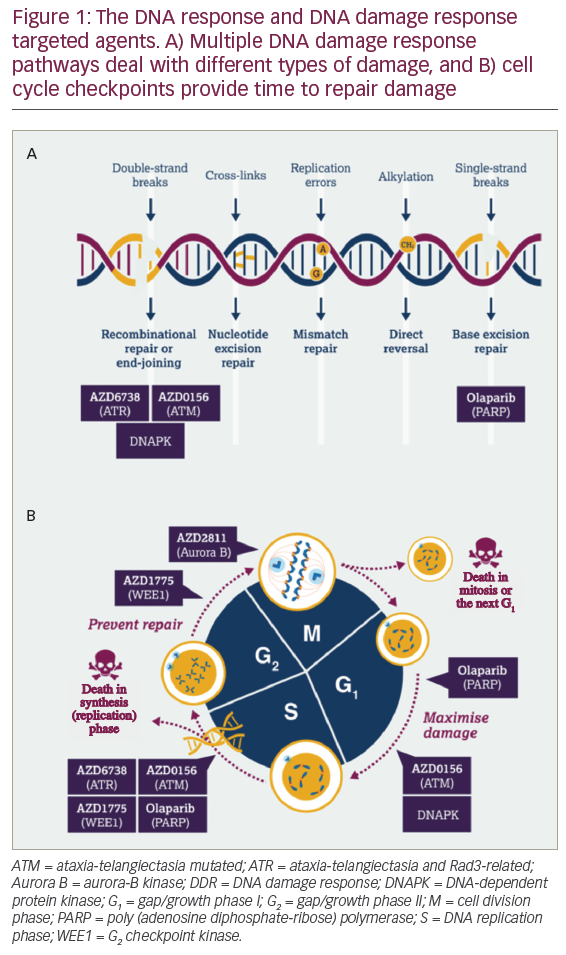
Breast cancers which lack expression of the estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor 2 (HER2)—so-called triple-negative breast cancers (TNBC)—make up 15–20% of the more than 230,000 cases of breast cancer diagnosed in the US each year.1 Metastatic TNBC remains an important clinical challenge as it is associated with a median survival of under 12 months. While breast cancer has not historically been thought of as an immunogenic tumor, there have been a number of observations made over the past decade that have provided the rationale for investigating immunotherapy as a viable therapeutic option for TNBC.
Loi and colleagues have observed that ER-negative breast cancers have a higher density of tumor infiltrating lymphocytes (TILs) than their ER-positive counterparts.2 The ability to profile breast tumors on a molecular level has led to two key observations: first, that programmed death ligand 1 (PD-L1) gene expression is significantly greater in TNBCs compared with non-TNBCs,3 and second, that a subset of TNBC tumors—the immunomodulatory subtype—is characterized by elevated expression of genes involved in T-cell function.4 Furthermore, TNBCs are genomically unstable and have high rates of genetic mutations, which can in turn lead to neo-antigen presentation and induction of an immune response through activation of the cancer-immunity cycle.5,6
The cancer-immunity cycle is a complex, multi-step, self-propagating cycle, which allows for the identification and eradication of cancer cells through the induction and amplification of T-cell responses. Key steps in the process include:
• release of cancer cell antigens;
• presentation of these antigens by antigen presenting cells;
• priming and activation of the T-cell response;
• trafficking of T-cells to tumors;
• infiltration of the tumor by T-cells;
• T-cell recognition of cancer cells; and
• cancer cell death, which in turn leads to antigen release (step 1), and propagation of the cycle.
Each step of this cycle is tightly regulated by both stimulatory and inhibitory factors, which lead to a delicate balance between immunity and unrestrained autoimmunity.
For many human cancers, the cancer-immunity cycle is intact up to the point of tumor cell killing by T-cells. Programmed death 1 (PD-1) is expressed primarily by activated T-cells, and binding of PD-1 to its ligands PD-L1 or PD-L2 has been show to impair T-cell function. PD-L1 is overexpressed by tumor cells and macrophages, and tumors can coop the PD-1/PD-L1 pathway to evade immune surveillance. Once the PD-1/PD-L1 interaction is blocked, preexisting anticancer T-cells can have their effector function rapidly restored.6 This concept has led to the development of a number of checkpoint inhibitors that target the interactions between PD-1 and PDL1. These anti-PD-1 and PD-L1 agents have now been tested in multiple tumor types with promising efficacy being seen in several tumor types, including melanoma and lung cancer. The PD-1 inhibitor pembrolizumab, and the PD-L1 inhibitor atezolizumab have both been studied for the treatment of PD-L1-positive TNBC.
The Study of MK-3475 in Subjects With Advanced Solid Tumors (KEYNOTE-012) study was a multi-cohort, phase Ib study of pembrolizumab in patients with PD-L1-positive advanced solid tumors, including triple-negative breast, head and neck, urothelial, and gastric cancers. The results of the TNBC cohort were presented at the San Antonio Breast Cancer Symposium in December 2014. Thirty-two women were enrolled in this study (median age 50.5 years, range 29–72) and were evaluated for safety and efficacy. Patients were heavily pre-treated, with almost 90% of patients having received therapy for early-stage disease, and 46% of patients having three or more prior therapies for metastatic disease.7 Treatment-related adverse events (AEs) were common, but generally mild and easily managed. The most common included arthralgias (18.8%), fatigue (18.8%), myalgias (15.6%), and nausea (15.6%). Grade 3 AEs included anemia, headache, asceptic meningitis, and pyrexia (n=1 in each). One grade 4 AE occurred and this was decreased blood fibrinogen ultimately resulting in death, and this was the only treatment-related AE that led to discontinuation. Pembrolizumab was associated with an overall response rate (ORR) of 18.5%, with one patient having a complete response (CR) and four patients having a PR. Responses were durable, with the median response duration not being reached (range 15 to 40+ weeks), with three of five responders on treatment for ≥11 months.7
Atezolizumab is an engineered human monoclonal antibody targeting PD-L1 that inhibits the binding of PD-L1 to both PD-1 and B7-1. The efficacy of atezolizumab has already been demonstrated in numerous tumor types, including bladder cancer, non-small cell lung cancer, renal cell carcinoma, and melanoma.8 A phase Ia TNBC cohort of a multi-cohort study was presented by Emens and colleagues at the American Association of Cancer Research meeting in 2015.9 A total of 54 patients were enrolled (median age 53 years, range 29–82). Patients were heavily pre-treated, with 89% of patients having been exposed to ≥4 systemic therapies. Similar to the pembrolizumab study, treatmentrelated adverse events were common, with 63% of patients experiencing a treatment-related AE of any grade. These were typically grade 1–2 and were easily managed, with the most common AEs including fatigue, nausea, and pyrexia. Eleven per cent of patients were found to have grade 3 treatment-related AE, and two deaths were reported and under further investigation at the time of presentation.9 Of the 21 patients considered evaluable for efficacy, the ORR was 19%, which included two CRs and two PRs. Responses were durable, with a median duration of response not yet reached at the time of presentation (range 18–56+ weeks).9
Blockade of the PD-1/PD-L1 axis has now been studied in two phase I clinical trials in TNBC and has resulted in promising, durable responses. In addition, treatment with these immune checkpoint agents appeared safe and tolerable, with the majority of side effects being mild and easily managed. Immunotherapy has arrived as an important tool in the fight against cancer, and will likely be an important treatment option for TNBC. Tumors evolve to evade the immune system in different ways and at different steps in the cancer-immunity cycle, and immune checkpointinduced T-cell anergy is relevant in TNBC. Combination therapies are exciting, and hold the promise of building on the modest responses seen with anti-PD-1/PD-L1 monotherapy. However, perhaps the greatest promise for the future is working to identify what limits effective immunity in an individual tumor, thus allowing for the greatest antitumor activity while limiting unrestrained autoimmunity.