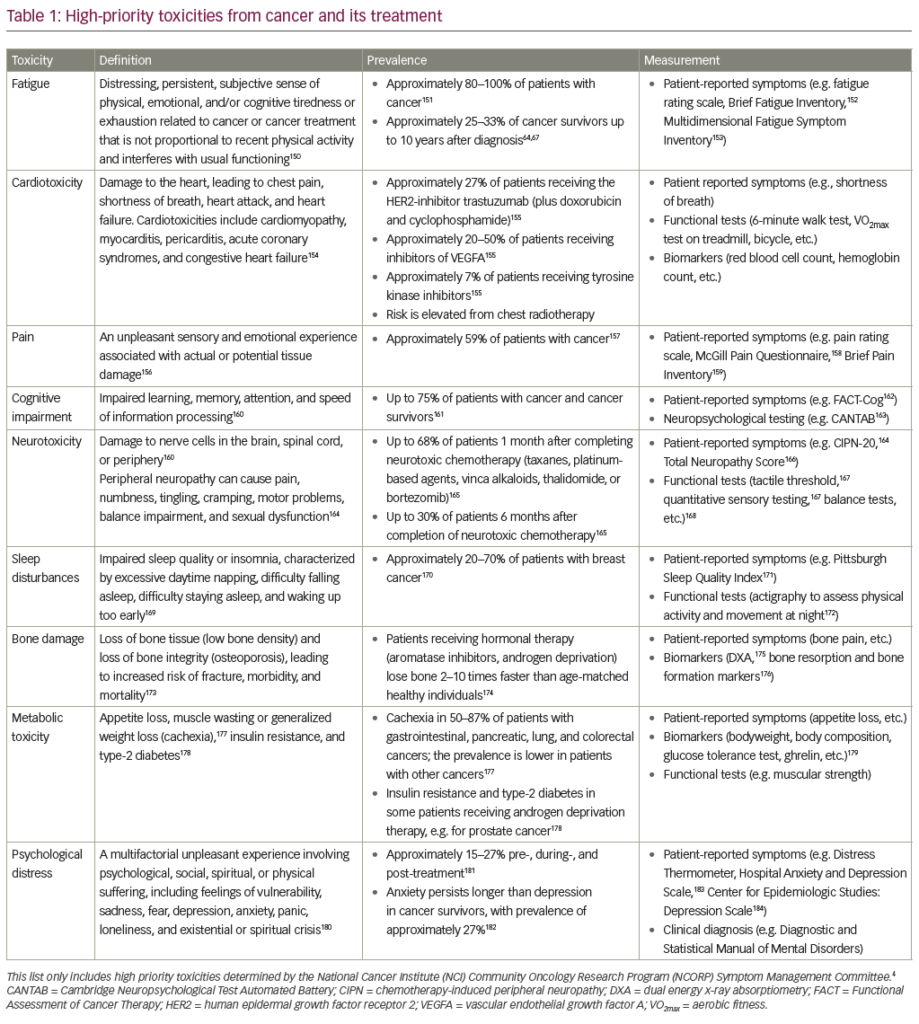
The current treatment for most cancerous growths consists of surgery, radio- and chemotherapy or combinations. The underlying rationales for those options remains, therefore, virtually unaltered since, respectively, the times of the Pharaohs.1 Emil Grubbe irradiated the first breast cancer in 18962 and mustard gas variants were first used therapeutically in 1946.3 These three strategies have in common that they are aimed at eradicating the maximum amount of cancerous growth in the shortest possible time. In the case of therapeutic options two and three by inducing lethal toxicity and thus killing the cancer cells, predominately via a form of programmed cell death termed apoptosis.4 Importantly, once a tumour has disseminated and metastasised, chemotherapy is the only of the three treatment options mentioned above that can be applied systemically, immunotherapy being an additional future option.
While there is no doubt that these strategies have been tremendously successful – over the last few decades, for example, mortality rates for several cancers have decreased by 30–60 %5 – cancer is still among the leading causes of death worldwide6 and – perhaps surprisingly, since we consider cancer a disease of old age – it is the most common cause of death in children and among the four most common causes of deaths in teenagers and young adults.7 While in absolute numbers quite rare, in terms of years of life lost and effects on quality of life pediatric cancers are clearly among the most devastating diseases faced by today’s clinicians.
Yet, the scope of further improving current therapeutic approaches seems rather limited. Large-scale sequencing efforts that delineated the mutational landscape of several cancer entities have yielded little with regards to potential new target molecules,8–11 while the use of small inhibitory molecules, for example the targeting of the phosphatidylinositol 3’-OH kinase (PI3K)/ v-akt murine thymoma viral oncogene homologue (Akt)/ mammalian target of rapamycin (mTOR) survival pathway, has shown some preclinical promise, but is far from being a paradigm shift in the clinic.12 The therapeutic use of pharmacologically active small molecules, such as tyrosine kinase inhibitors (TKIs), is most likely to be within the setting of combination therapy with existing chemo- and radiotherapy, as cancer cells have demonstrated the ability to rapidly develop resistance to those components, as seen, for example, in imatinib monotherapy.13,14
Interestingly, resistance can be mediated by de novo mutation, emergence of pre-existing rare sub-populations or via epigenetic alterations.15 Our only available responses to treatment failure are altering the treatment and increasing the dosage until the maximal tolerable dose is reached. Chemotherapy in particular is also frequently associated with significant side effects, which are often perceived by patients as worse than the actual disease, and have in the past led to the termination of treatment, particularly in under-aged patients.16 However, even with treatment success, long-term adverse effects and treatment-induced secondary cancers are, with extended life expectancies, also becoming increasingly a problem.16
Recent advances in the analysis of whole cancer genomes and protein expression patterns,17 combined with the increasing acceptance of the cancer stem cell model18 and increasing appreciation of the tumour microenvironment,15 have put to rest the notion of tumours as monolithic entities. We now understand cancer as a complex mixture of occasionally cooperating, occasionally competing subpopulations of cells that no longer necessarily harbour distinct oncogenic mutations. In essence, our understanding of what constitutes a tumour has drastically changed/been expanded and this new, encompassing view of cancer as an ecological system comprising mutated cancer cells and non-mutated cells of the microenvironment that, taken together, constitute a tumour, opens novel therapeutic avenues.
New Therapeutic End Points
While the tumour stem cell concept was first alluded to in 193719 it has only gained dominance in the last two decades, and its implications are still not fully understood. It seems generally agreed upon that somatic mutations and other forms of evolutionary pressure, such as the niche effect,20 can lead to tumour heterogeneity that even extends to the highest hierarchical order, that of tumour stem cell.21 Our therapeutic decisions are invariably based on the (molecular) histological features of the dominant, i.e. most numerous, sub-clone present. However, several lines of evidence have recently demonstrated that ignoring other, non-dominant sub-clones can have dire therapeutic consequences. Taking our cue from population genetics and ecology, we tend to assume a non-dominant, minor clone within a tumour has been out-competed by the dominant one and thus will be lost from the population within coming generations. However, sub-clones are not necessarily in direct competition; on the contrary, they have been shown to cooperate,22 and tumour growth can even be driven by a minor sub-clone.23 Importantly, different intrinsic chemosensitivities exist within the different subpopulations,24 implying that treatment resistance can and will arise, not necessarily due to the acquisition of de novo mutations, but a change in dominance of individual sub-clonal populations. We have argued elsewhere that the drastic environmental changes, i.e. the introduction of a lethal toxin in the form of drugs or radiation, changes the adaptive fitness requirements for a successful colonisation of an ecological niche.15 For example, mitochondrial dysfunction in tumour cells may have no evolutionary advantage in the absence of a potent selective force and lead to growth retardation due to reduced cell cycle progression.25 This sub-clone is unlikely to become dominant within a tumour. However, upon the introduction of a selective force, such as chemotherapy acting on the mitochondrial apoptosis pathway,4 the same mitochondrial deficiency might confer increased resistance to cell death,25 thus exerting a positive selective pressure, causing dominance of this particular sub-clone and thus the growth of a therapy-resistant tumour.
This, in turn, suggests the possibility of novel therapeutic approaches that take the aforementioned knowledge gleaned from population genetics and ecology into account.26 In particular, a stabilisation of the ecosystem of cancer should prevent the outgrowth of uncontrollable sub-clones, which would imply – in ecological terms – a regular culling of the dominant species to prevent its expansion into a neighbouring niche while maintaining the existing habitat, or – in therapeutic terms – chronifying the disease and blocking metastasis.
The mathematical and experimental foundations for a treatment based on these principles were put forward by Gatenby and co-workers in 2009. The adaptive therapy uses a reactionary schedule where treatment is only applied upon tumour growth and discontinued upon stabilisation, implicitly acknowledging the need to prevent sub-clonal outgrowth.27 This approach resulted in a considerable life extension in the experimental animals compared with standard therapy and – interestingly – resulted in increasingly longer gaps between treatments as time progressed.27 While generally speaking, a chronifying approach does not lend itself to pediatric cancers,16 it is, however, a variant thereof that shows clinical promise in the treatment of childhood glioblastoma. Glioblastoma multiforme is the most common tumour of the central nervous system in adults and unfortunately also frequently found in children.28 The current standard of care consists of tumour resection followed by radiotherapy and a course of temozolamide.29 However, with a mean patient survival of 15 months30 glioblastoma also remains one of the most lethal tumours. Pediatric glioblastoma is genetically similar, but distinct from the adult disease and while the importance of those differences is not fully understood, life expectancy is slightly better in children with this tumour.31 In our clinic we have introduced the rapamycin, irinotecan, sunitinib, temozolomide (RIST) protocol, which is a complex combination therapy consisting of combinations of chemotherapeutic agents and small molecule inhibitors and by metronomic dosage we could achieve a reduction of tumour burden with relatively few side effects.32 While patients are not cured and tumour recurrence occurs upon cessation of treatment, quality and quantity of life are greatly improved.32
Protocols that are aimed at the chronification of a malignancy should take ease of administration into account, ideally in the form of orally available medication, which eliminates the need for lengthy or regular clinic stays and improves patient compliance. However, it needs to be pointed out that the management of one of the diseases successfully controlled by chronification,33 type 1 diabetes, is based on regular subcutaneous injections of insulin. In addition, there are two further points of concern that need to be clarified or elucidated before one can consider the wider therapeutic application of a chronifying approach in cancer therapy. First, such a treatment is dependent on tight and regular monitoring of the tumour. Currently, this is time- and cost-intensive and not without logistic difficulties. In the case of glioblastoma, for example, magnetic resonance imaging (MRI) needs to be performed regularly, which can not be performed at a local general practitioner’s and thus necessitates that the patient travels a not inconsiderable distance to a clinic. Easier ways of monitoring tumour progression and composition are needed to make this option more attractive for patients and insurers. Interestingly, the emerging field of liquid biopsies might hold the answer to this particular problem.34,35 Second, which tumours should be treated with an aim of chronifying the disease, rather than curing the patient? With some malignancies at the extreme ends of the spectrum this appears to be rather obvious. For example, childhood leukaemias in general have a high cure rate, while glioblastoma do not, but these are rather exceptions? Hypothetically, should one suggest a cut-off point, e.g. if the chances of curing the disease are less than 50 % should one attempt chronification? How does one take patient age into account? Is it more acceptable to expect a 75-year old to take medication for the rest of his/her life than a 12-year old? These are important and difficult ethical questions, which need to be addressed before proceeding with such an approach. Crucially, a discussion in this regard cannot take place with a patient in attendance.
Targeting Cellular Interactions
Interestingly, one additional aspect that might make the RIST protocol a particularly suited tool for chronifying glioblastoma is that it targets the PI3K/Akt/mTOR pathway, which is activated in almost 90 % of all glioblastoma.36 This tumour entity is characterised by its highly invasive nature, within the boundaries of the brain, leading some to refer to glioblastoma as a systemic brain disease rather than a tumour within the brain (for example).37,38 Therefore, it is interesting to note that the PI3K/Akt/mTOR network not only mediates survival signals, but is also involved in cellular motility and invasion.12 Further studies are needed to ascertain whether the RIST therapy is – at least partially – effective in inhibiting invasion of tumour cells through their microenvironment. If this holds true, the treatment protocol should be expanded to targeting not the signalling pathway(s) activated by this interaction, but the interaction and the microenvironment itself.
It has been known for more than 40 years that resistance to cell death is not only mediated by acquired mutations, but also by cellular interactions, of which there are three distinct forms: homo- and heterotypic cell-cell – i.e. cancer cells interacting with each other, or non-cancerous cells, respectively – as well as cell-substrate adhesions.39 How failure to maintain these interactions leads to cell death is demonstrated in extremis by a process termed anoikis, the induction of caspase-dependent cell death upon loss of cell-substrate interaction.40 However more relevant in the context of cancer therapy is a phenomenon termed adhesionmediated apoptosis resistance (AMAR).15,39 AMAR has been found in a wide variety of malignancies where it can be induced by all forms of cellular interaction.15 Importantly, adhesions activate similar survival cascades as found mutationally activated in cancers; for example, the above-mentioned PI3K/Akt/mTOR pathway, the Bcl-2 family or inhibitor of apoptosis proteins (IAPs).15 It initially seems counter-intuitive to argue for targeting adhesions/interactions with the microenvironment, rather than the activated pathways within the cancer cells. As mentioned, these signalling networks can be either activated due to AMAR or mutations and epigenetic changes. Therefore the current strategy of targeting those signals directly41 should be more effective, as it affects the signal independently of the origins of its activation.
There are, however, several compelling reasons to consider targeting the microenvironmental interactions as preferable. First, taking the evolutionary perspective discussed above into account, it has been argued that targeted therapy does not work particularly well.42 Mutated cancer cells present a genetically highly unstable target and the increased selective pressure induced by blocking survival signalling frequently leads to the rapid development of resistant mechanisms.42 By contrast, the microenvironment, i.e. the part of a tumour that does not consist of mutated sub-clones, provides a much more genetically stable focus that is far less likely to develop resistances towards treatment.15 Second, if we are to understand the tumour microenvironment as those parts of the tumour that are not mutated cells, one must keep in mind that those components are nevertheless ‘abnormal’. Mutated cells influence the gene expression of cells in their vicinity, such as stroma cells or, as discussed below, macrophages. Vice versa these cells influence the classic tumour cells and mediate the main characteristics of the tumour. For example, some of the proteolytic proteins that mediate metastasis are provided not by tumour, but by stroma cells.43 Restoration of ‘normal’ microenvironment can prevent tumour progression and even formation,44,45 which is of particular interest if one considers the ‘premetastatic niche’. While the underlying dynamics of niche formation seem to be not without controversy,46 it appears that alterations of the microenvironment at a future site of metastasis can precede the arrival of mutant cancer cells.47,48 In particular, vascular endothelial growth factor receptor 1 (VEGFR1)-expressing haematopoietic precursor cells have been shown to home in on specific sites within the bone marrow, and form a niche there for migrating tumour cells.48 This indicates that targeting the interaction with the microenvironment can not only induce and/or sensitise for cancer cell death, but can also contribute to a more localised disease, either by inhibition of metastasis, thus, destroying potential refuge sites for cancer cells, or by blocking migration. An example of the latter is the formation of gap junctions between glioblastoma cells and astrocytes, which allows the cancer cells to use healthy cells as substrates during migration.49 Interestingly, not only heterotypic but also homotypic gap junctions can be found, and blocking these in an experimental setting can sensitise glioblastoma cells for chemotherapy and anoikis.50 Here, targeting one type of interaction, the gap junction, can have, depending on the microenvironment, different positive effects, i.e. the inhibition of migration and the induction of cell death. Importantly, we found that the points of interaction between tumour cells and their surroundings are not static: blocking one type of interaction, as in the previously mentioned example, the adhesion of glioblastoma cell to the extracellular matrix-protein fibronectin can lead to a switch in interaction towards the aforementioned gap junctions. Only by inhibiting both forms could cells be adequately sensitised for therapy.50 Therefore, one can easily imagine a combination therapy targeting multiple aspects of tumour cell/microenvironment interaction driving the cancer cell into isolation within the tumour.
The choice of target interaction is essential in this therapeutic approach, as loss of adhesion also occurs during initiation of invasion and metastasis. Epithelial-mesenchymal transition (EMT) describes the loss of cell polarity and cell–cell adhesion in epithelial cells, which then take on the characteristics of mesenchymal cells and become migratory and invasive.51 EMT is an essential process during several physiological events, but also contributes greatly to tumour progression and metastasis.52 Therefore it seems prudent to concentrate on the inhibition of forms of cell–cell interaction that are predominately modes of communication, like the gap junctions, and not forms of adhesion, like the adherens junctions.15 By contrast, targeting cell–substrate adhesion has been shown to be a highly effective way to block glioblastoma invasion53 and is currently being evaluated as a safe treatment option for several cancers.54
Focus on Tumour-associated Macrophages
To identify further potential targets within the tumour, the individual components that make up its microenvironment need to be better understood. To this group belong tumour-associated macrophages (TAMs) that appear to have a rather complex role within the tumour, being both promoters and inhibitors of tumour progression.
Variants of monocyte-derived macrophages can be found ubiquitously in the various tissues of a human body, where their function is to engulf foreign materials and consequently produce immune effector molecules. They have an essential role in, among others, antimicrobial defence, wound healing and tumorgenesis.55 Macrophages have usually been classified as anti-tumorigenic and pro-inflammatory M1 cells and pro-tumorigenic polarised M2 cells, which are involved in wound healing. While the role of (what are classically considered M1) macrophages in killing tumour cells has long been established56–58 and recently further highlighted in the context of mediating a bacteria-dependent immune response,59–61 it is the M2 phenotype that has been associated with TAMs.58 However, recent findings suggest that TAMs extracted from human pancreatic ductal adenocarcinoma are able to induce EMT in colorectal carcinoma cells, thus increasing the cancer cells’ migratory potential independently of M1 and M2 phenotype.62 While a recent meta-analysis suggested that high TAM infiltration in tumours was associated with a decrease in overall survival in terms of breast, gastric, bladder, ovarian and head and neck cancer, the data also indicated a more complex relationship between tumour cells and TAMs.63 In breast and bladder cancer TAM infiltration was found to be high at late clinical stages, ovarian carcinoma already displayed high TAM infiltration at a very early clinical stage, while high TAM infiltration in colorectal cancer was associated with a more favourable prognosis.63
Several independent lines of evidence implicate TAMs in promoting tumour progression and mediating therapeutic resistance. For example, it has been demonstrated that melanoma cells can recruit macrophages and induce cyclooxygenase-2 expression, which leads to increased angiogenesis and tumour growth.64 While they are probably not essential for tumour angiogenesis, TAMs certainly contribute to the speed of this process.65 Several of those secreted factors, growth factors such EGF, VEGF and cytokines such as interleukin (IL)-10 and IL-23, also contribute to tumour growth and metastasis.65 Therefore, it is not surprising that accumulating evidence suggests that, by targeting macrophage recruitment to cancerous sites through inhibition of colony-stimulating factor/colony-stimulating factor 1 receptor (CSF/CSF1-R), tumours seem to become less aggressive.66–68
Using a genetically engineered mouse model of breast cancer it could be observed that TAM infiltration of the tumour is paralleled by cytotoxic T cells.69 Interestingly, the functionality of the latter seemed to be compromised by the TAMs,69 arguing for their potential immunosuppressive role and, furthermore, linking them as a possible resistance mechanism towards cancer immunotherapy, such as application of programmed cell death protein 1 (PD1) inhibitors.70 While in glioblastoma, patients who received a VEGF-inhibitor upon relapse from standard therapy, the recurrent tumours contained a higher TAM infiltration than in those patients treated only with standard therapy.71 Based on these findings the hypothesis has been put forward that the modulation of VEGF signalling, such as the change in metabolism, might contribute to TAM infiltration.71 However, the precise role of TAMs remains unclear here, as modulation of the metabolism has yielded some conflicting results. Carmona-Fontaine and co-workers demonstrated that macrophages co-cultured with a breast adenocarcinoma cell line under normal and hypoxic conditions are not able to survive lactate accumulation such as in an hypoxic microenvironment,72 arguing against an increase in TAM infiltration due to an acidification of the microenvironment. By contrast, Wen and colleagues suggested that TAM infiltration is increased in an ovarian carcinoma xenograft, likely due to chemo-attraction of monocytes mediated by prostaglandin metabolites, produced under hypoxic conditions.73 In addition, it has even been shown that lactate abrogated the pro-inflammatory phenotype of macrophages.74
Taken together, it seems unclear under which circumstances TAM infiltration contributes to or inhibits tumour progression, or when it is a mere consequence of it. Until these questions are further elucidated, their full potential as therapeutic targets cannot be accurately gauged.
Conclusions
Alternative approaches to surgery, radio- and chemotherapy, all three of which aim to induce maximal reduction of tumour burden in the shortest time, have been shown to be experimentally viable. We have highlighted three areas of interest that lend themselves to future therapeutic developments. 1.
- The complex subclonal composition within mutant tumour cells that might suggest that the mutational landscape of tumours can be too rugged to allow targeted therapy to succeed. Here, future research is needed to understand when chronifying a malignancy can be a more promising therapeutic approach than healing. Importantly, these considerations are full of ethical implications and therefore should not be taken lightly. 2.
- Leading on from discussing the high genetic instability of mutant tumour cells, we consider an alternative therapeutic target that displays higher stability: the microenvironmental interactions of mutant tumour cells with cellular and extracellular components of the tumour. Here some promising targets have already been identified and are currently being clinically evaluated. 3.
- A better understanding of the individual constituents of the microenvironment is imperative in order to improve therapeutic options. Using TAMs as an example of potential future targets, we demonstrate how complex and contradictory the role of individual aspects of the microenvironmental can be.
Taken together, we are confident that further research into the above summarised aspects of tumour biology will open up new avenues in cancer therapy.