Incidence of Venous Thromboembolism in Patients with Cancer
The tumour itself is associated with the development of the hypercoagulable state, but cancer patients are also exposed to a range of iatrogenic factors known to independently increase the risk of VTE, including surgery, immobilisation, central venous catheters, chemotherapy, antioestrogen therapy or new antiangiogenic agents.6 Several sources of data are available for estimating the incidence and time course of symptomatic VTE among patients with different types and stages of cancer.
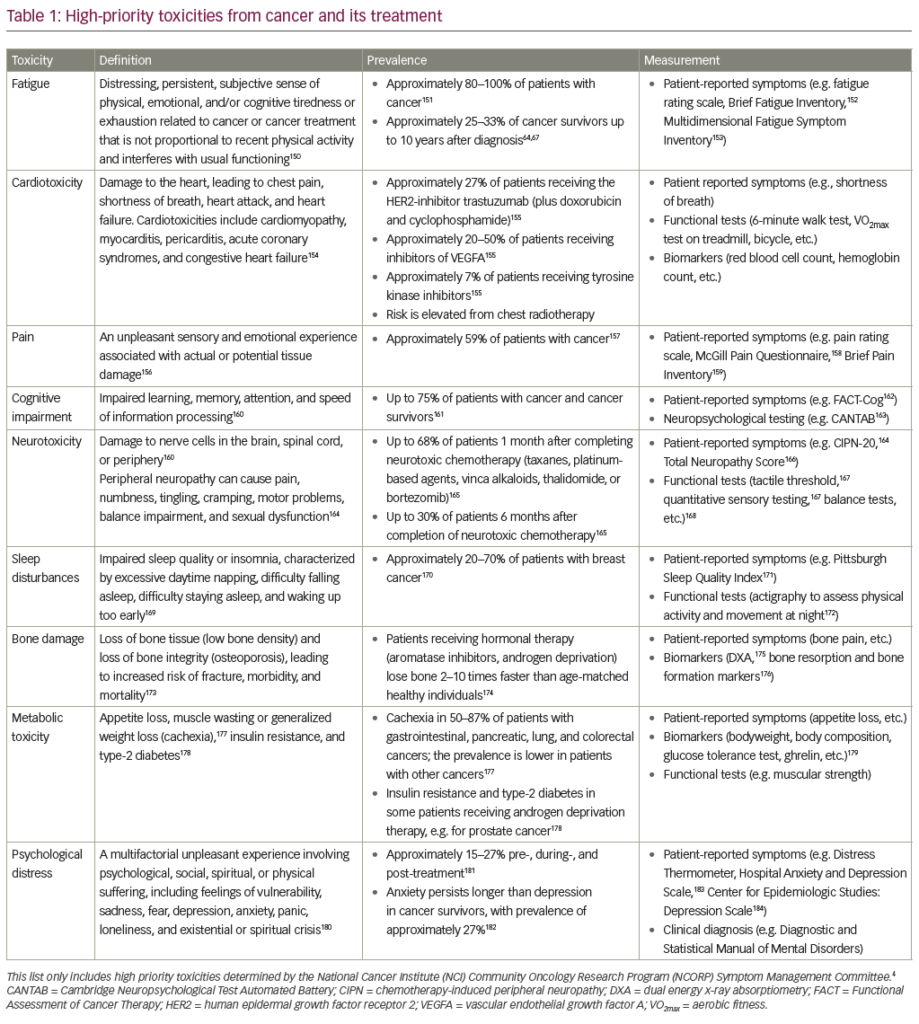
One large and recent study, based on the California cancer registry linked to the California patient discharge data set, showed that of 235,149 cancer cases, 1.6% were diagnosed with VTE within two years.7 Metastatic disease at the time of diagnosis was the strongest predictor of thromboembolism, with these patients having a 1.4–21.5- fold higher risk of VTE than patients with localised disease. The incidence of VTE also varied according to the type of cancer. The highest annual incidence of VTE was observed among patients with cancer of the pancreas (4.2% in localised disease versus 20% in metastatic disease), stomach (2.5 versus 10.7%), uterus (0.8 versus 6.4%), kidney (1.2 versus 6%) and lung (1.1 versus 5%).7 Further evidence of cancer-associated VTE was shown in a recent cohort study of patients with lung cancer, who had a 4.4% annual incidence of VTE, with the risk being greatest during the first six months after diagnosis and in patients with adenocarcinoma.8 In addition, the risk of VTE increased three-fold during chemotherapy, was doubled by radiotherapy and was six times higher for patients with distant metastases than for patients with localised lung tumours. The risk of thrombosis among acutely ill patients with cancer is being increasingly recognised. In a large randomised comparison of low-molecular-weight heparin (LMWH) and placebo in hospitalised medical patients with acute illness, cancer was independently associated with a 1.6-fold increase in the risk of VTE in the placebo group.9 In a study based on hospital discharge summaries of over a million patients with cancer, hospitalised in one of 133 academic medical centres in the US, symptomatic VTE was reported in 4.1% of patients.10 The rate of VTE increased by 28% from 1995 to 2003, due to a near doubling of the PE rate. Subgroups of cancer patients with the highest rates of VTE included those with black ethnicity and patients receiving chemotherapy.
Data from the same group previously reported a 5.4% incidence of VTE in 66,106 adult neutropenic cancer patients hospitalised in 115 medical centres.11 Clinical variables associated with thromboembolism included age over 65 years, primary site of cancer (including lung, gastrointestinal, gynaecological and brain cancer), co-morbidities and obesity. The in-hospital mortality rate was significantly greater among patients with VTE than those without VTE (odds ratio [OR] 2.01; 95% confidence interval [CI] 1.83–2.22), and this increase in mortality was observed in patients with localised cancer as well as those with advanced disease. In view of these risks, international guidelines recommend the use of prophylaxis with low-dose unfractionated heparin (UFH) or LMWH for patients with active cancer who are confined to bed in hospital.12,13
Treatment of Venous Thromboembolism in Patients with Cancer
Initial Treatment
Heparins are commonly used for the initial treatment of VTE in cancer patients. Compared with UFH, LMWH is easier to administer and does not require dose adjustment or monitoring.14 A meta-analysis of studies comparing UFH with LMWH for the initial treatment of VTE suggests that in cancer patients initial treatment with LMWH is at least as safe and effective as UFH. The same data suggest that LMWH may be associated with a reduced mortality risk in these patients.15
Long-term Anticoagulation
According to several cohort studies of patients with VTE, patients with cancer have a two to three-fold increase in recurrent VTE and a two to sixfold increase in major bleeding during oral anticoagulant treatment (OAC) compared with non-cancer patients.16,17 In general, vitamin K antagonists (VKAs) are difficult to use in patients with cancer due to the potential forinteractions of these agents with other medications, especially during chemotherapy, or problems with anticoagulant control arising from poor intestinal absorption or altered hepatic function, which are common in patients with cancer.
In patients with cancer receiving warfarin for VTE, the risk of bleeding and recurrent VTE remains high, even when the international normalised ratio (INR) is within the target range. As opposed to patients without malignancy, the bleeding rate is consistently high and is independent of the INR value in patients with cancer.18 At the time of recurrence, the INR value is within or above the target range in a higher proportion of cancer patients than in patients without cancer.16
In view of the poor results observed with the use of warfarin for the secondary prevention of VTE in patients with cancer, four randomised, controlled multicentre studies have compared the effects of LMWH with warfarin in patients with cancer and VTE (see Table 1). The first trial was an open-label, randomised comparison of warfarin and enoxaparin in which 146 patients with cancer and VTE were randomised to receive treatment with enoxaparin for three months, 1.5mg/kg once daily or warfarin, adjusted to achieve an INR of 2–3.19 n the warfarin group, all patients initially received LMWH at therapeutic dosage until an INR of at least 2 was achieved. During the three-month treatment period, 21.1% of patients in the warfarin group developed objectively confirmed symptomatic VTE and/or major bleeding compared with 10.5% in the enoxaparin group, although this difference was not statistically significant (p=0.09). During the same period, 22.7% of patients in the warfarin group died compared with 11.3% of patients receiving enoxaparin (p=0.07). Importantly, no atal bleeding was observed in the enoxaparin group, while six deaths due to bleeding occurred in the warfarin group (p=0.03).
The comparison of LMWH versus OAC for the prevention of recurrent VTE in patients with cancer (CLOT) trial was a large, international, open-label randomised study comparing dalteparin and OAC inpatients with VTE and cancer.20 All patients had symptomatic and confirmed VTE, and received dalteparin 200IU/kg once daily for one month, followed by approximately 150IU/kg once daily for five months (n=336). Patients allocated to OAC (warfarin or acenocoumarol) (n=336) initially received dalteparin 200IU/kg for five to seven days. In this group, OAC was started within 24 hours and adjusted to obtain an INR of 2.5 and continued for six months. The risk of objectively confirmed symptomatic recurrent VTE was significantly reduced in patients receiving dalteparin (8.8%) compared with patients receiving OAC (17.4%; p=0.002). The incidences of major bleeding and all bleeding complications were similar in both groups: 6 and 14%, respectively, in the dalteparin group, and 4 and 19%, respectively, in the OAC group (no significant differences).
The third study compared three treatments in 122 patients with VTE and cancer:
- enoxaparin 1mg/kg twice daily for five days, followed by 1mg/kg, once daily for six months;
- enoxaparin 1mg/kg twice daily for five days, followed by 1.5mg/kg, once daily for six months; and
- enoxaparin 1mg/kg twice daily for five days, followed by oral warfarin for six months.21
The primary objective of the trial was to evaluate the compliance with prolonged subcutaneous LMWH injection. Recurrent VTE was observed in one, one and two patients in groups (i), (ii) and (iii), respectively. There was also no difference in the incidence of bleeding. Interestingly, compliance was achieved in 97.9 and 97% of the patients allocated to he groups receiving a subcutaneous injection compared with 90.1% in the warfarin group. In the Long-term Innovations in TreatmEnt (LITE) programme, 200 patients with proximal deep vein thrombosis and cancer were allocated to tinzaparin, at a therapeutic dosage of 175IU/kg for three months, or to UFH followed by warfarin, adjusted to obtain an INR between 2 and 3.22 At the end of the three-month therapeutic period, physicians were allowed to stop anticoagulant treatment or to extend therapy with either LMWH or warfarin. At 90 days, 6% of the patients allocated to LMWH experienced recurrent VTE compared with 10.5% among the 100 patients who received warfarin, a non-significant difference. However, at 12 months the recurrent VTE rate was 7 and 16% in the two groups, respectively (absolute difference -9%, 95% CI -21.7 to -0.7; p=0.044). Major bleeding was observed in seven patients in each group.
The four studies above were reviewed in a meta-analysis, which reported a statistically significant and clinically important 50% reduction in the rate of recurrent VTE associated with prolonged LMWH administration in patients with cancer and VTE (6.5 versus 12.6%, relative risk [RR] 0.52, 95% CI 0.35–0.76; p=0.001).23 Conversely, no significant difference was observed between warfarin and LMWH for the risk of bleeding.22
Heparin-induced thrombocytopenia (HIT) may be a concern with the prolonged administration of LMWH in patients with cancer. Eight studies have now reported on prospective cohorts including a total of more than 900 cancer patients receiving prolonged treatment with LMWH, administered at therapeutic dosage, and no single episode of HIT was reported in those studies.19–22,24–27 However, thrombocytopenia is a common complication in cancer patients receiving chemotherapy and this may increase the risk of bleeding during LMWH treatment. For the use of dalteparin, Lee and co-workers suggested the reduction of the LMWH dosage during the period of thrombocytopenia, keeping to the standard dosage as long as the platelet count is above 100,000/mm3, to reduce the daily dosage by 3,000IU when the platelet count falls between 50,000 and 100,000/mm3, and to interrupt treatment for as long as the platelet count is below 50,000/mm3.20
The risk of drug accumulation has been addressed by a substudy of the CLOT trial.28 In this study, 24 patients receiving dalteparin 200IU/kg once daily had an anti-Xa measurement at weeks one and four. At week four, the mean anti-Xa level was 1.03 anti-Xa units/ml (95% CI 0.5–1.7) and did not differ significantly from the value obtained during week one.28
Renal impairment is a frequent co-morbid condition in patients having cancer. Renal function may be further impaired as a result of chemotherapy-related nephrotoxicity. In this context, LMWH accumulation may occur during prolonged treatment. Measurement of creatinine clearance is clearly needed before starting prolonged anticoagulant treatment using LMWH. Tinzaparin may be the most suitable option for patients with moderately impaired renal function. The results of a study completed in a group of patients with a wide distribution of creatinine clearance indicate that the anti-Xa effect of tinzaparin does not increase over 10 days of administration.29
Based on current data, the latest American College of Chest Physicians (ACCP) guidelines recommend that long-term anticoagulant therapy for patients with DVT and cancer should involve LMWH rather than VKAs, and should be continued for at least the first three to six months of long-term treatment.30 The recent American Society of Clinical Oncology (ASCO) recommendations also advise oncologists to use LMWH as initial and long-term treatment in cancer patients who experience VTE.12 The Effects of Low-molecular-weight Heparin Administration on Survival
Several studies have suggested a link between anticoagulant therapy and improved survival in patients with malignancy. One of the earliest studies was a prospective, randomised clinical trial that demonstrated that warfarin therapy was associated with significant prolongation of progression-free and overall survival in patients with small-cell lung cancer.31 However, in patients with other types of tumour, warfarin had no effect on survival. Furthermore, subsequent trials of VKAs in patients with cancer have provided conflicting results, and a systematic review concluded that there was insufficient evidence that long-term VKA administration provided a survival benefit in patients with cancer.32 Similar uncertainty surrounds the effect of UFH on survival.33
An analysis of patients with cancer who received LMWH for the initial treatment of acute deep vein thrombosis indicates that they may have prolonged survival compared with patients treated with UFH.15 More recently, a randomised clinical trial evaluated the effect of LMWH on outcomes in patients with small-cell lung cancer.34 In this study, both median progression-free survival and overall survival were significantly prolonged in patients who received LMWH in addition to chemotherapy compared with those who received chemotherapy alone. Furthermore, the improvement in survival was significant both in patients with extensive disease and in those with more limited disease.
In the Fragmin Advanced Malignancy Outcome Study (FAMOUS), a double-blind, placebo-controlled, multicentre trial, patients with advanced cancer received dalteparin 5,000IU once daily or placebo for 12 months in addition to the usual cancer treatment.35 Although LMWH administration was not associated with significantly improved survival at one year, post hoc analysis suggested that LMWH was associated with significantly increased median survival from 24.3 to 43.5 months in a subgroup of 102 patients with a good prognosis (i.e. surviving beyond 17 months from randomisation); however, this analysis was planned retrospectively, and the significance of these findings remains limited.
In the Malignancy and LMWH Therapy (MALT) trial, patients receiving the usual treatment for advanced cancer were randomised to receive either a full dose of nadroparin for two weeks followed by four weeks at half this dose or placebo.36 Median survival was 7.7 months in the nadroparin group versus 5.5 months in the placebo group (p=0.024). This beneficial effect was driven by the subgroup of patients who had a better prognosis at time of entry into the study.
Comparable findings were reported in a post hoc analysis of the CLOT trial. No difference in mortality was observed between patients with metastatic cancer receiving dalteparin or those receiving OAC. However, in the subgroup of 150 patients with non-metastatic solid tumours at randomisation, the cumulative mortality was significantly reduced in the dalteparin group (29%) compared with patients receiving OAC (35%; p=0.03).37 Conversely, Sideras et al. did not find any difference in the mortality rate of patients with advanced cancer who were randomised to receive dalteparin at prophylactic dosage or placebo in combination with the usual cancer care.38 These data are highly intriguing and warrant further investigation. Current data indicate that the improvement in survival associated with LMWHs may be independent of their anticoagulant effects, and may arise from antitumour effects of these agents, such as the inhibition of neoangiogenesis, inhibition of tumour-cell-derived heparanase activity, induction of apoptosis or inhibition of tumour cell adhesion to endothelial cells, whereas the effect of LMWH on tumour cell proliferation is debated.39 However, the clinical studies conducted so far have produced conflicting results, and evidence for prolonged survival in cancer patients with LMWH comes mainly from post hoc analyses or in studies conducted in patients with various types of malignancies. Additional studies conducted in patients with specific cancer types are needed to confirm these preliminary findings. Such studies are currently under way in several cancer types including lung, prostate, pancreas, ovary and stomach.
Conclusion
Patients with cancer are at high risk of thrombosis, and this is an important cause of avoidable mortality in these patients. These patients present unique challenges in the management of thrombotic risk that affect the safety and efficacy of antithrombotic therapies. Current data indicate that LMWHs offer several practical advantages for cancer patients, including a predictable anticoagulant effect, thus avoiding the need for regular laboratory monitoring, in addition to superior efficacy to OAC without increasing the risk of bleeding. Preliminary data suggest that LMWH administration may be associated with an increase in patient survival.