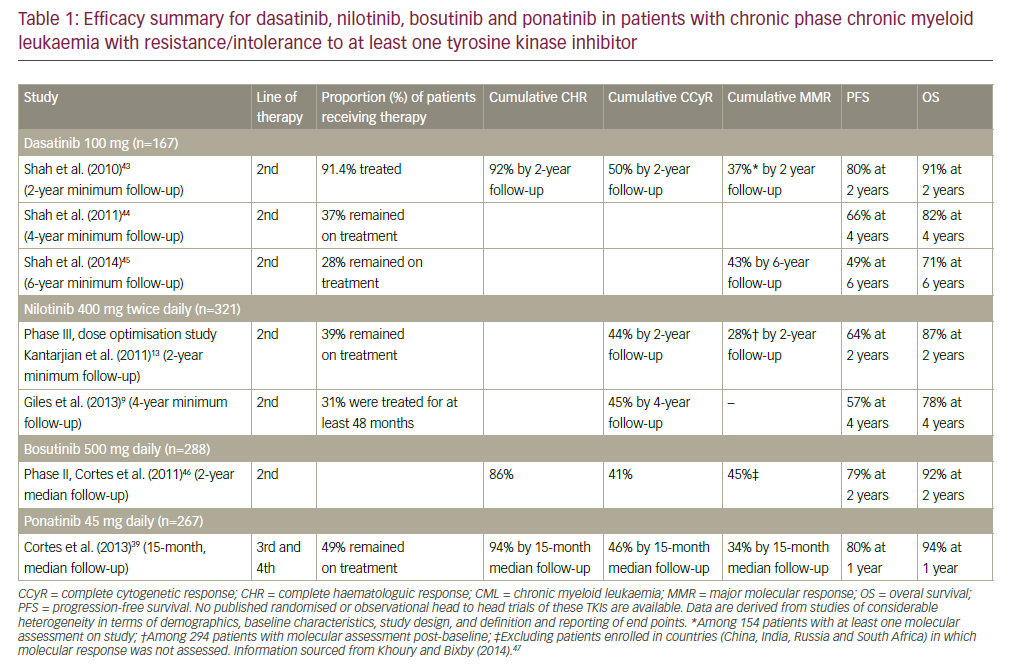
This latter observation is largely due to the ability to successfully re-treat relapsed patients. However, patients who become refractory to alkylator- and fludarabine-based treatments have traditionally had a poor response (<20%) to salvage therapy and a greatly shortened survival (median 10 months).1 Over the past decade this bleak situation for patients has been improved by the introduction of novel agents, including monoclonal antibodies. Alemtuzumab is a fully humanised monoclonal antibody directed against the CD52 antigen, which is widely expressed on B and T lymphocytes. It is licensed for the treatment of fludarabine refractory CLL and has been shown to induce remissions in 33–53% of patients in this setting.2,3 The standard dosing schedule is 30mg given three times a week intravenously for 12 weeks.
* CAM307 phase III prospective randomised trial comparing chlorambucil and alemtuzumab.CR = complete response; IV = intravenous; OR = overall response; SC = subcutaneous.
Alemtuzumab Monotherapy
The first report of the use of alemtuzumab as front-line therapy was in 1996 by Osterborg et al.4 Nine patients received the standard treatment, although in four patients the antibody was administered subcutaneously, and therapy was continued in all patients up to 18 weeks. The OR rate was 89% with three patients achieving CR. This group expanded the patient cohort and reported a further 41 patients treated with subcutaneous alemtuzumab as first-line therapy for a total of 18 weeks.5 The OR rate was maintained at 81% in 38 evaluable patients. Nineteen per cent of patients achieved CR and 68% a partial remission (PR). At the time of publication in 2002 the time to treatment failure had not been reached at 18+ months. These results are comparable to those observed for single-agent fludarabine6 and superior to those for single-agent rituximab7 (see Table 1). Interestingly, complete responders required 18 weeks of therapy to achieve their best response, with significant improvement in bone marrow clearance between the 12- and 18-week evaluation points. Furthermore, patients with low-volume lymphadenopathy also achieved complete remissions in contrast to the observation in relapsed refractory patients that lymphadenopathy predicted a poor response to single-agent antibody treatment. Ten per cent of patients developed cytomegalovirus (CMV) reactivation that responded rapidly to treatment with intravenous ganciclovir. There was no increase in bacterial sepsis. Although transient injection site reactions were observed with the subcutaneous administration in the majority of patients, many of the initial reactions associated with intravenous administration such as rigours, nausea and hypertension were not seen. One in five patients had a transient grade 4 neutropaenia, but other side effects were rare.
CR = complete response; F = fludarabine; IV = intravenous; OR = overall response;PFS = progression-free survival; SC = subcutaneous; TIW = three times a week.
At the American Society of Hematology (ASH) meeting in December 2006 the results of an international prospective, randomised, controlled trial (CAM307) comparing chlorambucil with intravenous alemtuzumab as front-line therapy for CLL were reported.8 Two hundred and ninety-seven patients were randomised to receive either alemtuzumab at the standard dose of 30mg three times per week for up to 12 weeks or chlorambucil 40mg/m2 once every 28 days up to 12 cycles. Response rates assessed by an independent panel showed OR of 83% for alemtuzumab compared with 55% for chlorambucil with CR rates of 24% and 2%, respectively (see Table 1). This translated into improved PFS for the patients who received alemtuzumab, with a 43% lower risk of progression or death. Notably, in patients who had the cytogenetic deletion of 17p (p53), there was a three-fold increase in OR with alemtuzumab (64%) compared with chlorambucil (26%). Statistically significant superior responses were also seen for patients with deletion 13q and deletion 11q treated with alemtuzumab compared with chlorambucil. Infections, including CMV, were reported in 76% of patients receiving alemtuzumab compared with 50% of chlorambucil patients while on study. Grade 3 and 4 lymphopaenia and neutropaenia were more common with alemtuzumab, but anaemia and thrombocytopenia were similar in the two treatment groups. Episodes of bacterial sepsis and febrile neutropaenia were comparable and the increase in infection in the alemtuzumab arm was almost entirely attributable to CMV reactivation. Although CMV reactivation occurred in half the patients, it was only symptomatic in 16%. This toxicity was therefore manageable by screening and pre-emptive treatment. Grade 3 or 4 infusion-related events were seen in 13% of patients receiving alemtuzumab and these were largely confined to the first few weeks of therapy. In contrast, adverse events increased over time in the chlorambucil arm where the median duration of treatment was twice as long. The toxicity profile of alemtuzumab in previously untreated patients appears to be much more acceptable with no increased treatment-related mortality compared with chlorambucil in the CAM307 randomised study. The Lundin study showed that efficacy for 18 weeks of subcutaneous alemtuzumab was equivalent to 12 weeks of intravenous therapy. Since subcutaneous administration results in fewer infusion-related side effects, this may be the preferable route.
Alemtuzumab Consolidation Therapy
Studies of alemtuzumab treatment of CLL, particularly in the relapsed/refractory setting, have consistently shown that patients with bulky nodal disease are less likely to respond to treatment compared with those patients who have low volume or no lymph node enlargement. This is in contrast to the excellent clearance of disease from the blood and bone marrow. In addition, in the Moreton study3 patients who achieved complete remissions and who were also negative for minimal residual disease (MRD) using a sensitive four-colour flow cytometry9 had significantly prolonged PFS and overall survival (OS). Several studies, using a variety of treatment regimens, have confirmed the observation that those patients achieving MRD-negativity have prolonged remissions compared with those patients who are MRD-positive at the end of therapy.10–12 These observations have provided a rationale for using alemtuzumab as consolidation of remissions achieved using fludarabinebased induction regimens. Four groups have published their results for this strategy using various treatment schedules, doses and routes of administration (see Table 2).13–16 Although not comparable, all have demonstrated the efficacy of such an approach in improving responses in around 50% of patients receiving consolidation. There has only been one randomised study which was reported by the German study group in 200413 and updated at ASH in 2006.17 Patients who had been treated to maximum response with fludarabine or fludarabine plus cyclophosphamide were randomised to receive alemtuzumab at the standard dose of 30mg three times per week for 12 weeks given intravenously at a median of two months following induction treatment or to have no further treatment. The trial was stopped prematurely having recruited 21 patients (11 to alemtuzumab and 10 to observation) because of a high infection rate in the alemtuzumab arm. However, despite the small numbers of patients, this trial did show a significant improvement in PFS for patients receiving alemtuzumab consolidation (median not reached) compared with 27.7 months for the observation arm. Factors contributing to the toxicity of alemtuzumab in this setting may be the dose and duration of therapy and, most importantly, the short interval between completing induction treatment and introduction of consolidation.
Consolidation after first induction treatment of CLL is therefore feasible and can deepen remission and increase PFS in a proportion of patients. However, there is a risk of toxicity and there are also financial implications. It may therefore be prudent to reserve this strategy for those patients with higher risk disease. Furthermore, a larger randomised study needs to be completed in such a group to confirm safety and efficacy as well as establishing the optimal timing, dose and delivery (intravenous or subcutaneous). Alemtuzumab for High-risk Chronic Lymphocytic Leukaemia
It is well recognised that patients who have deletions of chromosome 17p resulting in dysfunction of the p53 pathway have resistance to conventional chemotherapy such as alkylating agents or purine analogues and also have significantly shortened survival compared with those patients without this abnormality.18 Although p53 deletion in CLL is infrequent at initial presentation – occurring in 5–10% of patients – this abnormality becomes increasingly common as the CLL advances and becomes chemo-resistant. There has now been a number of reports showing that alemtuzumab has efficacy in this subgroup, probably by killing cells through a p53-independent mechanism.19–21 The only other therapy shown to be effective in this way is high-dose steroids.22 In CLL patients with p53 deletion who do not have bulky nodal disease, singleagent alemtuzumab may be an appropriate treatment choice either at first or subsequent line of therapy. However, in those patients with enlarged nodes it would seem logical to combine alemtuzumab with high-dose steroids, and this regimen does appear to be effective in a small number of patients who have been treated.23 It is now critical that patients who are about to embark on treatment, whether this is first- or subsequent-line, should have analysis to detect the presence of the p53 deletion since this will affect selection of treatment. The knowledge of specific genetic abnormalities will allow the prospective selection of appropriate treatments in order to reduce the damage caused by ineffective chemotherapy and to maximise the opportunity for achieving a good remission.
Alemtuzumab Combination Regimens
To date, experience with alemtuzumab in combination regimens is somewhat limited, and has been explored only in the relapsed patient setting. Single-centre data of combinations with fludarabine (FluCam – fludarabine and alemtuzumab24; C-FAR – cyclophosphamide, fludarabine, alemtuzumab and rituximab25) have all shown improved efficacy compared with alemtuzumab as a single agent in an equivalent clinical setting. There are currently a number of trials examining fludarabine/alemtuzumab-based combinations in the first-line setting and the results of these are awaited. Studies of high-dose steroids and alemtuzumab in high-risk CLL are ongoing.
Conclusion
There have been major advances in the management of CLL over the past decade. This includes the ability to stratify patients according to biological risk parameters such as genetic abnormalities, the introduction of novel therapies such as monoclonal antibodies and the ability to achieve a high proportion of good remissions, including eradication of minimal residual disease. All these strategies have resulted in improved selection of patients for appropriate therapy and better PFS. Alemtuzumab has been shown to be an effective monotherapy in both first-line and refractory CLL and it is now clear from several studies that toxicity is much reduced and more manageable when alemtuzumab is used earlier in therapy. More data are now needed on the use of alemtuzumab in front-line combination regimens. The most compelling data for alemtuzumab efficacy are in the high-risk cytogenetic group exhibiting p53 deletion when used either as a single agent or in combination with high-dose steroids. In addition, this antibody therapy has been shown to be effective in eradicating residual disease after completion of induction therapy, although the optimal regimen (dose, schedule and route of administration) is still to be determined.