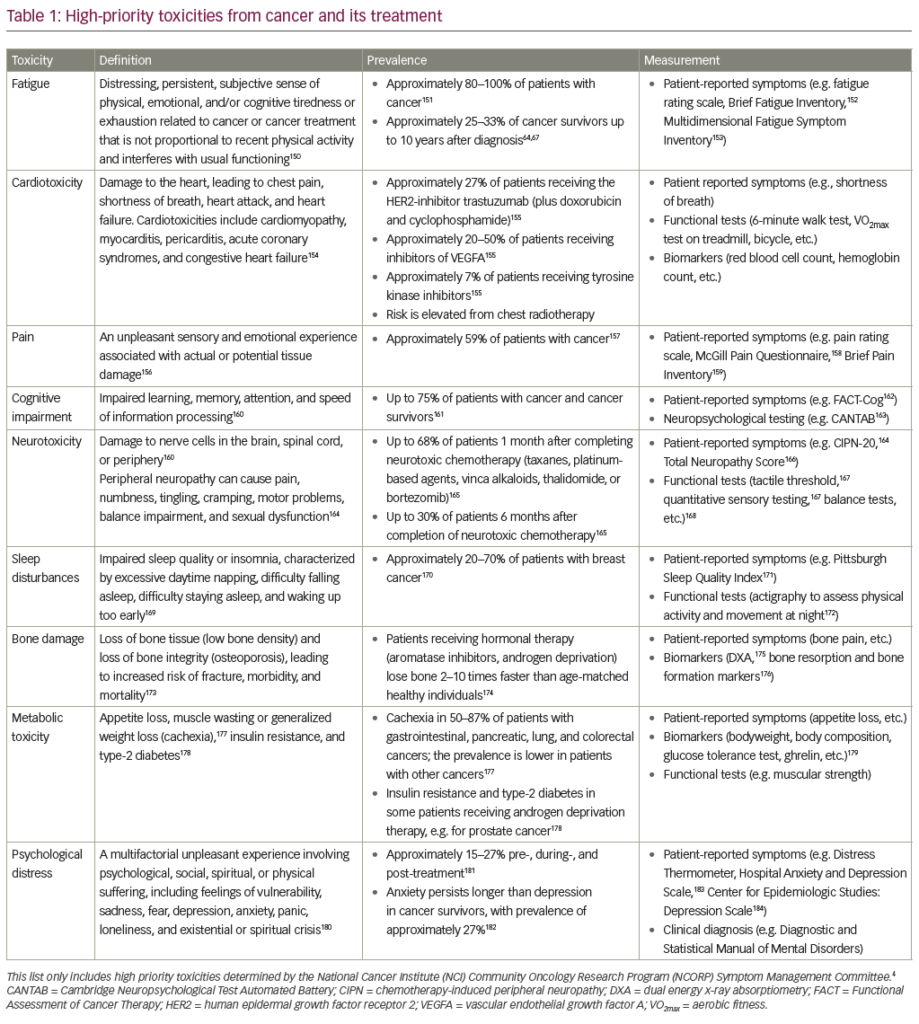
Cancer and its treatments result in side effects that impair quality of life (QoL). The most common side effect reported by cancer patients is cancer-related fatigue (CRF).1–4 Cancer patients report that CRF begins with diagnosis and worsens during the course of treatment. It can persist for months, and years, after treatment is complete.1–4 CRF often continues even when the patient’s cancer is undetectable or in remission.1–4 Cancer patients report a prevalence of CRF ranging from 60 to 100%, with 41% or more indicating severe CRF during treatment.1–4 As many as 81% of patients report persistent CRF, with 17–38% indicating persistent severe CRF more than six months after completing treatment.1–4
CRF is a multidimensional, subjective, and objective physiological state that is characterized by a persistent, overwhelming exhaustion and a decreased capacity for physical and mental work.1–4 CRF is characteristically different from the fatigue experienced by healthy individuals in its severity, its impact on QoL, and its lack of alleviation by rest or sleep.1–4 CRF often requires that survivors depend on others for simple activities of daily living, such as transportation, preparing food, or bathing.1–4 These difficulties with daily activities lead to a lack of self-sufficiency and can be demoralizing and discouraging for patients. CRF is frequently reported by patients as more distressing and having a greater negative impact on their daily activities and QoL than other cancer-related side effects including vomiting, nausea, pain and depression.1–4 The purpose of this article is to provide an overview of CRF and its pathopsychophysiology and to summarize the evidence for exercise as an effective intervention for managing CRF.
Pathopsychophysiology of Cancer-related Fatigue
CRF and its underlying pathopsychophysiological mechanisms may derive from the cancer itself, the side-effects of cancer treatment and other co-morbid conditions.4,5
Cancer-related Fatigue and Concurrent Side Effects
The concept of symptom clusters—two or more concurrent side effects strongly related to each other but not necessarily sharing the same pathopsychophysiology—has recently emerged.6–8 It is hypothesized that these co-occurring side effects may reciprocally influence the pathopsychophysiological development of each other (e.g. sleep disruption results in fatigue, depression results in fatigue, and fatigue results in sleep disruption and depression). CRF occurs simultaneously with an average of 13 additional side effects.9 These concurrent side effects may mediate the pathopsychophysiology of CRF.
CRF co-occurs with depression, which is reported by 10–25% of cancer patients.10 A recent review of 16 studies reported that CRF and depression were moderately correlated (r=0.55) among cancer patients.11 CRF also co-occurs with sleep disruption, which is prevalent in 30–50% of cancer patients depending on the definition of sleep disruption, mode of assessing sleep disruption, and cancer type and stage.9,12
CRF and insomnia are moderately correlated (r=0.26–0.54).11 This is exemplified in medically ill patients who spend a lot of time in bed, causing the distinction between sleep and wake cycles to blur: daytime activities are interrupted by napping/rest due to fatigue and the night sleep time is interrupted by activity resulting in fatigue.13
Pain is reported by 45–59% of cancer patients and frequently co-occurs with CRF.9–15 Forty-eight percent of cancer patients report anxiety and 18% meet criteria for anxiety disorder.16 CRF co-occurs with anxiety and the two are moderately correlated (r=0.48–0.62). Cognitive difficulties occur in approximately 25–80% of cancer patients and they often co-occur with CRF.5,17
Pathophysiological Mechanisms of Cancer-related Fatigue
Muscle wasting and muscle weakness are common results of cancer and its treatment and both may contribute to CRF.4,18–20 Adenosine triphosphate (ATP) is a key mediator in generating muscle mass and in contractile function. Decreased ATP synthesis may play a significant role in the development of CRF.4 A recent study showed that ATP infusion in patients with non-small-cell lung carcinoma improved elbow flexor muscle strength, increased energy, and reduced tiredness and shortness of breath.21
The immune system consists of interactions between various myeloid and lymphoid cell types and the effector molecules they produce (e.g. antibodies and cytokines) that are responsible for controlling infection and responding to injury. Controlled immune reactions are vital to preventing chronic inflammatory conditions. Numerous cytokines (e.g. interleukin [IL]-1β, tumor necrosis factor [TNF]-α, and IL-6) are present at higher levels in the serum of cancer patients experiencing CRF during and after treatments compared with the serum of cancer patients not experiencing CRF.22,23 These molecules are released as part of the innate response to tissue damage from site-specific macrophages, epithelial cells, and T cells in response to cancer and its treatments. Research has shown that cancer patients with CRF have significantly higher numbers of monocytes, CD4+ T-cells, and natural killer cells (which are all capable of cytokine production) compared with patients without CRF.24–28 High cytokine release (e.g. IL-1, IL-6, and TNF-α) is also linked to cognitive impairment and depression in cancer patients.29,30 This research suggests that CRF may share a common underlying pathopsychophysiology involving aberrant immune functioning.4
The hypothalamic–pituitary–adrenal axis is a major part of the neuroendocrine system, controlling many body processes including energy expenditure, mood, digestion, sleep, and immune function. In response to stress, the hypothalamus releases corticotrophin-releasing hormone, which then signals the anterior pituitary to release adrenocorticotropic hormone (ACTH). ACTH subsequently stimulates production of cortisol by the adrenal cortex. Cancer patients who experience CRF have significantly lower waking serum cortisol levels compared with patients without CRF,28 suggesting that disrupted cortisol and perhaps circadian rhythms are involved in the pathopsychophysiology of CRF.4 Abnormal cortisol levels and rhythms (measured throughout the day) have also been observed in metastatic breast cancer patients experiencing sleep problems, suggesting that CRF and sleep disruption share a common pathopsychophysiology.31 Chest radiation and certain chemotherapy agents have significant acute and chronic side effects that impair cardiovascular function and damage the heart.32–34 The acute cardiac effects of radiation include vascular tissue inflammation, vascular dilation, increased capillary permeability, and interstitial edema.32–34 These acute effects can progress to chronic problems leading to coronary artery disease, pericarditis, cardiomyopathy, valvular disease, and conduction abnormalities.32–34
Chemotherapy-induced cardiotoxic side effects are dose-dependent: cardiac damage increases with higher doses.33,34 Chemotherapy-induced damage results in sinus tachycardia, premature atrial and ventricular beats, supraventricular arrhythmias, coronary syndromes, heart failure, and pericarditis/myocarditis.33,34 Chemotherapy, particularly with anthracyclines, can also lead to chronic effects such as systolic and/or diastolic left ventricular dysfunction and severe congestive cardiomyopathy.33,34 CRF is a common symptom and an early pre-clinical indicator of cardiotoxicity in cancer patients.32 CRF severity is worsened by diminished cardiac function. Cardiac dysfunction causes more work and stress to the heart, which in turn leads to even more CRF, suggesting that CRF and cardiac damage may share a common pathopsychophysiology.32
Exercise and Cancer-related Fatigue
The current National Comprehensive Cancer Network (NCCN) guidelines recommend physical activity/exercise as a behavioral intervention for reducing CRF.35 Current scientific evidence suggests that physical exercise is safe for and well-tolerated by cancer patients with different types of cancer diagnosis throughout the cancer care continuum (post-surgery or transplant and during and after chemotherapy, radiation therapy, and/or hormone therapy) and that exercise shows great potential in mitigating CRF.1,36–46 Although the NCCN guidelines recommend physical exercise for the management of CRF, no clear evidence-based guidelines exist for prescribing an optimal dose (i.e. frequency, intensity, duration, and mode) of exercise for reducing CRF. The following summary provides a preliminary framework to assist oncologists and other healthcare providers in recommending physical exercise for patients with CRF.
More than 80% of cancer survivors prefer their oncologist to initiate discussion about participating in an exercise regimen.47 Patients who receive exercise instructions from their treating physician return to exercise more quickly after treatment and have better adherence.48–50 It is important for oncologists to discuss with patients how they can safely begin an exercise program and to inform patients of any potential limitations (e.g. orthopaedic, cardiopulmonary, oncological, etc.) that can affect exercise tolerance. Unfortunately, most cancer patients do not discuss initiating or continuing an exercise program with their treating oncologist or primary care physician.47,51–53
Cancer patients can benefit from an oncology referral to a qualified exercise specialist. The majority of cancer patients report preferring to receive exercise counseling from a qualified exercise professional affiliated with the cancer center in which they receive treatment.51 Qualified exercise professionals include individuals with formal education at bachelor’s level or higher in accredited exercise science or kinesiology programs. Additional certification by the American College of Sports Medicine with the Oncology Specialty would be preferable.43,54,55 Recently, the ACSM endorsed “exercise testing and prescription guidelines for cancer patients and survivors.”55 Generally, these guidelines recommend that exercise is safe and feasible for cancer patients and survivors and that they are not required to undergo exercise tolerance testing prior to initiating an exercise program. However, cancer patients and survivors with medical conditions placing them at moderate to high risk for complications should undergo preliminary screening for exercise tolerance and safety as suggested by the non-cancer specific ACSM guidelines. In addition, special consideration and additional screening should be given to cancer patients and survivors with peripheral neuropathies, musculoskeletal co-morbidities, increased fracture risk, arm and shoulder morbidity, wasting syndromes, lower extremity lymphadema and increased risk of infections secondary to cancer treatments.55
Exercise prescriptions for cancer patients should be individualized and tailored, considering the disease site and stage, planned treatments, and the individual’s current fitness level, along with past and present exercise participation and preferences. The ACSM guidelines recommend that cancer patients and survivors achieve 150 minutes a week of moderate physical exercise.55 These guidelines are not specific for fatigue reduction and may be difficult for many cancer patients and survivors to achieve. Research suggests that exercise interventions involving moderately intense (55–75% of heart rate maximum— corresponding to a rating of perceived exertion between 11 and 1456) aerobic exercise (e.g. walking and cycling) ranging from 10–90 minutes in duration, three to seven days a week are consistently effective at managing CRF among cancer patients with an early-stage diagnosis (i.e. non-metastatic disease).1,45 Stationary cycling may be a useful mode of physical exercise for patients with impairments such as ataxia or balance difficulties.49 Short bouts of activity (three to 10 minutes) accompanied by periods of rest culminating in a total of 30 minutes daily can also be effective at reducing CRF.57,58
Preliminary research suggests that progressive resistance exercise (e.g. therapeutic resistance bands, dumb-bells, fixed-weight systems) performed three times a week at a moderate to vigorous intensity (60–90% of one-repetition maximum) is suitable. This should be progressively increased up to two to four sets ranging from eight to 15 repetitions. This regimen has been shown to be effective at reducing CRF among cancer patients.1,45,59–61 Studies have also demonstrated that low intensity exercise is safe and well-tolerated by patients with metastatic disease.1,45 To decrease the risk for lymphadema, compression sleeves should be worn when appropriate.1,45,62,63 It is also prudent to advise cancer patients to avoid excessive high-intensity exercise that can potentially compromise the immune system and interfere with treatment and recovery.64
Although the exercise and cancer control literature provides consistent and growing support for the efficacy of exercise interventions in managing CRF during and after treatment, this body of literature remains relatively preliminary. Studies have small sample sizes, lack consistency in the exercise interventions employed, and vary widely in CRF assessment methods and statistical analyses.1,38–40,45 Despite these limitations, a meta-analysis by Schmidtz and colleagues41 reports that the evidence for exercise as an effective therapy for managing CRF is indeed consistently positive. Weighted mean effect size was 0.13 (confidence interval [CI] -0.06–0.33) during treatment and was 0.16 (CI -0.23–0.54) post-treatment, indicating that exercise is helpful in the management of CRF.41 More clinical research to develop specific exercise prescription guidelines is needed.
Cancer-related Fatigue, Aging, and Exercise
Patients who are >65 years of age are the fastest growing segment of the population and by 2030 will comprise 20% of the US population.65 A typical cancer patient today is over 65 years of age, has multiple medical problems, and is taking many medications simultaneously.66 Co-morbid conditions and medications coupled with age-related physiological decline have an aggregate impact in a vulnerable or frail elderly person who is now burdened with cancer and its treatment-associated complications.67 CRF is a common symptom reported by older cancer patients and frequently co-exists with other symptoms, such as pain and depression.68 Exercise has been shown to be useful in alleviating CRF and may be particularly helpful in preserving muscular strength, balance, and functional status among elderly cancer patients.
Little research has been devoted to the effects of exercise on CRF in older cancer patients and to the effects of exercise on physical performance and disability in this population.69,70 In one small study, a walking program improved CRF and sleep disruption in an older cohort of breast cancer patients receiving hormonal treatment.71 In one systematic review of the effects of exercise on CRF, nine experimental and 10 observational studies provided evidence that physical activity may be an effective intervention for CRF in older adults.72 The generalizability of these studies, however, was limited by poor representation of the older age group in the actual patient samples of these studies. Few studies provided an age-related analysis of the effects of physical activity on CRF, physical functioning, and QoL in older adults. There are growing numbers of elderly cancer patients receiving treatment and becoming survivors, so more research evaluating the effects of exercise on CRF and other health outcomes such as physical performance, disability, and health-related QoL is imperative.
Cancer-related Fatigue, Underserved and Minority Populations, and Exercise
Very little is known about the effectiveness of exercise interventions on CRF in minority and underserved populations. Clinical trials in this area either fail to report the racial and ethnic composition of their samples or are composed primarily of US non-Hispanic whites or homogenous international populations (e.g. Australians and Europeans). Well-documented cancer health disparities in incidence, mortality, and access to appropriate treatment, coupled with the high prevalence of CRF among cancer patients, make understanding the effectiveness of exercise as an intervention particularly important.
Several insights from the broader physical activity literature may help to guide the design and delivery of exercise interventions for CRF in minority cancer populations. Levels of physical activity are lower for ethnic minority populations, women, those with lower socioeconomic status, the elderly, and people with disabilities.73,74 The most successful physical activity interventions with ethnic minority and other underserved populations utilize a community-based participatory research (CBPR) model.74,75 In studies utilizing a CBPR framework, members of the target populations are involved in every phase of the research process, from design to dissemination.74 Members of the community (e.g. lay health advisors) often deliver the interventions.74 This allows for knowledgeable input from community members regarding the feasibility of particular interventions and the likely barriers to implementation.
Another emphasis of the minority physical activity literature has been the importance of using a social ecological theoretical model in order to understand health behavior and to design interventions. The social ecological model examines physical activity at the intrapersonal, interpersonal, community, environmental, and organizational levels. For example, intrapersonal barriers to physical activity among African- American women include multiple co-morbidities, lower ratings of health status, lower socioeconomic status, and lower levels of self-efficacy.75 On the interpersonal level, however, social support has been shown to be a facilitator of physical activity, with African-American women often engaging in activity with groups of family or friends and reporting companionship as one of the primary benefits of such activity.76 At the community and environmental level, the availability of safe, affordable, and convenient facilities for physical activity are a major concern for minority populations. Community resources, such as churches and other community organizations, may also provide an essential support for physical activity among minority populations.
Additional research is needed to determine the efficacy of exercise interventions for CRF among minority and underserved populations. The physical activity literature suggests that sustained and engaged involvement of the target population is critical to the success of interventions with these populations. Oncologists may need to go beyond the immediate clinical setting and consider partnering with community organizations to better assist these patients. It may also be helpful to recommend exercise interventions that incorporate a strong group component to take advantage of social support as a facilitator of physical activity. It will also be necessary to identify safe and convenient spaces for exercise in addition to recommending exercise appropriate for individuals with multiple co-morbidities, lower self-rated health status, and physical disability.
Summary
Major advances have been made in treating cancer and increasing survival. Unfortunately, most patients report CRF that begins during treatment and may persist for many years after treatment is complete. This CRF can interfere significantly with a patient’s ability to complete treatments for her/his cancer, recovery and QoL.
Strong preliminary evidence suggests that physical exercise can be effective in helping to manage CRF. NCCN guidelines recommend the consideration of physical exercise for managing CRF. At this time only general exercise participation guidelines can be summarized from current literature due to limitations in extant research. Further clinical research is clearly needed to develop specific exercise prescription guidelines for implementation in standard clinical oncology settings. ■