The exact cause of most leukemias is still not known, although there are a number of predisposing factors and exposures. In all likelihood, the cause or causes of most leukemias is multifactorial, including genetic, immune, infectious, and environmental factors.
Clinical Manifestation
The presenting clinical features of the acute childhood leukemias reflect the uncontrolled proliferation of malignant cells leading to replacement and suppression of normal hematopoietic progenitor cells, and infiltration into extramedullary spaces. Symptoms frequently accumulate in a matter of days or weeks, culminating in some event that brings the child to medical attention. Chronic Myelogenous Leukemia (CML) may present with a more insidious course. The differential diagnosis of an acute leukemia at presentation is included in Table 3.
Classification
ALL is the most common form of leukemia in childhood (see Figure 1). Subclassification into early pre-B-cell, pre-B-cell, B-cell, and T-cell lymphoblastic leukemias is essential to treatment, and prognostic stratification of patients.The myeloid leukemias can be categorized according to either the cell of origin (French-American-British, FAB, system) or cytogenetic and morphologic features (WHO (World Health Organization) Classification system). The distinguishing phenotypic features of each class of leukemia is listed in Table 4.
Cytogenetic and Molecular Markers
There are several well-described genetic mutations that not only offer insight into the pathogenesis of an acute leukemia, but which significantly influence prognosis. Treatment of ALL
Risk-Stratified Therapy
Risk-adapted therapies, taking into account clinical features (age, white blood cell count, and response to induction therapies) and biologic features (ploidy and mutations of known prognostic significance), are used to maximize cure rates while minimizing long-term side effects. Standard risk and high-risk patients are initially distinguished based on age (between 1 and 9.99 years of age is standard risk) and the initial white blood cell count (=50,000/µL is standard risk). Other clinical features used to stratify therapy include gender, leukemia phenotype, cytogenetics, and response to initial therapy.
Principles of Treatment
Standard therapy is divided into phases of remission induction, consolidation, delayed intensification, extramedullary disease treatment or prophylaxis, and maintenance. The timing, number, and chemotherapy components of each phase may differ between protocols, but the principles of therapy are similar. Remission induction in achieved in most cases with three drugs (vincristine, asparaginase, and a corticosteroid) in standard risk patients, or four drugs in high risk patients (the same three as in standard risk patients with the addition of daunorubicin).The goal is to induce a complete remission, defined as the reduction the leukemia cell burden below clinical, morphological, and molecular levels of detection after recovery of normal blood counts. With current chemotherapeutic regimens, remission induction is possible in more than 95% of children with ALL. Many protocols follow the induction phase, with a consolidation phase heavy in antimetabolites to secure or consolidate the remission. Stratification to higher or lower intensity therapies may occur at the end of induction based on molecular quantification of minimal residual disease.
Delayed intensification is a second phase of intensified chemotherapy similar to the induction and consolidations phases.The addition of a delayed intensification phase by the Berlin-Frankfurt-Munich (BFM) group has led to significant improvement in survival in all childhood ALL patients. Survival among patients with high-risk leukemia may be further improved by augmenting the regimen with a second delayed intensification.
In the past two decades, several trials have demonstrated that central nervous system (CNS) radiation may be reduced (to 1200cGy to 1800cGy) or eliminated in patients with low-risk B-cell ALL with a rapid early response to induction therapy. Elimination of cranial radiation in patients with T-cell ALL has not proven feasible. Reduction in cranial radiation is designed to reduce long-term endocrine and cognitive effects, as well as reduce the risk of secondary malignancies
Unlike treatment paradigms for most malignancies, successful therapy for childhood ALL includes a protracted maintenance period. Attempts to reduce therapy to less than 24 months have never been successful. In current Children’s Oncology Group (COG) Studies, boys with standard risk ALL receive three years of postremission therapy, while girls with ALL receive two years of post-remission therapy. The intensity of therapy is greatly reduced during maintenance, and many patients may return to school during this period.
Adapted from Aesselin BL. Leukemias. In Hoekelman RA, Adam HM, Nelson NM, et. al. Primary Pediatric Care, 4th Ed. Mosby, (2001). ALL = acute lymphoblastic leukemias, AML = acute myelogenous leukemia.
Infant ALL
Despite intensified therapy, infants less than six months of age with ALL have an event-free survival of less than 10%, while those older than six months have an eventfree survival of 40%. Currently, all infants still have a higher incidence of relapse, death from toxicity, and long-term side effects when compared to older children.
Infants under six months old, with abnormalities involving 11q23, appear to be at highest risk.
Treatment of AML
Principles of Therapy
The event-free survival rate in childhood AML has reached a plateau at 60%, despite aggressive intensification of therapy. Cytarabine and daunorubicin, in combination with either etoposide or thioguanine, will induce a complete remission in more than 90% of patients. Attempts to substitute idarubicin or mitoxantrone for daunorubicin, or increase the dose of cytarabine, have largely proven to be of comparable efficacy, and, occasionally, greater toxicity. Intensive post-remission therapies have been developed using multi-agent regimens cycled every four to six weeks. There have been no randomized, head-to-head comparisons of the different regimens, but none have succeeded in improving long-term, event-free survival beyond 60%. Most studies of allogeneic transplant in children with AML non-randomly assign patients with matched sibling donors to undergo transplant, and those without sibling donors to continue with conventional chemotherapy. Relapse-free survival is generally better in the patients who undergo transplantation, but overall survival is comparable due to transplant-related mortality.
cy = cytoplasmic, mem = membrane,TCR = T cell receptor, MPO = myeloperoxidase,TdT = terminal deoxynucleotidyl transferase
Acute Promyelocytic Leukemia
Patients with acute promyelocytic leukemia (APML) do not do well with conventional AML therapy. Rather, regimens intensifying the anthracycline dose in induction and incorporating retinoic acid, mercaptopurine, and methotrexate into maintenance therapy have led to five-year survival rates exceeding 80% among children with APML.
CML
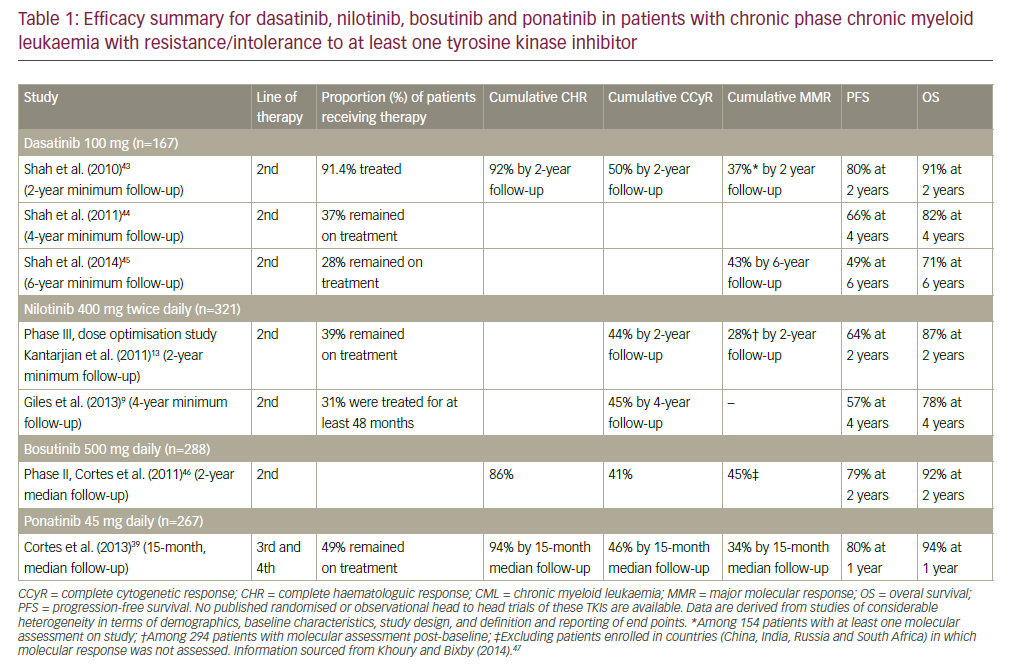
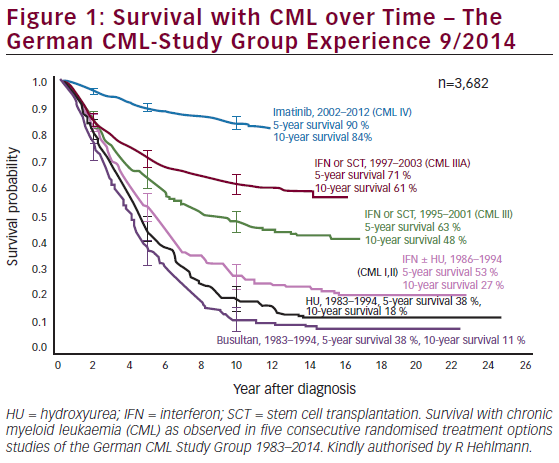
In children and adults, CML is characterized by the presence of the Philadelphia (Ph) chromosome. Until recently, standard therapy for CML in children and adults included hydroxyurea, interferon, and low-dose cytarabine. While clinical improvements in hematologic parameters were possible with these therapies, cytogenetic remission were infrequent. More recently, imatinib mesylate was developed as a specific inhibitor of the BCR-ABL (breakpoint cluster region- Abelson) tyrosine kinase. Results from recent clinical trials have rapidly established imatinib as the standardof- care for CML, and cytogenetic remissions are now expected. Limited data are available on imatinib mesylate in children, but preliminary data are promising. Imatinib mesylate has dramatically changed therapy for CML, but it is still not a cure. The only proven curative strategy for children with CML is allogeneic stem cell transplantation (SCT). Survival after SCT for CML is reported to be 70–80% with a matched sibling donor and 40–60% when unrelated donors are used.
Relapsed Leukemias and STC
ALL
Despite development of successful therapies in childhood leukemias, 20–25% of children with ALL and 50% of children with AML will relapse after chemotherapy treatment. In fact, more children are diagnosed with relapsed leukemia than Hodgkin’s disease (HD) and pediatric sarcomas. In most children with relapsed leukemia, long-term, event-free survival is less than 50%, even with SCT. Remission re-induction following relapse is relatively good, but overall survival remains poor, owing to a deficiency in effective therapies.The three-year, event-free survival for patients with a first remission less than 18 month is 4%; 18–30 months, 10%; and greater than 30 months, 41%. Most events occur within six months of relapse and fewer than half of the patients assigned to unrelated donor transplants actually made it to transplant in second remission.These results are striking and speak strongly to the need for additional therapies for ALL.
AML
As is the case with ALL, hematologic relapse remains the most common adverse event in patients with AML. About 30–40% of patients who achieve a remission will relapse, while 25% will have residual disease after intensive induction therapy. Remission re-induction rates for all patients with AML in first relapse ranges from 50–80% using multiagent chemotherapy. Overall survival following relapse is a dismal 30–40%.
New Agents
As high-dose chemotherapy with SCT is not enough for high-risk, relapsed, and refractory leukemias, new agents are clearly needed. One of the driving forces in the identification, rational synthesis, and development of new agents—many of which are the prototypes in entirely new classes of antineoplastics—is an improved understanding of the biological mechanisms involved in leukemogenesis.
Next Generation Cytotoxic Therapies
One strategy employed in new agent development is the encapsulation of proven agents in liposomes containing polyethylene glycol (PEG). Pegylated forms of doxorubicin, vincristine, all-trans retinoic acid (ATRA), and asparaginase are available. Pegylation increases the half-life, enhances tumor delivery of the compounds, decreases the vesicant effect of doxorubicin and vincristine, decreases the cardiac toxicity of doxorubicin, and reduces the number of intramuscular injections of asparaginase .
Cytarabine is a purine nucleoside analog with an excellent track record in pediatric leukemias, both AML and ALL. Fludarabine, cladribine, and clofarabine are nucleoside analogs developed more recently that are also effective in leukemias and have more favorable pharmacologic features than cytarabine. Based on a response rate of 31% (12% complete remission), clofarabine was recently approved for use in relapsed ALL in children. This the first drug approved for such an indication in the US for more than two decades. Like cladribine and fludarabine, clofarabine may be used effectively in combination with cytarabine. As a potent inhibitor of ribonucleotide reductase, clofarabine may decrease intracellular deoxynucleotides, decreasing feedback inhibition of deoxycytidine (dCyd) kinase, and thus allowing for an increase in the accumulation of ara-CTP.
Nelarabine (compound 506U78) is a pro-drug of cytarabine. Early studies demonstrated that nelarabine is selectively toxic to lymphoblasts with an early T-cell phenotype. Initial Phase I and II trials conducted by the COG demonstrated activity with an overall response rate of 55% in first relapse, 25% in second relapse, and a few responses in third relapse. Unfortunately, there was significant neurotoxicity, including ataxia, confusion, and coma. The effect appears dose-dependent, and the incidence and severity decreased with dose deescalation. Nelarabine is currently being tested by the COG in the backbone of an intensive BFM regimen for patients with newly diagnosed T-cell ALL. Agents Targeting Specific Molecular Events
The remarkable success of imatinib mesylate in patients with CML demonstrated the potential of targeted therapies.
Tyrosine Kinase—results from recent clinical trials have rapidly established imatinib as the standard-of-care for CML. A recent phase III prospective trial of >1,000 patients conclusively demonstrated that imatinib is superior to interferon alpha (IFN-a) plus low-dose cytarabine in inducing cytogenetic disease remission and prolonging progression-free survival in adult patients with chronic-phase CML.
Dasatinib—(BMS-354825) is a potent inhibitor of Abl and Src (sarcoma) kinase activity. Imatinib resistance is frequently mediated by mutations in the Abl kinase domain. Unlike imatinib, dasatinib has affinity for all mutations of the Abl kinase domain. In a phase I trial of dasatinib in 36 imatinib-resistant (or intolerant) patients with CML in chronic phase, a complete hematologic response is reported in 86% of the patients, and a complete cytogenetic response by three months is reported in 28% of patients.These compounds will have affinity for other tyrosine kinases (e.g. Src, c-kit, plateletderived growth factor receptor (PDGFR)) involved in the pathogenesis of all leukemias. However, it is not yet known what the clinical applications of these compounds will be when targeting non-Abl tyrosine kinases.
Thalidomide and Lenalidomide—thalidomide belongs to the class of compounds called imids.While the mechanism of action is largely unknown, imids do appear to inhibit cytokine production and angiogenesis, while enhancing stromal cell function. Thalidomide experienced a revival after demonstrating a 66% response rate in patients with multiple myeloma. Lenalidomide, is a synthetic compound with less neurotoxicity and teratogenicity.An 83% response rate is reported among patients with myelodysplastic syndrome (MDS) due to 5(q-).
Proteosome Inhibitors—proteosome inhibitors, like bortezomib, inhibit the breakdown of proteins through the ubiquitin-proteasome pathway. Inhibitors of cell cycle progression and the transcription factor NF-_B accumulate in cells treated with a bortezomib. In an open-label trial of patients with multiple myeloma, a 28% response rate is reported. In a subsequent phase III randomized trial comparing bortezomib to high-dose dexamethasone, a superior response rate is seen in the patients treated with bortezomib. A phase I trial has been completed by the COG, and phase II combinations are underway.
Arsenic Trioxide—as single agent therapy, arsenic trioxide may induce a complete remission in 90% of patients with APML. Success of this agent in APML has prompted multiple on-going studies in MDS, myeloma and AML.
Inhibitors of mTOR—the mammalian target of rapamycin (mTOR) is a serine-threonine kinase involved in the activation of Akt and downstream pathways. Rapamycin, an mTOR inhibitor, has demonstrated significant clinical responses in a few patients with relapsed and refractory AML.
Farnesyltransferase inhibitors—ras mutations are common in AML and MDS, and targeting Ras farnesylation is a novel approach currently under investigation.Tipifarnib (R115777) is a farnesyltransferase inhibitor with initial clinical responses in 10 out of 34 elderly patients with AML. Subsequent trials in patients with MDS, CML, and high risk AML were less encouraging. However, additional studies evaluating combinations with other agents are on-going. Targeting Gene Expression
Histone acetylation/deacetylation and deoxyribonucleid acid (DNA) methylation are epigenetic phenomena that can effect gene expression. Chromatin remodeling agents targeting DNA methylation and histone acetylation are at present in clinical trials. Currently, there are two hypomethylating agents with activity in hematologic malignancies. 5-azacytidine is US Food and Drug Administratioin (FDA)-approved for adults with MDS and 5-aza-2’-dexoycytidine (decitibine) is currently in clinical trials in adults with MDS. Suberoylanilide hydroxamic acid (SAHA), high-dose valproate, and sodium butyrate are histone deacetylase inhibitors. Early investigations into these agents alone and in combination with hypomethylating agents are under way.
Monoclonal Antibodies
Rituximab is a chimeric monoclonal antibody that recognizes the CD20 antigen expressed on mature and immature B-cells. CD20 is expressed in some B-cell ALLs and B-cell non-Hodgkin’s lymphoma. Initial studies demonstrated that the addition of rituximab to the standard CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) backbone, enhanced survival among patients with B-cell lymphomas.Alemtuzumab is a monoclonal antibody that recognizes CD52 expressed on T-cells, some B-cells and NK cells. Epratuzumab is a humanized monoclonal antibody directed against CD22, an antigen expressed on mature and immature B-cells. In a phase I/II trial of epratuzumab in adult patients with lymphoma, there was a 33% response rate.
Gemtuzumab ozogamicin (GO) is a humanized monoclonal antibody, directed against the myeloid antigen CD33, conjugated with the cytotoxic agent calicheamicin. It is approved for use in adults with AML over age 60 years of age. In a phase I trial in pediatric patients GO demonstrated a 28% response rate. However, six of the 13 patients who later underwent SCT developed veno-occlusive disease (VOD). Following promising phase I results, the COG is currently evaluating the role of GO in the backbone of a daunorubicin, etoposide, and cytarabine induction. Humanized monoclonal antibodies conjugated to radioisotopes have also been developed.
Conclusions
Treatment of childhood leukemias is widely touted as a great success in oncology clinical trials, largely due to the continued incremental increases in long-term survival in children with ALL. Significant challenges still lie ahead as we define new strategies for treating AML and relapsed leukemias, as well as the myeloproliferative and myelodysplastic disorders. Application of riskadapted therapy to patients with ALL and AML has allowed for continued improvement in long-term, event-free survival and a reduction in long-term side effects. What has been learned about leukemia biology, host pharmacogenetics, and environmental influences on leukemia development, has led to the development of effective strategies treatment and preventive strategies for childhood leukemias. There is still a significant amount of work to be done, but the future is bright.