Pain Management Strategies
Common non-invasive strategies include oral, rectal and transdermal administration of analgesics, whereas intranasal and nebulised applications are not customary in Germany. Invasive and destructive measures such as chordotomy and neurolysis are now very rare in cancer pain therapy, and the indication and execution of these measures should therefore be limited to specialists. The most common invasive but non-destructive measures are intravenous and subcutaneous administration of drugs, while intramuscular application is rarely considered in cancer pain therapy. Spinal applications (epidural or intraspinal) are well established routes of administration but – compared with administration by mouth – rarely chosen strategies. In exceptional cases, intraventricular application of opioids may be indicated.
Oral Administration
In short, the above-mentioned factors indicate that physicians in the out-patient sector should make use of orally administered pain therapy whenever possible and should evaluate its advantages and disadvantages in relation to other methods, such as transdermal, rectal, intravenous, subcutaneous, epidural or intraspinal application of drugs. Furthermore, it is advisable that physicians select a few substances from the large portfolio of each substance group for use in pain control, to be able to gather extensive experience in their use.
Opioids for the Treatment of Moderate Pain
For the treatment of moderate pain, dihydrocodeine has been well established for several years, bearing in mind that it causes severe constipation and, frequently, nausea and vomiting. These side effects can be prevented by prophylactic treatment with laxatives and anti-emetics. As short-acting analgesics, immediate-release tramadol and tilidine are of no significant use for cancer pain control but, since slowrelease formulations of tramadol and tilidine were launched onto the market, tramadol has become the opioid of choice for the treatment of moderate pain. This is due to its analgesic effectiveness of eight to 12 hours and the low occurrence of side effects. Opioids for the Treatment of Severe Pain
Morphine
To this day, morphine is considered the reference opioid for the treatment of severe cancer pain with opioids. By international consensus, oral morphine is the standard by which all other opioid treatments for cancer are measured. It is well established worldwide, it has a predictable efficacy and its well known side effects can be targeted by prophylactic measures or are easily treated. Furthermore, morphine is produced in various immediate and slow-release formulations and has various routes of administration.fentamyl
Treatment with transdermal fentanyl is particularly indicated when the administration by mouth is not or is no longer possible, for example, in patients with tumours in the head and neck area, or if persistent nausea and vomiting are undermining the effectiveness of orally administered drugs. A fast-acting oral transmucosal delivery system for fentanyl for the management of breakthrough pain has recently come onto the market. Oral transmucosal fentanyl citrate (OTFC) consists of a sweetened matrix containing fentanyl that is attached to an applicator. When OTFC is placed in the mouth and sucked by the patient, the matrix dissolves and fentanyl is absorbed through the oral mucosa to provide fast-acting analgesia within no more than five minutes. OTFC is provided in six release rates: 200μg, 400μg, 600μg, 800μg, 1,200μg and 1,600μg fentanyl.
Dosage Titration
For the physician in the out-patient sector, the following refers to recommended procedure for the treatment of moderate to severe pain.
Moderate Pain
Treatment of moderate pain should start with 2 x 100mg controlled-release tramadol. If no sufficient pain relief can be achieved, titration should be carried out as follows:
3 x 100mg; 2 x 200mg; 3 x 200mg; and 2 x 400mg.
The ceiling effect of controlled-release tramadol is expected to occur at a dosage of approximately 800 mg to 1,000mg per day. Alternatively, morphine may be chosen for the initial treatment of moderate pain. If the patient is in good general health, the initial dose should be 2 x 30 mg controlled-release morphine tablets. Patients in poor general health should initially be given not more than 2 x 10 mg controlled-release morphine tablets per day. If pain relief continues to be insufficient after one day of such treatment, titration of the dosage is required. This should be performed in intervals of one or two days according to the following progression, until a satisfactory result is achieved and there are no side effects preventing titration:
10 – 20 – 30 – 40 – 50 – 60 – 70 – 80 – 100 – 120 – 140 – 160 – 180 – 200 – 220 – 250 – 280 – 300 – 330 – 360 – 400 – 430 – 460 – 500mg per day. Severe Pain
For the management of severe pain, a short-acting morphine preparation should be used initially. The recommended initial dose is 10mg. The efficacy of this dosage should be checked every two hours, thus allowing a titration after four hours, if required. If there is sufficient pain relief after administration of 10 mg, this dosage of short-acting morphine tablets or morphine drops should be given every four hours. If no sufficient pain relief is achieved, the morphine dosage should be increased as follows:
15 – 20 – 25 – 30 – 40 – 50 – 60 – 75 – 90 – 100 – 120 – 150 – 180 – 200mg.
Doses of 200mg every four hours amount to 1,200mg of morphine per day. In exceptional cases this requires further increments in the daily dosage, but it is recommended to discuss this with a colleague experienced in cancer pain control with morphine. If pain relief proves sufficient, the daily dosage of morphine is converted to controlled release tablets by the conversion factor 1:1. Hydromorphone is increasingly popular as an alternative to morphine. The initial dose should be 2 x 4mg per day, titration should be graded as follows:
3 x 4mg/day; 2 x 8mg/day; 2 x 12mg/day; 3 x 12mg/day; 2 x 24mg/day, etc.
Side Effects
The most common side effects of strong opioids are initial nausea and vomiting, as well as persistent constipation. Tiredness – another initial side effect – usually disappears after about five to seven days. As a prophylactic measure against nausea and vomiting, a course of haloperidol 3 x 0.5mg by mouth is offered. It is administered for the duration of seven days, because a tolerance against the emetic effects of morphine will normally develop.
Due to the constipation caused by opioids, patients receiving strong opioids will also be given laxatives from the beginning of their therapy. In patients who are still able to take sufficient liquids, macrogol is the drug of choice. Patients who are no longer able to do so, either due to the progression of their cancer disease or as a result of increasing weakness, are given natrium-picosulphate, often in combination with lubricant laxatives, such as liquid paraffin.
Along with non-opioids and opioids for cancer pain control, adjuvant substances such as glucocorticoids, anticonvulsants and antidepressants are good complementary therapeutic options, above all for the treatment of neuropathic pain.
Pain Treatment for Terminal Cancer Patients
Abstract
Overview
Strategies for the Treatment of Cancer Pain
Causal Pain Management
If the patient’s pain is the result of progression of the cancer disease, then surgical interventions, chemotherapy, hormone therapy, radioisotope or radiotherapy may be considered, provided they seem to be adequate causal or palliative therapeutic options. Minimising or even removing a tumour will normally lead to a decrease in the pain intensity. In the advanced stages of cancer, the benefit of such measures must be carefully weighed against possible complications and burdens on the patient. Palliative care does not necessarily preclude such therapeutic approaches. The expected benefits of such measures, however, must be greater than the potential disadvantages. Of these therapies, radiotherapy is considered a rather important aspect of palliative medicine. Whenever cancer patients are suffering from pain, an adequate analgesic therapy is required, irrespective of the progressive stage of the disease.
Article
Further Resources

Trending Topic
Chemotherapy-induced myelosuppression has long been considered a toxicity that limits the dose of cytotoxic agents, which contributes to significant morbidity and mortality.1 Sub-optimal dose intensity may decrease the efficacy of therapy, thereby compromising progression-free survival (PFS) and overall survival (OS) benefits.2 Chemotherapy-induced myelosuppression is often managed with granulocyte-colony stimulating factors (G-CSFs) and erythropoiesis-stimulating agents (ESAs) […]
Related Content in Supportive Cancer Care

Welcome to the latest edition of touchREVIEWS in Oncology & Haematology. Despite the success of the human papillomavirus vaccination programme, cervical cancer remains one of the most common cancers in women, and advanced disease carries a poor prognosis. Lebreton et ...

Welcome to the winter edition of touchREVIEWS in Oncology & Haematology. Despite the continued impact of the COVID-19 pandemic on cancer care, 2021 has seen important advances in many areas of oncology, which is reflected in our diverse range of articles. ...

Welcome to the inaugral issue of touchREVIEWS in Oncology & Haematology (formerly Oncology & Hematology Review [US]), which aims to summarize the latest clinical data and provide practical advice to help busy physicians in their daily practice. As co-editors-in-chief of ...

Cancer is a growing problem that demands the best from all sectors of the global health workforce.1 Cancer nurses make up the largest proportion of this workforce and, as well as being responsible for direct care delivery, also aim to ...

Biosimilars, which are copies of patent-expired large-molecule biologic drugs (Table 1),1–8 offer the same rationale as generic drugs. They can give patients, hospitals and healthcare systems the opportunity to expand patient access to care and offer potential budget savings to reinvest (...
Forty percent of Americans will be diagnosed with cancer in their lifetimes.1 Due to continued advances in cancer detection and treatment, there are an unprecedented number of cancer survivors. In fact, the 5-year survival rate for all cancers has increased ...

Despite the increasing availability of precision medicine and the recent introduction of immune checkpoint inhibitors in the treatment of many tumours, chemotherapy remains the standard of care for most patients with cancer. Chemotherapy-induced nausea and vomiting (CINV) is highly prevalent ...

Dear Colleagues, European Oncology & Haematology is now accepting submissions to the Summer and Winter 2018 editions. European Oncology & Haematology aims to publish topical articles, which are chosen for their evaluation of current practices and research that directly affect practitioners ...

Dear Colleagues Oncology & Hematology Review (US) is now accepting submissions to the Spring and Fall 2018 editions. Oncology & Hematology Review (US) aims to provide practicing oncologists and hematologists with concise and timely articles that provide practical advice relevant to ...

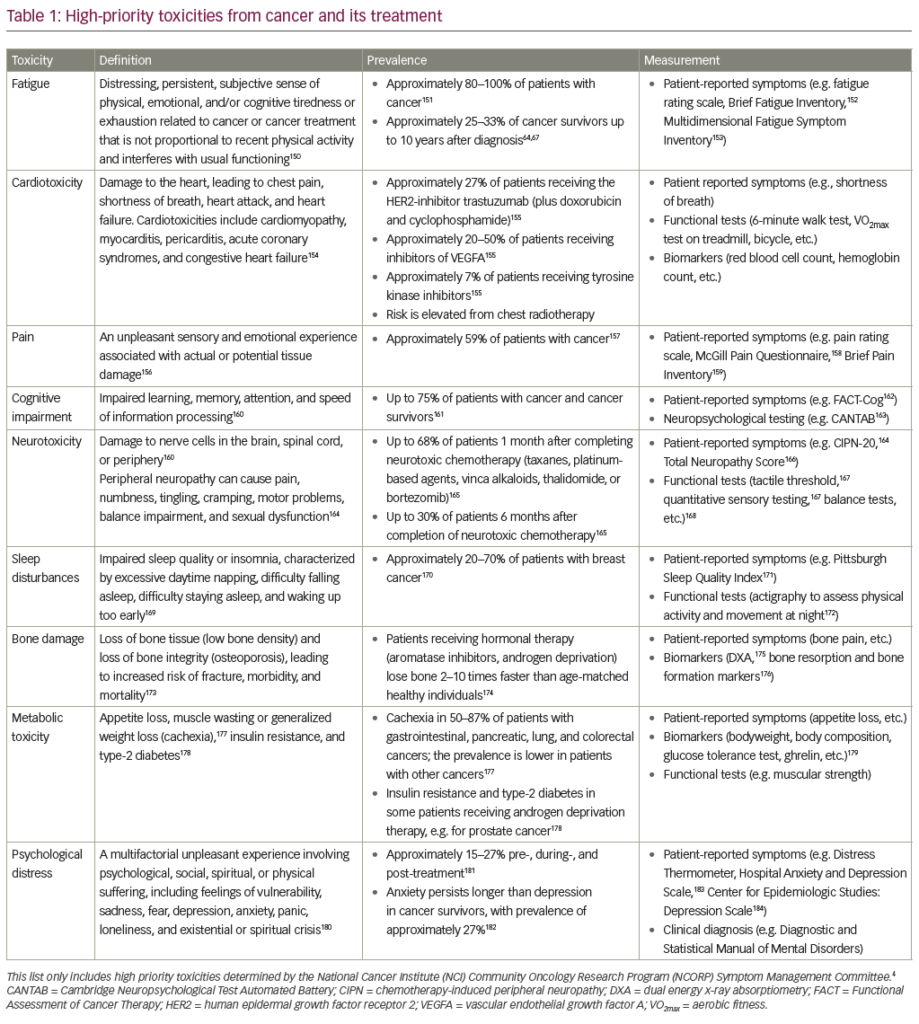
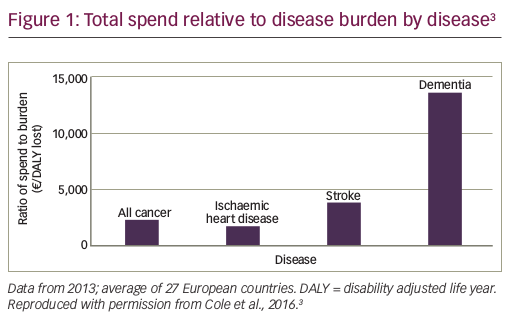
The estimated incidence of cancer increased by almost one third across Europe between 1995 and 2012, and this growth is predicted to continue.1,2 Compared with other diseases and disabilities, the lost years of life and productivity resulting from cancer represent a heavy ...
Innovation in healthcare is highly beneficial from a patient perspective; however, the introduction of new drugs, often at high cost, is placing increasing pressure on many healthcare systems.1 As a result, these systems are facing a growing crisis of affordability, ...
Latest articles videos and clinical updates - straight to your inbox
Log into your Touch Account
Earn and track your CME credits on the go, save articles for later, and follow the latest congress coverage.
Register now for FREE Access
Register for free to hear about the latest expert-led education, peer-reviewed articles, conference highlights, and innovative CME activities.
Sign up with an Email
Or use a Social Account.
This Functionality is for
Members Only
Explore the latest in medical education and stay current in your field. Create a free account to track your learning.

