Over the last 10 years, the availability of neurokinin-1 receptor antagonists (NK1 RAs) has led to marked improvements in the treatment of chemotherapy-induced nausea and vomiting (CINV).
In clinical trials with the NK1 RA aprepitant in combination with ondansetron and dexamethasone (DEX), more than 70 % of patients treated with highly emetic chemotherapy (HEC), such as cisplatinbased regimens, achieved overall complete response (CR) (no vomiting, no rescue medication during the overall phase).1,2 Warr et al. showed that women receiving anthracycline and cyclophosphamide (AC)-based moderately emetic chemotherapy (MEC), patients receiving a 3-drug antiemetic regimen with aprepitant had overall CR rates over 50 % compared with 42.5 % with a two-drug antiemetic regimen without an NK1 RA (p=0.015).3 Similar outcomes were demonstrated in a study by Rapoport et al.4 The demonstrated advantage of adding a NK1 RA to antiemetic regimens has led to its inclusion as an essential component in CINV prophylaxis guidelines, in combination with a 5-HT3 RA and DEX.5–7
Aprepitant was initially approved by the US Food and Drug Administration (FDA) in 2003 for the treatment of CINV in combination with other antiemetic agents. It is currently administered as 3 doses taken orally: 125 mg before chemotherapy on day 1, and 80 mg on days 2 and 3.1–3 Fosaprepitant is a prodrug of aprepitant that is readily and rapidly converted to aprepitant via phosphatase enzymes in the bloodstream. Fosaprepitant is available as an intravenous (IV) formulation that can be used in place of the oral dose of aprepitant on day 1. It may be particularly advantageous in patients who cannot accept or tolerate an oral formulation.
A phase III study with another NK1 RA, casopitant, was completed for treatment of CINV in both 3- and 1-day dosing schedules. In July 2008, GlaxoSmithKline (GSK) filed a marketing authorisation application with the European Medicines Agency. The application was withdrawn in September 2009 because GSK decided that further safety assessment was necessary, and that it would take considerable time to produce these data. Nevertheless, the potential convenience of administering dosing the NK1 RA in the clinic without having to prescribe additional NK1 doses for patients to take home was of substantial interest to oncologists.
Other NK1 RAs are in phase III clinical development for CINV, including rolapitant and netupitant (NETU).8,9 Rolapitant is a potent, selective NK1 RA that is rapidly absorbed, has a remarkably long half-life (up to 180 hours) and appears to have a low potential for drug–drug interactions. Rolapitant has been free of clinically significant drug interactions in studies conducted to date. Specifically, it does not alter the pharmacokinetics of midazolam or other CYP3A4 substrates and, consequently, is unlikely to have an effect on the pharmacokinetics of drugs metabolised by cytochrome P450 3A4. Therefore, administration of rolapitant is unlikely to cause a clinically significant pharmacokinetic interaction with many commonly used drugs intended for cancer patients undergoing chemotherapy.10NETU is a potent and selective NK1 RA under development in combination with a fixed dose of palonosetron (PALO) for the prevention of CINV. NEPA, a fixed-dose combination of NETU – a new NK1 RA – and PALO – a pharmacologically distinct 5-HT3 RA – targets these dual antiemetic pathways and has been shown to uniquely work synergistically in vitro.9
Other NK1 RA, such as vestipitant, have been under investigation for other indications (e.g. tinnitus).11 Olanzapine is a thienobenzodiazepine that can act on multiple receptors including dopamine (D1, D2, D3, D4) and the serotonin receptors (5HT2a, 5HT2c, 5HT3, 5HT6) as well as adrenergic, muscarinic and histamine receptors.12 Common side effects are weight gain, sedation and impaired glucose tolerance. Its major use is as an oral antipsychotic agent. It has been found to be active in the prevention of both acute and delayed emesis in early phase trials, with chemotherapy of high emetic potential including cisplatin, and chemotherapy of moderate emetic potential including cyclophosphamide, doxorubicin or irinotecan.
Neurokinin-1 Inhibitor Clinical Pharmacology
Most of the current published NK1 clinical pharmacology literature is with the use of aprepitant. Both substance P and serotonin (5-HT3) have been implicated in CINV by triggering the corresponding receptors in the brain and in the gastrointestinal tissues, respectively.13 Substance P and 5-HT3 are generally thought to have different time courses of action due to the biphasic nature of cisplatin-based HEC;14,15 the 5-HT3– mediated effect occurs within a few hours of the administration of chemotherapy, early in the acute phase. The NK1-mediated effect starts at approximately 15 hours following chemotherapy, and continues into the delayed phase.15,16
While the 5-HT3 mediated phase is quite short and essentially complete in the first 24 hours after chemotherapy, the substance P/NK1-mediated phase in HEC extends for 60 or more hours postchemotherapy. 17,18 The acute phase of chemotherapy has been defined as the first 24 hours post-chemotherapy, and the delayed phase as any time in the 96 hours after that. These phase definitions are rooted in the need for readily measurable endpoints, rather than the biological phases associated with 5-HT3 and NK1. However, the 5-HT3 RA and NK1 RA are often associated with acute and delayed CINV, respectively. However, it is clear that the NK1 mediated effect spans both phases in HEC.17,19 AC-based MEC on the other hand is monophasic, with both the 5-HT3 and NK1-mediated effects occurring within a few hours after chemotherapy, early in the acute phase.15 When the same effects are evaluated in patients receiving AC-based MEC, aprepitant was effective earlier in the acute phase – starting as early as 6 hours post-chemotherapy – compared with approximately 18 hours in HEC studies.15,17 In a Kaplan-Meier analysis of time to first emesis in HEC studies, the curves representing the regimen with aprepitant and active control were clearly separated as early as 15 hours.15,17 This highlights the differential time course of action of 5-HT3 and NK1 RAs when used with HEC and MEC.15 Hence NK1 RAs, although often associated primarily with the delayed phase, also have a critical role during the acute phase in both HEC and MEC as it was shown in the registration trials.
A single 125 mg dose of oral aprepitant has been shown to have a mean plasma half-life of 14.0 hours; furthermore, at 24 hours after dosing the aprepitant concentration is 36 % of the peak concentration (Cmax).19 The pharmacokinetic profile of aprepitant after administration of IV fosaprepitant is shown in Figure 1. This is similar to that of oral aprepitant, because fosaprepitant is rapidly converted to active aprepitant following IV administration. In fact, the mean plasma halflife for fosaprepitant was found to be 2.3 minutes, suggesting that complete conversion occurs in less than 30 minutes.19 The maximum concentration of aprepitant following IV fosaprepitant administration takes place at 15 minutes compared with 4 hours for oral aprepitant administration. The aprepitant mean half-life was similar for the IV fosaprepitant 115 mg and oral aprepitant 125 mg doses (13.6 hours compared with 14.0 hours, respectively). These doses had nearly identical mean concentrations at 24 hours; 504 ng/ml compared with 494 ng/ml, respectively.19
More importantly, however, aprepitant not only remains detectable in the bloodstream for a relatively long period of time, it also binds tightly at the site of action in the brain. In vivo animal studies show that aprepitant may occupy up to 100 % of striatal NK1 receptors for at least 48 hours after exposure despite the fact that aprepitant levels were no longer detectable in the tissue after 24 hours.20 Figure 3 shows the results of a positron emission tomography (PET) tracer study in healthy individuals taking aprepitant 125 mg long term.21 In this study, an aprepitant plasma concentration of 216 ng/ml corresponded to an estimated NK1 receptor occupancy of 95 %.21 Although this study was conducted in healthy volunteers taking aprepitant long term, this plasma concentration is about half that reported by Lasseter et al.19after 24 hours following a single dose. Similar studies have reported =90 % receptor occupancy with single oral doses greater than 100 mg.20 Taken together with the favourable plasma pharmacokinetics, the slow dissociation and lasting functional inhibition at the NK1 receptors may facilitate the capacity for administration of aprepitant as a single dose prior to chemotherapy.
Clinical Data
Aprepitant
In two phase III trials, aprepitant plus ondansetron and DEX was shown to provide significantly greater rates of CR than ondansetron plus DEX alone in patients receiving cisplatin-based HEC.1,2 In these trials, patients were randomised to receive standard therapy of ondansetron (32 mg IV) and DEX (20 mg oral) on day 1 followed by DEX (8 mg bid) on days 2 through 4. Patients in the aprepitant group received aprepitant (125 mg oral), ondansetron (32 mg IV) and DEX (12 mg oral) on day 1, followed by aprepitant (80 mg oral) and DEX (8 mg oral) once daily on days 2 and 3, followed by DEX (8 mg oral) on day 4.1,2 The first of these studies evaluated 523 patients for efficacy and 568 patients for safety.2 The second study evaluated 521 patients in the efficacy analysis with 525 patients in the safety analysis.1 In a planned combined analysis of these two trials, the CR rates for the aprepitant group compared with the active control group were 86 % versus 73 % for the acute phase, 72 % versus 51 % for the delayed phase and 68 % versus 48 % for the overall 5-day period (p<0.001 for all time periods).17 In both studies, aprepitant was generally well tolerated and the incidences of adverse events and discontinuations were similar between the two treatment groups.1,2
Another large trial compared 3-day aprepitant plus day-1 ondansetron and 4 days of DEX to 4 days of ondansetron plus DEX in patients receiving HEC (n=484).22 CR rates for all time phases (overall, acute and delayed) were significantly higher in the aprepitant group. In the overall phase (days 1–5), 72 % of patients receiving aprepitant had CR compared with 61 % of patients receiving 4-day ondansetron plus DEX (p=0.003).22
Aprepitant has also been assessed in patients undergoing AC-based MEC.3 In this phase III study, patients undergoing breast cancer chemotherapy were randomly assigned to receive either an aprepitantcontaining or active-control regimen for prevention of CINV (n= 857).3 The treatment group received aprepitant (125 mg) plus ondansetron (8 mg) and DEX (12 mg) before chemotherapy and ondansetron (8 mg) eight hours after chemotherapy on day 1, followed by aprepitant (80 mg qd) on days 2 and 3.3 The control group received ondansetron (8 mg) and DEX (20 mg) before chemotherapy and ondansetron (8 mg) 8 hours later on day 1, followed by ondansetron (8 mg bid) on days 2 and 3.3 The aprepitant group had a greater CR rate over the 5-day assessment period than the control group: 50.8 % versus 42.5 %, respectively; p=0.015.3 A recent randomised, placebo-controlled study in 848 patients studied the efficacy of the same antiemetic regimens in a range of MEC regimens: including AC-, oxaliplatin- and carboplatinbased chemotherapies.4 This study found that significantly more patients receiving a regimen with aprepitant experienced no vomiting (76.2 %) compared with active control (62.1 %; p<0.001), which indicates that antiemetic regimens with aprepitant were also effective with other chemotherapy regimens.4 A study with aprepitant administered as a single dose on day 1 in combination with PALO and DEX in HEC has recently been reported.23 In this pilot trial, all 75 patients received PALO (0.25 mg IV) on day 1 and DEX on days 1.4.23 No significant difference was detected between the single and 3-day aprepitant dose groups.23 In both aprepitant groups, 93 % of patients were emesis free during 5 days of follow up compared with 50 % in the control group.23
A variation of this aprepitant single-dose protocol in patients receiving MEC has also been reported.24 Forty-one subjects received aprepitant (285 mg oral), DEX (20 mg oral) and PALO (0.25 mg IV) on day 1, with no antiemetics administered on subsequent days.24 This was a pilot study with no control group.24 In the overall phase, CR was seen in 51 % of patients. In the acute and delayed phases, 76 % and 66 % of patients, respectively, had CR.24 Although this study employed a high dose of aprepitant, no apparent safety concerns emerged during this study. While exploratory, these studies suggest that aprepitant as a single dose prior to chemotherapy is feasible.
Other Neurokinin-1 Receptor Antagonists
Fosaprepitant
Single day 1 dosing of fosaprepitant for HEC was investigated in phase III.25.26 Grunberg et al. conducted a randomised, double-blind, active-control design to test whether fosaprepitant is non-inferior to aprepitant.25 HEC-naive patients receiving (cisplatin .70 mg/m2) received ondansetron and DEX with a standard aprepitant regimen (125 mg on day 1, 80 mg on day 2, 80 mg on day 3) or a single-dose fosaprepitant regimen (150 mg on day 1). The primary endpoint was CR. A total of 2,322 patients were enrolled, while 2,247 were evaluable for efficacy. Antiemetic protection with aprepitant and fosaprepitant were equivalent within predefined bounds for non-inferiority. More frequent infusion site pain/erythema/thrombophlebitis was seen with fosaprepitant relative to aprepitant (2.7 % versus 0.3 %, respectively).
In a separate study Saito et al.26 evaluated the efficacy and safety of single-dose fosaprepitant in combination with IV granisetron and DEX in patients receiving HEC chemotherapy (cisplatin =70 mg/m2). In total, 347 patients were enrolled in this trial to receive the fosaprepitant 150 mg, IV, on day 1 in combination with granisetron, 40 µg/kg, IV, on day 1 and DEX, IV, on days 1–3 or the control regimen of placebo plus IV granisetron and DEX.
The percentage of patients with a CR was significantly higher in the fosaprepitant group than in the control group (64 % versus 47 %, p=0.0015). The fosaprepitant regimen was more effective than the control regimen in both the acute phase (94 % versus 81 %; p=0.0006) and the delayed phase (65 % versus 49 %; p=0.0025). The authors concluded that single-dose fosaprepitant used in combination with granisetron and DEX was well-tolerated and effective in preventing CINV in patients receiving HEC.
Casopitant
In phase II trials, various casopitant dosing schemas, in combination with ondansetron and DEX, significantly reduced both HEC- and MECassociated CINV as measured by CR compared to ondansetron plus DEX alone.27,28 These trials included an exploratory single-day regimen of casopitant (150 mg oral) (see Figure 2). Having demonstrated efficacy in these trials, both the casopitant 3-day IV plus oral and a single oral dose were incorporated into the phase III trial design. Results of phase III trials of casopitant in HEC have shown that both the single oral dose and the 3-day IV/oral regimen, in combination with ondansetron and DEX (also dosed on days 2-4), provided significantly improved CR rates compared with ondansetron and DEX alone: 85.7 % and 79.6 % versus 66.0 % (p<0.0001 and p=0.0004).29 Although casopitant was discontinued for further development, these data provide a proof of concept to support the usage of NK1 inhibitors over 1 day. Treatment was well tolerated with similar adverse event and discontinuation frequency across study arms.29 Rolapitant Rolapitant is a promising agent and was studied in a multicentre, randomised, double-blind, placebo-controlled, dose range-finding study of rolapitant administered orally in subjects receiving highly emetogenic chemotherapy (HEC =70 mg/m2 cisplatin-based chemotherapy). The study included a total of 454 subjects who were randomised to receive ondansetron, DEX and either placebo or 10, 25, 100 or 200 mg of rolapitant prior to the administration of cisplatin on day 1 of each cycle. The rolapitant 200 mg group had significantly greater CR in the overall, acute and delayed phases compared with the control group (62.5 % versus 46.7 %; p=0.032; 87.6 % versus 66.7 %; p=0.001 and 63.6 % versus 48.9 %; p=0.045, respectively).30
Of interest, these patients had greater rates of no significant nausea (a maximum visual analog score [VAS] <25 mm) in the overall, acute and delayed phases compared with the control group (63.2 % versus 42.2 %; p=0.005; 86.5 % versus 73.3 %; p=0.029 and 64.4 % versus 47.8 %, p=0.026, respectively). This would suggest a benefit over current therapies as nausea remains a clinically significant problem in these patients. Rates for no emesis and no nausea for the 200 mg dose group in cycles 2 to 6 were sustained in subsequent cycles.
Netupitant
Recent data in a phase III multinational, randomised, double-blind, parallel group study, assessed the efficacy and safety of a single oral dose of NEPA (NETU 300 mg + PALO 0.50 mg) versus a single oral 0.50 mg dose of PALO in 1,455 chemotherapy-naive patients receiving anthracyclinebased chemotherapy (all patients received oral DEX 12 mg and NEPA or 0.5 mg PALO on Day 1). The primary efficacy endpoint was CR during the delayed (25–120 hour) phase. The majority of the patients were white females undergoing treatment for breast cancer. NEPA showed superior CR rates compared with PALO during the delayed, acute and overall phases. NEPA was also superior to PALO during the delayed/overall phases for complete protection defined as no emesis, and no significant nausea. The incidence of headache and constipation was low (3.3 % and 2.1 %, respectively). The authors reported no evidence of any cardiac safety concerns for NEPA or PALO, they concluded that NEPA is superior to PALO in preventing CINV in breast cancer patients receiving MEC.31
Olanzapine
Olanzapine may prove to be useful in the management of acute and delayed nausea, where as conventional antiemetics (5-HT3 RA, steroids and NK1 RA) are more efficacious in preventing vomiting. Olanzapine has been used in a phase II trial with granisetron and DEX with high response rates. In a phase II trial of Olanzapine in combination with PALO and DEX, the CR in eight patients receiving HEC was 100 % with 75 % CR in the delayed and overall phases.32
Navari et al. recently conducted a phase III study in 247 patients receiving cisplatin or doxorubicin plus cyclophosphamide. The trial showed the superiority of the olanzapine over aprepitant for the prevention of delayed nausea.33 In this trial, patients were randomly assigned to olanzapine (10 mg orally on the day of chemotherapy, and then 10 mg once daily on days 2 through 4), or aprepitant (125 mg orally prior to chemotherapy, followed by 80 mg orally on days 2 and 3), both arms received PALO (0.25 IV on the day of chemotherapy) plus DEX. DEX (20 mg) was only given on the day of chemotherapy, while the aprepitant group received DEX 12 mg on the day of chemotherapy followed by DEX 4 mg twice daily on days 2 and 3. Olanzapine may also be useful for management of breakthrough emesis.
The olanzapine data provides an avenue for future research and the improvement of patient outcomes. This needs to be confirmed in multicentric randomised phase III studies.
Current Trends and Future Directions
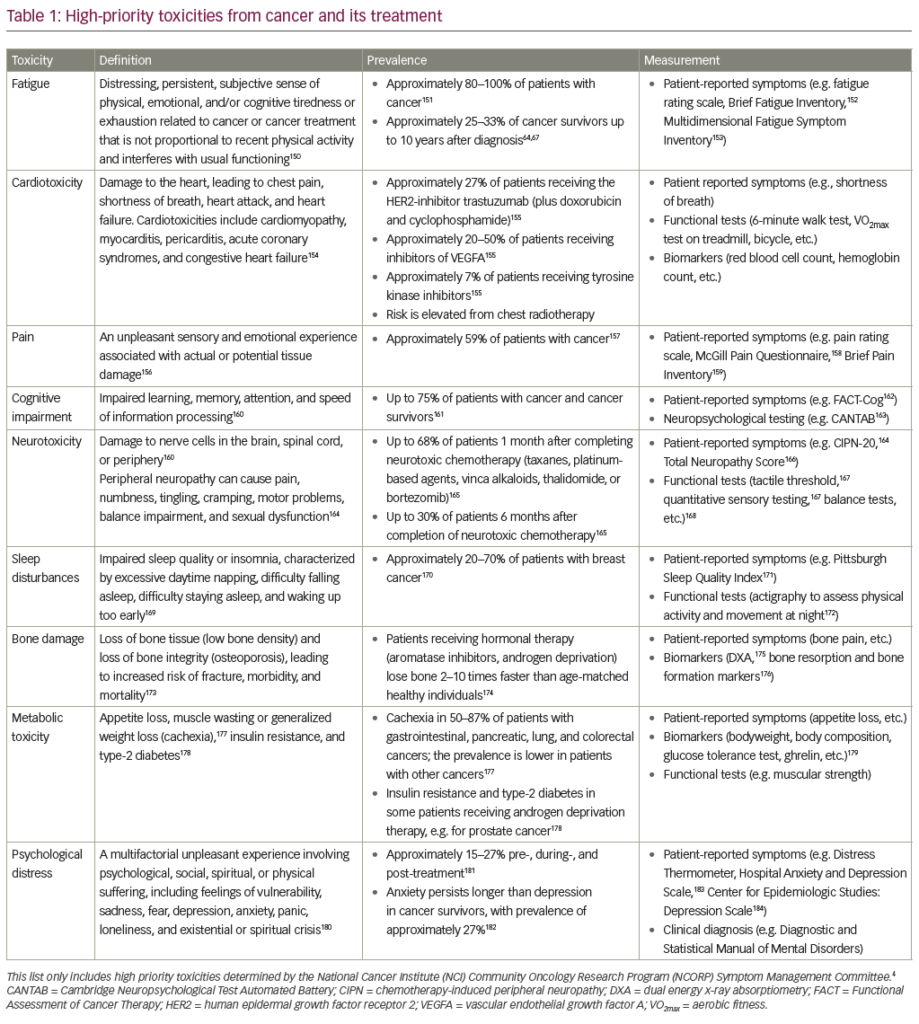
Current evidence-based indications for the usage of NK1 RA are summarised in Table 1 according to the Multinational Association of Supportive Care in Cancer (MASCC), the National Comprehensive Cancer Network (NCCN) and the American Society for Clinical Oncology (ASCO) guidelines.5–7 Aprepitant remains the only NK1 RA currently licensed for CINV. The other agents are under phase III development. The utility of aprepitant was recently re-demonstrated in an extensive meta-analysis.34 Aprepitant administered on 1 day may be preferable to the 3-day regimen.
Nausea and vomiting remains the most feared side effects of chemotherapy.35 Nausea alone may affect over 50 % of patients during the acute and delayed phase following chemotherapy.36 5-HT3-mediated CINV occurs in the hours immediately following chemotherapy. As a result, 5-HT3 RA are mostly effective during the early part of the acute phase of CINV. Substance P-mediated CINV occurs later in the acute phase and continues into the delayed phase, especially in HEC, so the NK1 RA may provide clinicians and patients with an important tool against nausea and vomiting that continue after the patient leaves the clinic.
NK1 RA in combination with 5-HT3 RA and DEX are now the preferred treatment for preventing CINV associated with HEC and MEC ACbased chemotherapy. Aprepitant is available in an oral formulation dosed on days 1 through 3, or an intravebously administered prodrug fosaprepitant, which can be used in place of the aprepitant dose on day 1. Casopitant was withdrawn for further development. Other promising NK1 inhibitors are in clinical development.
Recent studies have investigated use of a single dose of NK1 RA in combination with other antiemetics prior to therapy on day 1 in both HEC and MEC. This strategy is supported by the pharmacology. The plasma half-life of aprepitant of 14 hours, and of casopitant of 16-17 hours, supports the fact that high levels of the drug remain in the blood beyond 48 hours following a single dose.19,22 More importantly, in vitro and in vivo data (as well as PET imaging data in humans) suggest a tight binding affinity of the NK1 RA for the NK1 receptor, with receptor occupancy as high as 95 % or more for over 2 days after dosing.20–22 Clinical data also support the effectiveness of single, day- 1 dosing regimen, with efficacy demonstrated following single-dose administration of aprepitant and casopitant.23,24,37
Newer NK1 RAs are also administered on day 1. Olanzapine is a thienobenzodiazepine that can act on multiple receptors including dopamine and the 5-HT3 receptors as well as adrenergic, muscarinic and histamine receptors. This agent’s precise role may be defined in future trials for the prevention and rescue of CINV, in combination with or replacing centre inhibitors in the management of CINV.