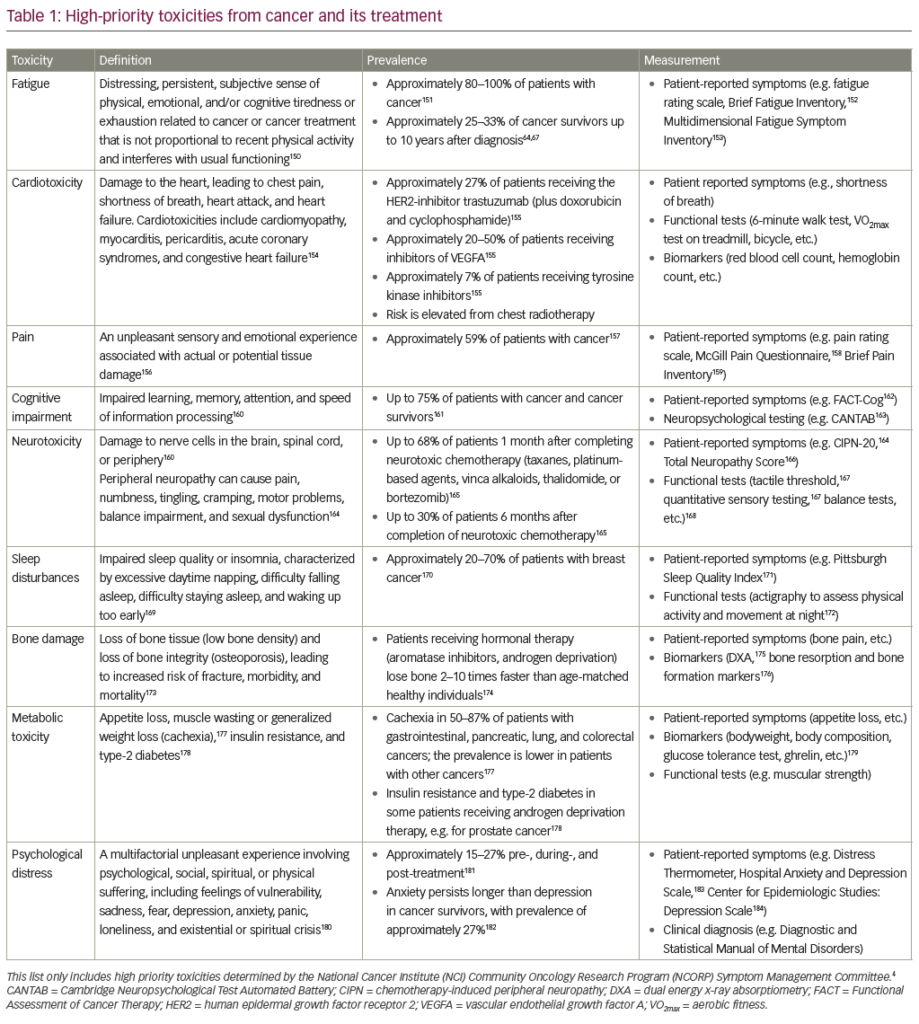
Cancer treatments and their side effects are often perceived as more debilitating than the actual cancer by many patients.1–3 Nausea and vomiting (NV) are still among the most common, expected, and feared side effects among patients receiving chemotherapy.1–3 In fact, some cancer patients who experience chemotherapy-induced nausea and vomiting (CINV) will delay chemotherapy treatments and contemplate refusing future treatments because of a fear of further NV.1–4 Anticipatory nausea and vomiting (ANV) are also problems among chemotherapy patients. By the fourth treatment cycle, anticipatory nausea has been reported in up to 30% of patients who experienced nausea following earlier chemotherapy cycles,5 while rates of anticipatory vomiting have been reported to be as high as 20%.6,7 CINV leads to poorer chemotherapy adherence, impaired functional activity, increased anxiety and depression, diminished quality of life (QOL), and increased utilization of healthcare resources, all of which substantially increase the burden of cancer and hinder its effective treatment.4,8–11 A great deal of progress has been made in the effective management of vomiting; however, nausea remains a substantial problem.1,2 The purpose of this article is to provide an overview of the pathopsychophysiology of CINV and the recommended guidelines for standard treatment, and to highlight newer targeted treatment approaches.
Pathopsychophysiology of Nausea and Emesis
Pathopsychology of Nausea and Emesis
Conditioning and Anticipatory Nausea and Vomiting
ANV is an unfortunate consequence of CINV, defined as the occurrence of nausea and/or vomiting in patients prior to treatment. The majority, if not all, of researchers agree that the development of ANV is strongly related to the Pavlovian conditioning model (see Figure 1), although it is still possible for patients to experience ANV without actually having previously experienced post-treatment NV.12 ANV can begin while the patient is still at home or on the way to the cancer clinic. ANV often increases in intensity as the patient moves closer in proximity to the actual room in which he or she receives treatment. ANV is a problem that adversely affects the QOL of patients7,13 and can interfere to the point at which a patient discontinues treatment.14 Once ANV develops, it cannot be controlled by normal antiemetic medications, including 5-hydroxytryptamine-3 (5-HT3) receptor antagonists.15
Expectancy and Nausea
Placebo and/or nocebo effects may contribute to the ineffectiveness of antiemetics in managing ANV, particularly nausea. Kirsch16 suggests that an anticipation or response expectancy for a physiological sensation such as nausea can generate corresponding subjective experiences that are then self-confirming. When such expectancies are non-volitional in nature (e.g. nausea from a noxious stimulus), Kirsch theorizes that expectancies directly affect both physiological and psychological outcomes, are self-confirming, and account for the placebo effect. Stewart-Williams asserts that expectancies are at the core of most placebo effects in human beings.17 Forgione18 suggests that “a placebo may be thought of as an accidental creation of expectancy.” Hahn19 and Barsky and colleagues20 propose that the expectation of developing side effects can cause side effects to manifest and elicit a nocebo. A review of 93 studies examining expectancy and placebo in the clinical environment concluded that expectancy plays a central role in the development of medical side effects and is the principal mechanism by which placebos elicit physiological effects.21
The classic conditioning paradigm does not fully explain the development of ANV,22,23 and a role for cognitive factors such as anxiety and response expectancies in ANV development exists.7,24–26 Anxiety is known to affect the development of NV at least in part through negative expectancies24,27–29 and, reciprocally, negative expectancies are instrumental in the development of anxiety.16,30 Expectancies affect the generation of conditioning effects31–33 and, conversely, conditioning influences response expectancies.16,34,35 Therefore, anxiety and response expectancies may mediate the conditioning patients toward ANV. Several studies have revealed that response expectancies significantly predict the occurrence of ANV.23,26,36–38 A descriptive study conducted by our research group with 63 female cancer patients with various types of cancer showed that expectancy strongly predicted the development of nausea prior to the third chemotherapy (p=0.001). Expectation of nausea was assessed prior to the first treatment on a five-point semantic rating scale anchored at one end by ‘1: I am certain I will not experience nausea’ and at the other end by ‘5: I am certain I will experience nausea.’13
Variations in the frequency and severity of CINV are not fully accounted for by the pharmacological properties of the chemotherapeutic agents or physiological characteristics of patients.39,40 The expectations of patients about developing CINV were postulated to account for some of this variance, and numerous studies have shown a positive relationship between expectations and the development of CINV.41–43 For example, Roscoe and colleagues44 studied 194 breast cancer patients about to begin their first cancer treatment regimen containing doxorubicin (see Figure 2). The results showed that a patient’s expectancy of nausea assessed prior to receiving the first chemotherapy cycle was a strong predictor of subsequent nausea and was a more robust predictor of severe nausea than previously reported factors such as age, nausea during pregnancy, and susceptibility to motion sickness. Patients who thought it was ‘very likely’ that they would experience severe nausea following chemotherapy were five times more likely to experience severe nausea compared with those who indicated that it would be ‘very unlikely’ that they would experience severe nausea following chemotherapy. These results suggest that patients’ expectancies related to nausea development are significant predictors of and likely contributing factors to CINV, especially nausea.
Pathophysiology of Nausea and Vomiting
The pathophysiology of vomiting is more straightforward than that of nausea, and is thought to involve mostly lower brain structures without general involvement of the cerebral cortex and other areas of higher development.45 Vomiting is a reflex triggered by toxic substances within the body such as chemotherapeutic agents. Broadly, these agents are sensed in the gastric or small bowl mucosa and cause the stimulation of vagal afferents that interact with the central nervous system (CNS), resulting in efferent vagal action that ultimately leads to an emetic response.8,46 Temporal occurrence, severity, and degree of emesis and/or nausea vary and are highly influenced by gender, race, age, and current and past health.47 Current knowledge of the biological properties of the emetic response and how individuals respond differently to chemotherapy has aided the development of therapeutic regimens for treating NV. Progress in preventing and controlling chemotherapy-induced vomiting has resulted in fewer patients experiencing vomiting during chemotherapy, whereas nausea continues to be reported by a majority of patients receiving emetogenic chemotherapy at some time during a course of treatment.48
Compound/receptor signaling is key to initiating and controlling the emetic response. The most widely studied compound is serotonin (5-hydroxytryptamine [5-HT]), which is produced by enterochromaffin cells and is a unique cell type dispersed throughout the enteric epithelium. These cells constitutively express 5-HT and, on exposure to a chemotherapeutic agent, the 5-HT is expressed more abundantly. At elevated levels, 5-HT is released from the basal surface into the lamina propria. There, secreted 5-HT binds to cognate 5-HT3 receptors located on vagus nerve terminals, thus acting as a neurotransmitter transducing a signal to the hindbrain. In turn, the translated signal triggers a motor response of NV carried by efferenting vagal nerves.49 In order to endogenously terminate the 5-HT-mediated NV response, the compound must be taken up by intestinal enterocytes through binding of the serotonin transporter (SERT) receptor.50
For approximately 20 years, 5-HT3 antagonists have been extremely useful for curbing NV in patients receiving chemotherapy. These drugs exert their antiemetic potential by competing with 5-HT for the binding of 5-HT3 receptors and thereby blocking a pro-emetic signal to the CNS. The newest 5-HT3 antagonist, palonosetron, has a higher receptor binding affinity than other commonly used 5-HT3 antagonists, which may make it more effective at preventing NV. Moreover, this drug has a half-life of 40 hours, which may allow more effective prevention of delayed NV than achieved with other 5-HT3 antagonoists.51 Since 5-HT synthesis is increased significantly after chemotherapy, another method of potential therapeutic benefit would decrease 5-HT synthesis in the gut. Since HT synthesis is dependent on tryptophan hydroxylase (TPH), this enzyme may represent a viable target. In fact, pre-clinical studies have taken place using a TPH inhibitor to selectively inhibit 5-HT in the gut using a ferret model of chemotherapy-induced emesis.52
Substance P is another strong regulator of the emetic response; it binds to the neurokinin-1 receptor (NK-1). Both compound and receptor are found within the CNS and the gut. Unlike 5-HT/5-HT3 receptor interaction, less is known about how and where substance P and neurokinin-1 act in promoting emetic potential, although peripheral and central components may be involved. Pre-clinical studies suggest that antagonizing neurokinin- 1 receptor action in the CNS is key to preventing NV, as agents that are incapable of crossing the blood–brain barrier do not protect against emesis.53 Clinically, the administration of aprepitant, the first drug devised to antagonize the NK-1 receptor, has proved to be effective in preventing NV when combined with currently used therapies.54
Dopamine release and cognate dopamine receptor-2 signaling may also play a role, as dopamine antagonists have been shown to be effective in treating NV, but far less is known about this mechanism. While participation of the CNS is clearly a major contributor to the emetic process, it is also possible that the enteric nervous system (ENS) itself may be able to control NV effects without CNS interplay. Further understanding of the role of more mysterious mediators may contribute to a broader range of antiemetic treatments for complete prevention of NV and to the treatment of patients with varying degrees of NV.
Current Scientific Evidence Regarding the Treatment of Chemotherapy-induced Nausea and Vomiting
Behavioral
A growing body of literature provides support for the efficacy of behavioural interventions for the treatment of CINV, especially when treating ANV. Behavioral interventions are especially appropriate for addressing ANV because it is a conditioned response,55,56 and interventions are best implemented prior to the complete/full development of the undesired conditioned response. Many of the same interventions that are effective in controlling NV have been used with success in treating ANV.57,58
One such intervention that might delay or prevent conditioned side effects of treatment59 is progressive muscle relaxation (PMR), in which one learns to relax by tensing and releasing specific muscle groups in a progressive manner.56 Patients who practice PMR have significantly less nausea during and immediately after chemotherapy,60 and experience a reduced duration of NV.61 PMR has a significant effect size in controlling nausea.62 Relaxation is often combined with guided imagery (focused attention designed to elicit specific physiological responses and attain specific health goals). Imagery mentally addresses all sensory modalities such that both psychological and physiological responses are generated as if an actual external stimulus had been presented.63 Guided imagery visualizing quiet scenes55 and imagery that is patient-specific and tailored around meaningful images patients have of their side effects or disease are both beneficial.58,64 Patients who receive a combined intervention of relaxation plus guided imagery have a reduced incidence of nausea during chemotherapy, lower incidence, duration, and severity of post-chemotherapy nausea, shorter duration of post-treatment vomiting,65,66 and a more positive experience with chemotherapy than patients who receive antiemetic treatment alone.67 Other interventions include hypnosis, suggestive therapy,68–71 systematic desensitization,72 biofeedback,56 wristbands,73 and electrical stimulation at the wrist.74
No single mode of delivery of these behavioral interventions is universally effective or flawless. One-to-one instruction (clinician to patient) of these strategies allows the clinician to tailor them to individual patient preferences and is more effective than instruction by audiotapes,75 but audiotapes allow for independent practice after live intervention59 and are cost-effective.56 The needs, resources, abilities, psychological history, and condition of the patient and treatment setting must be considered when choosing how to implement these interventions.64
Pharmaceutical
Advances in first-generation antiemetic agents and the introduction of new therapies have facilitated CINV control; palonosetron (Aloxi) and aprepitant (Emend) are the two newest antiemetic agents. Palonosetron, a second-generation 5-HT3 receptor antagonist, is a potent antiemetic agent with 100-fold greater binding affinity to type 3 serotonin receptor and a five times longer half-life (~40 hours) than other drugs in its class.8,10,11,76 A single dose (0.25mg intravenously [IV]) of palonosetron has been shown to effectively prevent acute and delayed NV associated with moderate to highly emetogenic chemotherapy, and is as effective as older antiemetic agents.8,10,11,76
Some studies have indicated that palonosetron is superior to first-generation 5- HT3 receptor antagonists (ondansetron and dolasetron), but only for secondary end-points (prevention of delayed emesis and overall tolerability) because primary end-points (no vomiting and no rescue medication required) were met in all studies.8,76,77 The more important issue is not the possible superiority of palonosetron to older 5-HT3 receptor antagonists, but rather whether palonosetron is better than earlier 5-HT3 receptor antagonists when combined with other drugs (specifically dexamethasone, a corticosteroid, and aprepitant).76 There have been no prospective trials specifically designed to prove the superiority of palonosetron over any 5-HT3 receptor antagonist, and no prospective trials comparing palonosetron with another 5-HT3 receptor antagonist when both are combined with dexamethasone.8 Future studies need to investigate the effects of various combinations of antiemetic agents (using older and newer agent combinations) and determine the optimal combination in preventing and controlling CINV.
Aprepitant, a neurokinin-1 (NK-1) receptor antagonist, represents the newest class of antiemetic agents. Phase III trials have shown that aprepitant is effective in controlling CINV. In several comparative trials over multiple treatment cycles, sustained antiemetic protection was better in the aprepitant group than in the control group.8,78–81 According to current guidelines,82–84 a three-drug combination of a 5-HT3 serotonin receptor antagonist, dexamethasone, and aprepitant is recommended before chemotherapy in high-emetic-risk patients; a three-drug combination of a 5-HT3 serotonin receptor antagonist, dexamethasone, and aprepitant or a two-drug combination of 5-HT3 serotonin receptor antagonist and dexamethasone for moderate-emetic-risk patients (depending on the chemotherapy regimen); and dexamethasone only for low-emetic-risk patients.
Despite the introduction of new antiemetic therapies, first-generation antiemetic agents continue to be used for controlling CINV. Two older 5-HT3 receptor antagonists (ondansetron and granisetron) continue to provide effective antiemetic coverage for CINV, can be less expensive than the newer agents, and are more likely to be covered by the patient’s health insurance.85 Several studies indicate that ondansetron and granisetron are both effective in meeting primary (complete control or no vomiting) and secondary (preventing delayed emesis) end-points. Additionally, the side effects (headache, hiccups, diarrhea, constipation, drowsiness, hypersensitivity, and tremors) of both drugs are mild and brief in duration.80 Nevertheless, despite the continued effectiveness of older antiemetic agents and the promise of new ones, CINV persists as a major problem in cancer care. While the primary concern is preventing vomiting, the problem of nausea becomes a greater challenge as strategies to control vomiting improve.1
Treatment Guidelines for Chemotherapy-induced Nausea and Vomiting
Clinical practice guidelines have been developed and published by panels of experts in order to facilitate implementation of currently accepted and evidence-based methods to control CINV (see Figure 3).82–84 Guidelines for the management of CINV classify this side effect into four broad categories based on time of onset (acute and delayed), expectation (anticipatory), and resistance to antiemetic treatment (breakthrough and refractory). Acute NV occurs within a few minutes to several hours following chemotherapy and is alleviated within 24 hours. Delayed NV starts >24 hours following chemotherapy and can last for up to seven days. ANV is conditioned or learned from previous experience with NV. Breakthrough NV is drug-resistant and more difficult to control. Refractory NV occurs despite active effort to control this side effect. Refractory patients do not respond to antiemetic treatments. Guidelines for the treatment of CINV also classified the likelihood of this side effect in the absence of antiemetic treatment for patients undergoing chemotherapy into four broad categories: minimal (10%), low (10–30%), moderate (30–90%), and high (>90%). Guidelines for the treatment of CINV also categorized different chemotherapy agents based on risk levels (i.e. minimal, low, moderate, and high).
Conclusions
Despite major advances in the treatment of CINV, including the development of new pharmaceutical and behavioral therapies and the establishment of standard clinical guidelines for effectively managing the illness, patients continue to experience CINV. Although the introduction of 5-HT3 and NK-1 antagonists has considerably reduced the incidence of CINV and ANV, they remain troublesome side effects among cancer patients. Clearly, there is a need for focusing attention on better understanding ANV, nausea alone, and other cases where antiemetics such as 5-HT3 and NK-1 antagonists become less effective.
Behavioral interventions that have been shown to be effective in helping to manage ANV, nausea alone, and CINV need to be included in standard clinical practice guidelines. Further research is needed that focuses on the development of targeted therapies that integrate pharmaceutical and behavioral interventions to maximize the effective reduction of ANV and CINV, not just vomiting alone.■