Programmed death 1 (PD-1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) are immune checkpoints that are expressed on the surface of antigen-presenting cells in the initiator and effector phase of T-cell activation respectively. They are responsible for “switching off” the T-cell. Inhibition of these checkpoints allows for overexpression of the immune system. The mechanism of defeating tumour cells can be understood by the three E’s of immunoediting.1 This over activity of the immune system may result in effects on normal body tissues, which is possibly how toxicities arise, although the exact mechanism is largely unknown.2
The incidence of grade 3 or 4 immune-related adverse events (irAEs) is higher with CTLA-4 blockers, whereas and PD-1 inhibitors appear to have better tolerability.3–5 The grade of irAEs varies according to the dose of drug administered to patients, where smaller doses of drug are used, side effects are similar but are less frequent.6 The combination of PD-1 inhibitor with a CTLA-4 inhibitor was recently approved in melanoma, however more adverse reactions were seen when the two drugs were used together especially grade 3 or 4 irAEs (55%),7 although greater overall response rates were seen.7–9 The incidence of irAEs can vary with tumour type and between different classes of drugs.
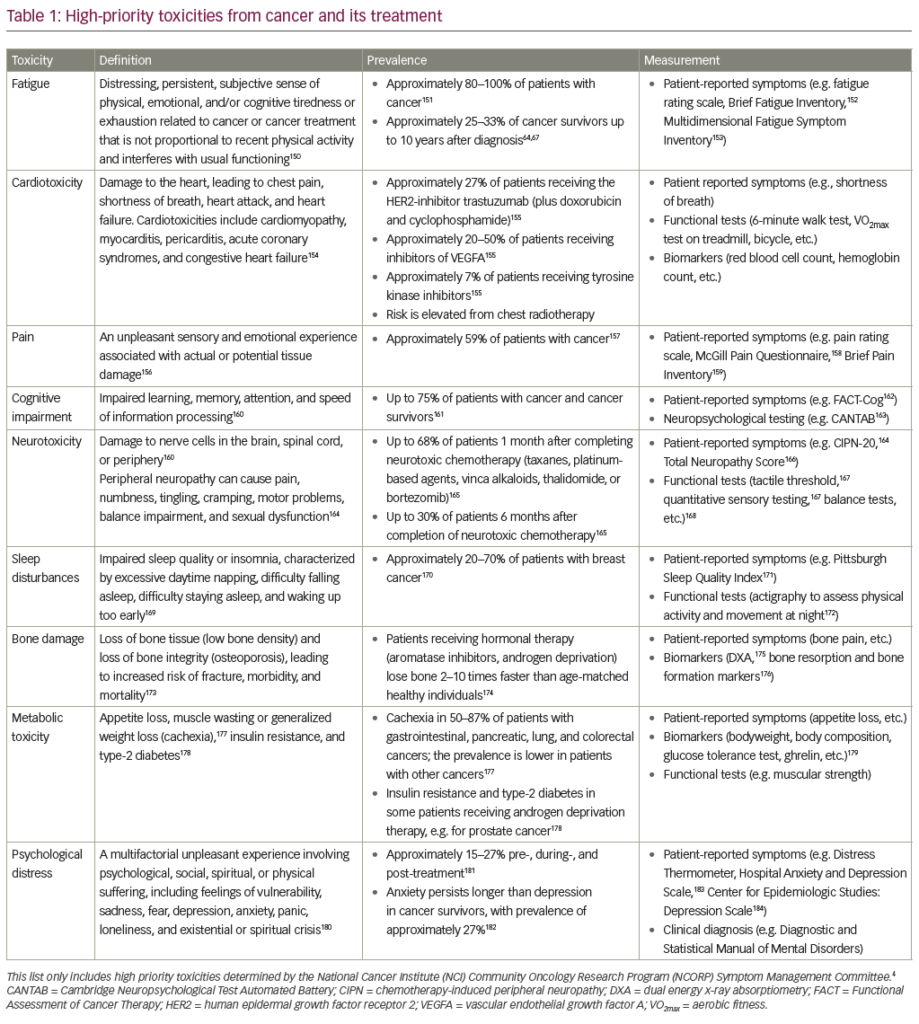
The most frequent irAEs are seen in the gastrointestinal (35%) and dermatological (44%) systems.6 The incidence of hepatic and endocrine system involvement follows with about 5–6%. Other systems less commonly affected are neurological, ophthalmologic, pulmonary, renal, haematological, cardiovascular, respiratory and musculoskeletal.,2,6,8
irAEs usually develop within 6–12 weeks of initial dosing and resolve within 12 weeks of onset, but may appear after the first dose.10,11It is also a postulation that the more severe the adverse event is, it correlates with a better response to treatment.3,9,12
When managed correctly, promptly and with close monitoring, most irAEs are reversible.2,6,12 In general, the optimal management of irAEs includes early recognition – by far being the most important, proper assessment of severity so that the choice of therapy, either supportive or immunosuppressive, can be quickly and correctly implemented. Mild irAEs can be observed and treated symptomatically with supportive care. As a guide, moderate irAEs, usually require stopping the offending agent implementing oral corticosteroid therapy and restarting therapy once symptoms have resolved. Severe irAEs warrant permanent discontinuation of the drug, hospitalisation and high-dose intravenous corticosteroids, with slow weaning. In very severe cases other immunosuppressive agents such as infliximab or mycophenolate mofetil may be warranted.13
Dermatological immune-related adverse events
These can occur after the initial dose of treatment and can be ongoing. Rashes are frequently maculopapular and mild.3,8,10,12 Rash and generalised pruritus can occur in up to 50% of patients with CTLA-4 inhibitors and 37% with PD-1 inhibitors.10 Symptomatic treatments for grade 1 rash; such as topical corticosteroid creams can be used. Oral antihistamines can be used if pruritus is severe.8 Rare cases of serious skin reactions such as Stevens-Johnson syndrome and toxic epidermal necrolysis have been reported.5,10,14 A moderate rash is usually non-localised and covers more than 50% of the skin surface area and the recommendation is to omit the offending agent. If there is no improvement within a week, it usually requires topical or oral corticosteroids at a dose of 1 mg/kg/day. The drug can be resumed immune related symptoms become mild. Dose reductions are not usually advocated. When high dose corticosteroid therapy is used, once symptoms are controlled, tapering of the steroids should occur over a one-month period at least.13
Of note, the development of vitiligo may be associated with clinical benefit. Although it occurs in a small percentage of patients undergoing immunotherapy, there is a clear survival benefit in patients who develop vitiligo during treatment.14,15
Gastrointestinal immune-related adverse events
Side effects can occur anywhere along the gastrointestinal tract, ranging from mucositis, aphthous ulcers, gastritis and abdominal pain. More commonly diarrhoea related to colitis can be observed and patients can also have blood or mucus in their stool. In severe conditions perforation can occur and lead to death and must be excluded in patients with symptoms of peritonitis.2,10 Other infectious causes of diarrhoea must be excluded for instance Clostridium difficile infection in severe cases.3,8,10
A colonoscopy can be considered in patients with severe symptoms or if the cause is unclear.8,10 Mild symptoms can be treated symptomatically with rehydration, replacing electrolyte losses and loperamide.2,3,13 Grade 2 irAEs require the immunotherapy agent to be omitted. If symptoms are ongoing for more than one week, there should be an immediate commencement of oral corticosteroid therapy at a dose of 1 mg/kg/ day. When symptoms are resolved, the drug can be recommenced.2,3,10,13
Severe or life-threatening colitis is a serious complication as well as symptoms consistent with perforation, ileus or fever. High dose intravenous corticosteroids commenced at a starting dose of 2 mg/ kg/day is recommended.10,13 If symptoms persist, a single dose of immunosuppressive infliximab therapy at 5 mg/kg must be considered unless there is a contraindication.10,13 The dose of infliximab can be repeated after two weeks if symptoms persist.8,10 Mycophenolate mofetil can also be considered.10,16
Hepatic immune-related adverse events
It is important to remember that liver toxicity can occur in the absence of clinical symptoms. A baseline liver functions test (LFT) should be taken.10,13 When derangements occur, other infectious causes, medicinal use by patients and disease progression must be excluded. These should be investigated appropriately.10,13 Severe hepatotoxicity requires permanent discontinuation of the immunotherapy drug, commencement of high dose intravenous corticosteroids and monitoring until LFT returns to baseline. If LFT derangement is significant or refractory to steroid treatment, alternative immunosuppressive therapy such as mycophenolate mofetil must be considered.13 Other hepatotoxic agents can be avoided. A liver biopsy is indicated where aetiology is unclear.10
Endocrine immune-related adverse events
Symptoms are nonspecific such as fatigue, headache, mental state changes or dizziness from hypotension.10,13 The most common abnormalities of the endocrine system are hypophysitis and hypothyroidism.8,10 Workup should include screening for thyroid abnormalities and baseline thyroid function tests are also recommended. Pituitary hormones, if thyroid functions are normal, are indicated. Primary adrenal and primary pituitary insufficiency can be differentiated with an early morning cortisol assay.3,8,10 Magnetic resonance imaging (MRI) can be obtained to visualise the pituitary gland to confirm the diagnosis of hypophysitis.3,10 MRI findings can be non-specific but can show a general enlargement of the pituitary gland.17,18In a review, about 85% of patients had pituitary gland abnormality on MRI.19 A retrospective analysis also postulated that radiological findings of irAEs could also be associated with a better clinical response.11
Treatment usually requires replacement of thyroid hormone and in mild cases of adrenal insufficiency, oral corticosteroid replacement therapy can be used.13 Adrenal insufficiency or crisis is a medical emergency. This warrants hospitalisation and high dose intravenous corticosteroids with mineralocorticoid activity. Infection or sepsis should be excluded. A consultation with an endocrinologist is needed to ascertain if long-term hormone replacement is required.8,10,13
Pulmonary immune-related adverse events Pulmonary irAEs are more common with PD-1 blockers, although the incidence is <1% and presents far later into treatment phase.10 In patients with new respiratory symptoms, infection or disease progression should be excluded before diagnosing immune related pneumonitis. A bronchoscopy and biopsy can be done in patients where aetiology is unsure.8,10
Ophthalmological immune-related adverse events
Episcleritis, conjunctivitis and uveitis has been described, and according to severity, topical steroids or systemic steroids can be utilised.3,18
Neurological immune-related adverse events
Sensory symptoms and motor symptoms can be experienced. Other irAEs such as posterior reversible encephalopathy syndrome, aseptic meningitis, enteric neuropathy, and transverse myelitis have been described. Cases of Guillain-Barre syndrome have also been noted on numerous occasions. In our opinion it is worthwhile to consult a neurologist to assist with diagnosis, differentiating between paraneoplastic phenomenon and treatment implementation. Individual cases may warrant plasmapheresis or intravenous immunoglobulin.3,8 Although immunosuppressive therapy or drug omission suffices in most cases.13
Haematological immune-related adverse events
Red cell aplasia, neutropenia, acquired haemophilia A, thrombocytopenia have all been described.3,8 Recently cases of haemolytic-uraemic syndrome occurring in patients receiving ipilimumab have been reported.20 All these haematological complications can be managed effectively with immunosuppressive therapy.
Others less frequent irAEs include pancreatitis, asymptomatic raise in amylase and lipase, renal insufficiency and nephritis.
Conclusion
When managing a patient with suspected irAEs, each case should be individualised. A thorough workup of each side effect should be done to ascertain whether or not there is truly an irAE. Most importantly, a high index of suspicion of irAEs must always be kept in mind. Although most irAEs are self-limiting, in severe cases if treatment is not given promptly and correctly, it can be associated with life-threatening complications and death. Early recognition and aggressive treatment with immunosuppressive agents is vital to prevent morbidity and mortality.