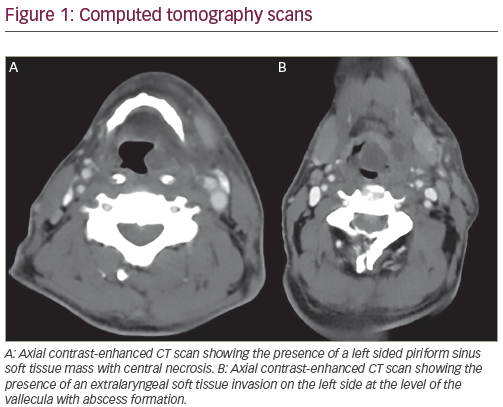
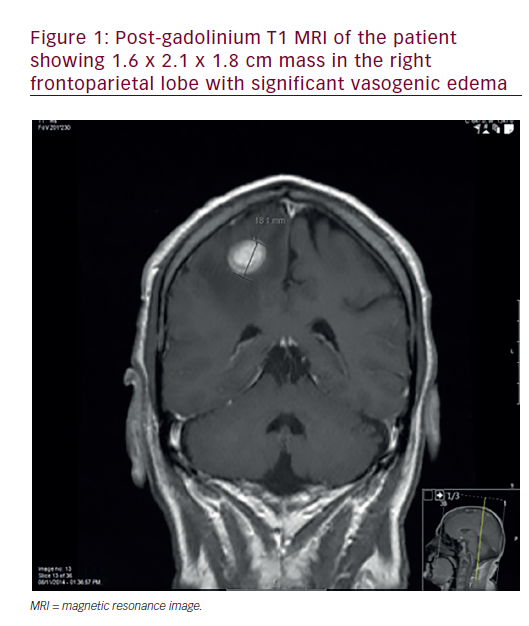
Patients with brain metastases do poorly, with median survival of only one month if treated with steroids only.5 In an effort to better stratify patients into prognostic groups, the Radiation Therapy Oncology Group (RTOG) conducted a recursive partitioning analysis and found age, Karnofsky Performance Status (KPS), and systemic disease status to be important.6 Even in the best group, recursive partitioning analysis (RPA) class I, median survival is only 7.1 months (see Table 1).
Corticosteroids
Steroids are often used to ameliorate symptoms when patients are diagnosed with brain metastasis. Corticosteroids control peri-tumoral edema, usually providing symptomatic relief within hours, but long-term efficacy is poor.7,8 Prolonged steroid use results in several negative consequences, described in detail elsewhere. Steroid administration, with or without whole brain radiation therapy (WBRT), has been compared in one trial. WBRT plus prednisone (compared with prednisone alone) did not improve performance status (61 versus 63%) but improved median survival (14 versus 10 weeks).9 Patients with a short life expectancy may be appropriately palliated with steroid therapy alone, while those with a longer projected lifespan are more likely to benefit from WBRT.10Whole Brain Radiation Therapy
The efficacy of WBRT in the treatment of brain metastases was first demonstrated over half a century ago, with improvement in symptoms and functional status and minimal morbidity.11 Multiple non-randomized studies have shown that WBRT increases survival by up to four to six months, compared with only one or two months with conservative therapy.5,12 Multiple fractionation regimens have been studied and are summarized in Table 2; little change in median survival has been realized. The 30Gy in 10 fractions dose schedule used in these trials has become a standard. As WBRT is usually palliative, treatment schedules should generally be selected to maximize quality of life, and regimens that provide a slight survival advantage at the expense of this end-point should not be routinely employed.
Surgery
Since the 1980s, surgery has been a standard option for patients with a single brain metastasis.13 Patchell and colleagues randomized 48 patients to biopsy plus WBRT versus resection followed by WBRT and found that resection significantly improved overall survival (40 versus 15 weeks, p<0.01) and local recurrence (20 versus 52%, p<0.02).14 Similar results were obtained in another trial randomizing patients to 40Gy in 2Gy twice-daily fractions with or without resection. Combined modality treatment improved median survival (10 versus six months, p=0.04) and functionally independent survival (FIS) (p=0.06); maximum gain was observed in patients with stable extracranial disease (median survival 12 versus seven months, p=0.02; FIS nine versus four months).15
Another randomized trial, however, did not support these findings. Mintz and colleagues randomized 84 patients to 30Gy in 10 fractions with or without resection and found that resection did not significantly improve median (5.6 versus 6.3 months, p=0.24) or one-year survival (12.2 versus 30.2%).16 Most patients in this trial had active extracranial disease and poor performance status and almost half of the patients randomized to the radiation-alone arm eventually underwent surgery because of patient choice or symptomatic deterioration.
Retrospective data indicate that the intracranial failure rate for patients undergoing resection alone is unacceptably high.17 Patchell et al. demonstrated significant reduction in intracranial failure with WBRT added to resection (18 versus 70%, p<0.001).18
The value of resection in patients with multiple or recurrent metastases is less clear. A retrospective analysis found that patients with resection of four metastases showed similar survival to those with a single metastasis (14 versus 14 months, p<0.5), while those with one or more lesions left unresected did not perform as well.19 Therefore, aggressive surgical treatment is generally reserved for patients with good functional status and controlled extracranial disease. Sterotactic Radiosurgery
Stereotactic radiosurgery (SRS) allows for the delivery of a single, highly focused, high-dose fraction of radiation within the target volume with low doses to normal tissue. RTOG 90-05 established the dose guidelines to be utilized.20 RTOG 95-08 evaluated radiosurgery in a randomized context in patients with one to three brain metastases <4cm in diameter (WBRT with or without SRS boost).21 SRS plus WBRT resulted in better survival (6.5 versus 4.9 months, p=0.0393) and local control (82 versus 71% at one year, p=0.01), primarily in patients with single metastases. Improvement in performance status and reduction in steroid use was also noted in patients receiving SRS. Subgroup analysis showed benefit from SRS in RPA class I patients (p=0.0453) and those with non-small-cell lung cancer (NSCLC) primaries (p=0.0508).
As a result of the available evidence, RPA class I patients with single brain metastasis are currently treated with resection or SRS followed by WBRT. Patients with multiple intracranial lesions may be treated with WBRT alone, or, depending on performance status and extracranial disease, may receive a combination of SRS, WBRT, and resection.
Controversies
Can Whole Brain Radiotherapy Be Withheld?
The rationale for adding WBRT to other modalities is to treat ‘micrometastases’ not visible on imaging. Patchell et al. showed that the addition of WBRT to resection improves local and regional control and reduces neurological death. Aoyoma and colleagues randomized 132 patients with one to four brain metastases <3cm in diameter to WBRT plus SRS or SRS alone, and found that WBRT plus SRS significantly reduced brain tumor recurrence (46.8 versus 76.4%, p<0.001).22 It has been argued that intracranial failure and neurological deterioration is more directly related to progression in the brain, and therefore maximizing local control (with WBRT, for example) is paramount.23 This concept has been further supported by a recent randomized trial evaluating motexafin gadolinium as a radiosensitizer.24 The most significant predictor for neurological and neurocognitive decline as well as deterioration in quality of life was disease progression in the brain. The switch to surgery or radiosurgery alone advocated by some is occurring in the absence of randomized data. This strategy reflects concerns about possible neurological and neurocognitive decline secondary to WBRT; much of the data for this comes from unconventional fractionation schedules. WBRT is considered to be the standard of care for the management of patients with brain metastasis (either adjuvantly or alone) and further studies are needed before it is routinely omitted; at the least, patients should be appropriately informed of the high rate of intracranial failure when WBRT is withheld.Surgery versus Stereotactic Radiosurgery
Both resection and SRS offer good results for selected patients with brain metastases. Although no randomized controlled trials have compared these modalities, several non-randomized comparisons have been reported. The MD Anderson Cancer Center reported a small retrospective experience comparing SRS and surgery with better survival for surgery (16.4 versus 7.5 months), but inherent selection bias, rather than true efficacy, was probably the reason for this.25 Most other retrospective studies have found nearly equivalent rates of local control and survival (see Table 3).26 One of these reports reviewed the outcomes for 122 patients who underwent SRS plus WBRT for a single brain metastasis; patients were eligible for resection by criteria used in the 1990 Patchell et al. trial.14 SRS plus WBRT produced outcomes similar to those of resected patients.14,27 Median survival (56 versus 40 and 43 weeks), duration of functional independence (44 versus 38 and 33 weeks), neurological death (25 versus 29 and 35%) and in-field recurrence (14 versus 20% and unreported) were all comparable.14,17,27
As surgery provides immediate symptomatic relief, this modality is clearly preferable in patients with severe neurological deficits, significant mass effect, or ventricular obstruction.26 Surgery is also generally preferred for lesions >4cm in diameter, as some data suggest SRS for larger lesions results in higher complication rates and decreased local control. For most other patients, especially those with deep-seated tumors, SRS is often the favored option due to its minimal invasiveness, favorable toxicity profile, and reduced hospitalization time and costs. For patients in whom one metastasis is symptomatic, this lesion can be resected and the others treated with SRS.13
Future Directions
Given the poor outcomes with standard therapies, novel approaches in the form of radiation sensitizers, cytotoxic chemotherapy, and target therapies have been and are currently being investigated. One such agent is the radiosensitizer motexafin gadolinium, a metalloporphyrin redox modulator that selectively localizes in tumors and catalyzes the oxidation of a number of intracellular reducing metabolites necessary for repair of cytotoxic damage.28 A recently completed randomized trial has shown that patients with brain metastases from NSCLC treated in a timely fashion had an improvement in neurological progression.29 Another radiosensitizer, RSR-13 (an allosteric modifier of hemoglobin), has shown promising results in patients with brain metastasis from breast cancer primaries.30
Cytotoxic therapies are also being actively investigated in the management of brain metastasis. Temozolomide, an oral alkylating agent, has shown activity in patients with brain metastasis and multiple trials are ongoing to determine its safety and efficacy.31 A variety of targeted agents, such as lapatanib, sorafenib, and Sutent, are currently under investigation for use in certain subsets of patients with brain metastasis.
Brain metastasis continues to be a significant medical problem affecting approximately 170,000 Americans annually. Whole brain radiation remains the standard of care either in the up-front or adjuvant setting. Patients with limited disease and good performance status may realize a survival advantage with either surgery or radiosurgery. Several new radiation sensitizers, cytotoxic agents, and molecularly targeted agents are currently being evaluated.