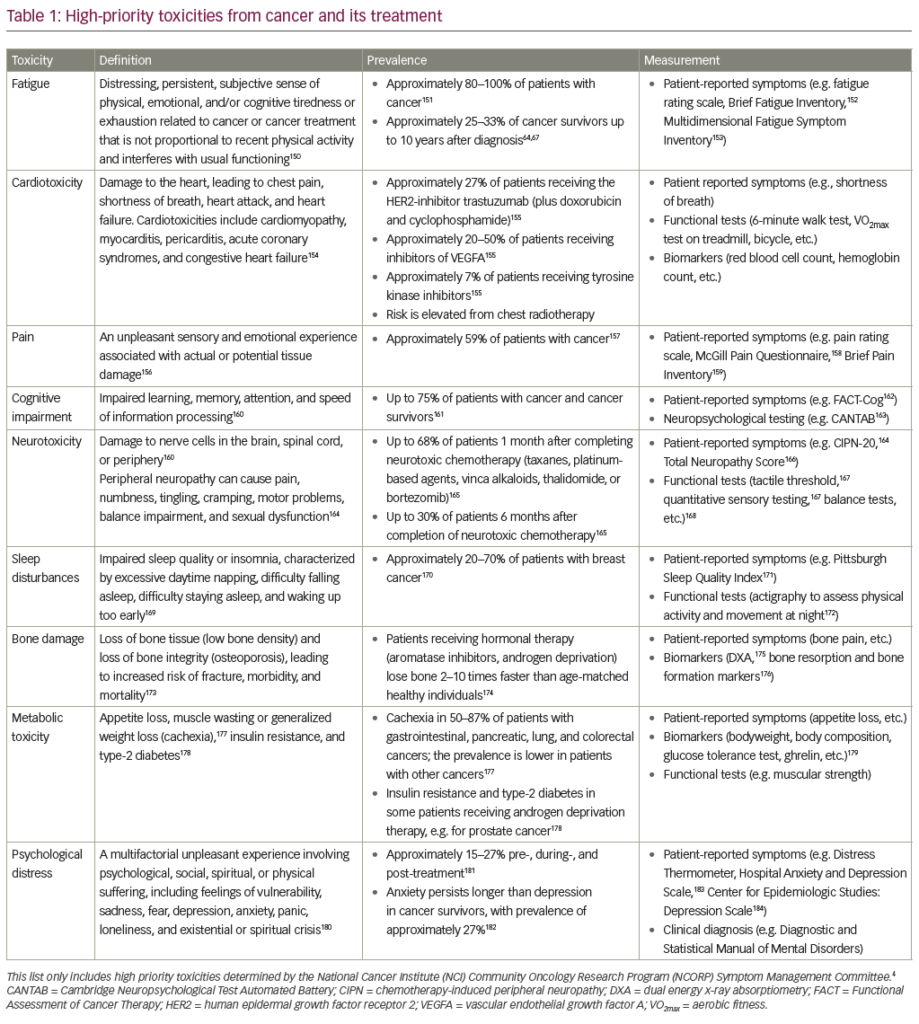
Antineoplastic therapy is frequently complicated by the development of cardiotoxicity, either as the direct effects of chemotherapy on the heart, such as left ventricular dysfunction (LVD), heart failure (HF) or ischaemia, or as indirect effects due to haemodynamic changes, thrombotic events or hypertension.1 Simultaneously, with the improvement in the management of cancer patients and prolonged survival, cardiotoxicity has become increasingly important, as the adverse effects on the heart may create new challenges in the long-term management of the patients, reducing the effectiveness of treatment and survival, due to the appearance of comorbidities.
Chemotherapy-Related Left Ventricular
Dysfunction and Heart Failure
Over the last 20 years, considerable progress has been made in the management of cancer due to the introduction of targeted molecular therapies.2 However, even these new target drugs have been shown to have a cardiotoxic potential, especially when used as combination therapies, by inhibition of transduction pathways in normal or non-cancerous cells.3
Monoclonal Antibodies
Trastuzumab is a humanised monoclonal antibody against the human epidermal growth factor receptor (HER2), a tyrosine kinase receptor overexpressed at the time of diagnosis in 15 % of breast cancer (BC) and up to 20–30 % of metastatic BC,4 which is associated with more aggressive disease, poor response to therapy and survival. Trastuzumab is used to treat BC cells that overexpress HER2, increasing the overall survival, but at the same time, the risk of cardiotoxicity.5,6 A 2001 pivotal study reported a gain of survival of 4.8 months when trastuzumab is administered with anthracycline; however, 27 % of patients who received the combination therapy developed signs of HF (16 % were in New York Heart Association [NYHA] class III–IV).4 Pooled data on the use of trastuzumab have reported an incidence of LVD of 3–7 % (trastuzumab alone), up to 27 % when administered with anthracyclines;5,7 while adjuvant trastuzumab after anthracyclines and paclitaxel is associated with an incidence of 4–18.6 % for LVD and 1.6–3.3 % for HF.6,8,9 Analysis of more than 45,000 cases of early-stage BC treated in woman >65 years old has shown that patients who received both trastuzumab and anthracycline had an absolute 23.8 % higher rate, and those treated with anthracycline chemotherapy alone has an absolute 2.1 % higher rate of HF or cardiomyopathy (CM) events over 3 years compared with patients who received no adjuvant chemotherapy or trastuzumab.10 Use of trastuzumab was associated with an absolute 14 % higher adjusted incidence rate for HF or CM over 3 years.10 Furthermore, among BC survivors >65 years old, 29.2 % were observed to have preexisting cardiovascular diseases (CVDs) or diabetes at the time of diagnosis.11
The risk factors described for the development of trastuzumab-induced cardiotoxicity include age >50 years, borderline left ventricular ejection fraction (LVEF) before treatment, history of CVD, cardiovascular risk factors (CVRFs) such as diabetes, dislipidaemia or elevated body mass index (>30), prior treatment with anthracyclines12–14 and, possibly, genetic background and immune status.15
There are some differences between anthracycline and trastuzumab cardiotoxicity: the first is irreversible, dose-related and associated with ultrastructural changes at the endomyocardial biopsy, while the second is reversible, responsive to medical therapy and does not appear to be dose related.16,17
What are the causes of trastuzumab’s cardiotoxicity, especially in coadministration with anthracyclines? HER2 plays an important role in embryonic development of the heart and it forms heterodimers with HER4 after the union with neuroregulin 1 (a ligand of HER4 expressed by endothelial cells).18 The heterodimerisation of HER2–HER4 leads to activation of different pathways, such as Src-FAK, which increases the cell–cell contact, or phosphoinositide 3 kinases – protein kinase B (PI3K-Akt) and mitogen-activated protein kinase (MAPK) – which promotes the proliferation of cardiomyocytes, their survival, contractile function, angiogenesis and development of the conduction system.19,20
The HER–neuregulin 1 axis is a critical component of the response of cardiac tissue to stress. When the heart is exposed to anthracyclines, pathways that lead to cardiomyocytes death are activated.21 Signalling from the HER2–HER4 dimerisation are activated as a counter-measure, but they are blocked by trastuzumab. In view of these circumstances, there is a growing possibility of using recombinant neuregulin-1 in severe HF, which use has demonstrated to increase the LVEF of 12 %.22 Further investigations are required to determine safety.
Given these assumptions, the administration of trastuzumab after anthracyclines may be related to an increased risk of heart damage compared with the opposite sequence. Clinical evidence to support this hypothesis comes from the comparison of the 0 % incidence of HF in a Finnish trial with only 9 weeks of herceptin (FinHER) (in which trastuzumab preceded anthracyclines) and 3–4 % incidence of HF in trials where trastuzumab was given after.5,23,24
Bevacizumab is a humanised monoclonal antibody against vascular endothelial growth factor (VEGF). The uncontrolled hypertension (discussed below) and the inhibition of VEGF signalling, both able to alter the adaptive responses of the heart, seem to be potential mechanisms able to explain its cardiotoxicity. Bevacizumab-related HF ranges from 1.7 % to 3 %1; a recent meta-analysis finds in metastatic BC patients an overall incidence of high-grade HF of 1.6 % (relative risk [RR]=4.74).25
Small Molecule Tyrosine Kinase Inhibitors
Lapatinib is an inhibitor of the epidermal growth factor receptor, and has been shown to produce a low rate of cardiotoxicity, with 1.4 % of asymptomatic cardiac events and 0.2 % of symptomatic HF (0.3 % in patients pre-treated with anthracyclines and 0.1 % in those pre-treated with chemotherapy and trastuzumab); the mean time of onset was 13 weeks.26 Differences in the pharmacokinetics of duration or degree of inhibition of HER2 by lapatinib compared with trastuzumab may partly explain the different incidence of HF induced by the two drugs.15
A retrospective review of six trials showed an incidence of imatinibrelated HF of 0.5 %.27 Imatinib leads to cardiomyocyte death through a stress response of the endoplasmic reticulum in which an accumulation of misfolded proteins induces apoptosis, mediated by the c-Jun N-terminal kinase (JNK).28,29
Retrospective analysis of patients treated with sunitinib showed an incidence of HF from 2.7 % to 8 %.30,31 No change in LVEF has been reported in a trial of patients with advanced gastrointestinal stromal tumour (GIST) treated with sunitinib for 8 weeks.32
Schmidinger et al.33 estimate an incidence of LVEF drop of 5 % for sorafenib and 14 % for sunitinib. Particular attention should be paid when patients are sequentially treated with those two drugs, as additive cardiotoxicity has been reported.34 Sunitinib induces an intracellular signalling cascade that releases the pro-apoptotic factor BCL2. This could induce the activation of the intrinsic apoptotic pathway, adenosine triphosphate (ATP ) depletion and myocyte that could lead to LVD.15 In patients with leukaemia treated with dasatinib, 2 % to 4 % in developed HF.1 Desatinib cardiotoxicity appears to be due to inhibition of ABL and SRC.35
Chemotherapy-Related Myocardial Ischaemia/ Tromboembolism
Monoclonal Antibodies
Bevacizumab is associated with an increased risk of arterial thromboembolism (AT E), and its safety has been called into question: a pooled analysis of randomised controlled trials36that evaluated combination treatment with bevacizumab versus chemotherapy alone in patients with metastatic colorectal, breast and non-small-cell lung cancer, shows an increased risk of AT E (hazard ratio (HR)=2.0; 95 % confidence interval [CI]) in the bevacizumab group (most of them were myocardial or cerebrovascular events), which can occur at any time during therapy, but in most cases after 3 months. Proposed risk factors include age >65 years old and a previous history of AT E. A meta-analysis by Ranpura et al. assesses an incidence of all-and high-grade AT E of 3.3 and 2 %, respectively, with a RR of 1.46 for fatal adverse events in combination therapy with bevacizumab compared with chemotherapy alone.37 Looking at venous thromboembolism, a systematic meta-analysis of 15 trials38 found rates of all-and high-grade VTE of 11.8 and 6.3 %, respectively.
The proposed mechanism for the development of ischaemic phenomena is a decrease in the regeneration process of endothelial cells during anti-VEGF treatment, through a loss of integrity of the endothelium, which allow the exposure of subendothelial collagen, activation of tissue factor and, therefore, of the coagulation cascade.39,40
Small Molecule Tyrosine Kinase Inhibitors
A higher frequency of AT E have been found in patients treated with sorafenib for metastatic renal cancer (3 %)41 compared with placebo. Choueiri et al.42 find an incidence of 1.3 and 1.7 %, respectively, with no statistically significant difference.
Chemotherapy-related Hypertension
Treatment of cancer patients with inhibitors of angiogenesis, both antibodies or tyrosine kinase inhibitors (TKIs), is commonly associated with systemic arterial hypertension, with potential organ damage.1,43,44
Hypertension is a known complication of bevacizumab (VEGF inhibitor) with 4 % to 35 % of patients with raised blood pressure (BP) during treatment and 11–18 % of patients with grade 3 hypertension,1 of which 11–16 % require de novo treatment with antihypertensive medication, and 1 % that develop a hypertensive crisis.44 Ranpura et al. 45 found high-grade hypertension in 7.9 % of patients, without significant differences between high and low dose.
For sutinitib, incidences of all-and high-grade hypertension are 21.6 and 6.8 %, respectively, with relative risks of 3.44 and 22.72, respectively.46 However, studies and meta-analysis used different diagnostic criteria, with a large variability of results. For example, a retrospective analysis on sunitinib30 found that 47 % of patients developed hypertension, defined as BP >150/100 mmHg. Sorafenib is widely associated with this adverse effect (17 % to 43 % of patients in clinical trials), often severe (grade 3 and 4 occurs in 1.4 % to 38 %).1
Several theories have been put forward: the increased arterial resistance can be explained by the inhibition of VEGF, with a decrease of the nitrous oxide (NO)-synthase activity in the arterioles and loss of the contribution of this vasodilating molecule and/or the reduction of the arteriolar and capillary density.47,48 In this regard, Mourad et al.49 have demonstrated that hypertension induced by bevacizumab was associated with capillary rarefaction measured by capillaroscopy. Veronese et al.50 found that hypertension induced by sorafenib is mainly due to an increase in vascular rigidity.
Chemotherapy-related QT Interval Prolongation
The main pro-arrhythmic adverse effect induced by anti-cancer therapy is the prolongation of the QT interval, which is a measure of the total duration of ventricular depolarisation and repolarisation, from the beginning of the QRS complex to the end of the T wave, the duration of which is affected by the heart rate.
Multitargeted Tyrosine Kinase Inhibitors
Prolongation of QTc (QTP) >60 msec was found in, respectively, 1.9 % and 2.5 % of patients with chronic- and accelerated-phase chronic myelogenous leukaemia treated with nilotinib,51 while in another phase II trial, the percentage rose to 4 % (no patient had QTc >500 msec).52 A phase I/II trial reported five cases of sudden cardiac death (SCD) probably or potentially related to nilotinib.53 A phase II study of dasatinib showed an increase >60 msec of QT interval in 3 % of patients and <1 % had a QTc >500 msec.52
Sunitinib is associated with various effects on electrocardiogram (ECG), such as bradycardia, increased PR and QT interval and changes in the ST segment and T wave.30 However, Torsades de Pointes (TdP) has been observed in <0.1 % of patients. The effect of sutinib on the QT interval is dose dependent.33
Vandetanib is a selective inhibitor of VEGFR, EGFR and (receptor) RET tyrosine kinase that interacts with ion channels leading to repolarisation abnormalities and QTP.54 Analysing phase II trials, vandetanib showed a QTP in 20.6 % of patients treated55and 11.4 % in another trial in combination with docetaxel.56
Evaluation of Patients before Treatment with Anticancer Drugs and Identification of High-risk Populations
All patients who are undergoing chemotherapy with drugs known to induce cardiotoxicity should receive an initial clinical evaluation and assessment of comorbidities and CVRF, such as hypertension, diabetes, dyslipidaemia or electrolyte disturbances that should be corrected before initiating anti-cancer therapy and monitored during the same.1,57,58 As seen above, some antineoplastic drugs are responsible for a lengthening of the QTc interval, a substrate for malignant ventricular arrhythmias. These patients should be evaluated for the identification of correctable risk factors known to induce QTP, and an ECG should be performed periodically.
Drug-related cardiotoxicity has been defined by the Cardiac Review and Evaluation Committee supervising trastuzumab clinical trials as one or more of the following features:7,58
• cardiomyopathy in terms of a reduction in LVEF, either global or more severe in the septum;
• symptoms associated with HF;
• signs associated with HF (such as S3 gallop, tachycardia); and
• decline in LVEF of at least 5 % to less than 55 % with accompanying signs or symptoms of HF, or a decline in LVEF of at least 10 % to <55 % without accompanying signs or symptoms of HF.
However, recent definitions have varied and include a greater change in LVEF below the lower limit of normal (LLN) or LVEF <50 %, and represents the upper limit of normality recognised by different authors in recent years.24,58,59,60
Monitoring and Management of Cardiotoxicity During and After Therapy
Management of Hypertension
It is recommended to perform a careful cardiologic evaluation, including monitoring of BP before treatment with VEGF inhibitors, and renal function assessment, as a glomerular disease may be the cause of worsening hypertension. This evaluation should not exclude patients from therapy, but enable to plan a monitoring and early treatment. The strategy, in accordance with the current guidelines endorsed by the European Society of Cardiology (ESC), is to maintain BP <140/90 mmHg (<130/80 in patients with diabetes or renal disease), choosing the most appropriate drug on patient characteristics and comorbidities. Antihypertensive therapy includes the use of angiotensin converting enzyme inhibitors (ACE-In), to use as first-line therapy61, and beta blockers. Non-dihydropyridine calcium channel blockers should be avoided, as they are able to interfere with cytochrome P450 and CYP3A462, of which the antiangiogenic drugs are a substrate. Regular monitoring is recommended weekly during the first cycle of therapy, and then at least every 2 or 3 weeks for the duration of drug regimen. Discontinuation must be considered if systolic BP is >200 mmHg or diastolic BP >100 mmHg. At earlier stages, discontinuation remains controversial, as grade 2 and 3 hypertension seems to be associated with a good response to therapy63 and it is important to allow continuous and safe administration of VEGFtarget drugs without dose modification. Treatment with bevacizumab has been associated with proteinuria in patients who develop hypertension (up to 41–63 %),64 with a causal and dose-dependent relationship.
Management of Myocardial Ischaemia
The diagnosis and management of acute coronary syndrome (ACS) is based on clinical presentation, ECG changes and elevation of myocardial necrosis enzymes, in the manner indicated by the recent ESC guidelines. The treatment of ACS provides the use of percutaneous coronary intervention or thrombolysis and the administration of antiplatelet and anticoagulants agents, all of which may be a problem (or contraindicated) in cancer patients with thrombocytopenia, recent surgery or brain metastases.1 The use of stents (especially drug-coated stents) may be limited in cancer patients due to the need for longterm use of clopidogrel and aspirin, which may hamper administration of anticancer therapy.39 At the moment there are no specific recommendations. However, a retrospective study on aspirin and beta blockers in patients with cancer and ACS showed an improved 7-day survival rate without increasing of the bleeding risk.65
Management of QT Prolongation
QT prolongation is a commune-adverse effect of many drugs (see www. qtdrugs.org) such as antidepressants, antibiotics, antifungals, serotonin agonist/antagonists, antipsychotics, antiarrhythmics, anti-emetics), and is also associated with several risk factors that should be carefully investigated and, if possible, removed, such as baseline QT prolongation, subclinical QT syndrome, female gender, diabetes, cirrhosis, myocardial ischaemia or hypertrophy, HF, myocarditis, bradycardia, hypokalaemia, hypomagnesaemia, hypocalcaemia, hypothyroidism, hyperparathyroidism, hyperaldosteronism, subarachnoid haemorrhage, stroke and intracranial trauma.
An ECG should be performed in order to monitor the duration of the QT interval at baseline, one week after the beginning of treatment and periodically at each dose adjustment. Close monitoring and adjustment of electrolytes is necessary before starting and during treatment. The QT should be measured and corrected (QTc) for heart rate according to Bazett formula (QT/RR1/2) or Fridericia (QT/RR1/3). The definition of prolonged QTc is quite variable in the literature, but many authors consider a normal QTc ≤400 ms and prolonged ≥450 ms in men and ≥470 ms in women. Ventricular arrhythmias, in particular TdP, are associated with QTc ≥550 ms.66
In cases of TdP, the offending drug should be discontinued and the patient should be monitored in an intensive care unit. Initial therapy consists of intravenous administration of 2 g of magnesium sulphate. Unsynchronised defibrillation may be indicated in cases of ventricular fibrillation or haemodynamically unstable polymorphic ventricular tachycardia. Another approach is an overdrive pacing to reduce the QTc interval, especially when the TdP is precipitated by bradycardia. Isoproterenol titrated to a heart rate ≥90 beats per minute is another option when the temporary stimulation is not available.1
Management of Heart Failure
Echocardiography
A Doppler ECG (DEcho) evaluation should be performed before, during and after treatment with drugs known to induce LVD and HF, in order to identify early signs of ventricular dysfunction.60,67
Among all imaging modalities, DEcho offers several advantages, in that it allows an assessment of systolic and diastolic function, valve disease, pericarditis, pericardial effusion and carotid lesions.
The most-common methods of cardiac monitoring of LVEF are echocardiography, multigated acquisition (MUGA) scan or cardiac magnetic resonance imaging (MRI); however, the last two methods, first, expose the patient to radiation or, second, are associated with high costs. Echocardiography provides a wide spectrum of information on cardiac morphology and function. However, LVEF measurement may underestimate the early heart damage1,57 so, as a consequence, we must take into account recent techniques, such as 2D speckle tracking echocardiography (2DSTE) or tissue doppler imaging (TDI).
A seriated echocardiogram study in patients with BC treated with anthracyclines and subsequently with trastuzumab68 shows that 10 % of patients experienced a decline in LVEF >10 %, yet still more than 50 %. However, longitudinal 2DSTE decreased significantly at 3 months and systolic strain rate by TDI decreased at 6 months, suggesting evidence of occult LVD associated with therapy with trastuzumab despite preserved LVEF, proving that these two techniques are more sensitive in detecting occult LVD.69 For further evidence, a recent prospective study by Fallah- Rad et al.,70 showed that 25 % of patients treated in the same way who developed cardiotoxicity (decrease in LVEF >10 % or a value <55 % with signs or symptoms of HF) showed LVEF variations at 6 months after therapy with trastuzumab; however, after 3 months the peak systolic velocity of the side wall (S’) to the TDI and global peak longitudinal and radial strain by 2DSTE had already declined, proving indices of early systolic dysfunction. Systolic and diastolic function should be assessed at baseline, before and during treatment with monoclonal antibodies or anthracyclines. Further evaluations, through conventional methods, TDI and 2DSTE are recommended, even in asymptomatic patients. Patients with LVD should be evaluated by coronary angiography or dobutamine stress echocardiography to exclude coronary artery disease, which could lead to specific treatment.1
Biomarkers in Evaluation of Cardiotoxicity
Troponin is the gold standard for detection of myocardial ischaemia and damage: increased levels has been observed in patients treated with standard dose of anthracyclines and with the newly targeted therapies. Detemination of troponin levels can be useful to identify patients at risk for HF and to discriminate between reversible and irreversible cardiac damage in BC survivors treated with trastuzumab.71 Periodic assessment of troponin levels during high-dose chemotherapy allows the identification of patients at risk of cardiac dysfunction and cardiac risk stratification, proving as an early predictor of LVD and subsequent reduction in LVEF.72 Sawaya et al.73 have combined biomarkers with echocardiography in a multicentre study of patients treated with anthracyclines and trastuzumab, which showed that a decrease in longitudinal and radial strain and the elevation of troponin at 3 months is predictor of reduction in LVEF at 6 months.
B-type natriuretic peptide (BNP) is a cardiac hormone released by the myocardium in response to volume overload, so increased levels of BNP reflects an augmentation of the filling pressure and LVD. A useful approach, but still controversial, is performing the basic assessment of these biomarkers and periodic measurements during therapy to identify patients who require further cardiac evaluation.58
Management/Treatment Recommendations
Patient undergoing chemotherapy known to induce cardiotoxicity should have a careful clinical assessment of CVRF and comorbidities and those that are reversible should be suitably treated before the start of chemotherapy.58,74 Patients should also be evaluated for the presence of risk factors associated with the development of target drug-induced cardiotoxicity. Drugs associated with a high risk of HF should be avoided in patients with pre-existing LVD and used with caution in those with cardiovascular comorbidities.75
Summarising the evidence described above, the LVEF is an index of cardiac function widely used in clinical practice and in experimental studies. However, other echocardiographic techniques have been shown to be able to identify early signs of cardiotoxicity and should be used, when possible, in the monitoring of the patient at risk of developing LVD and HF. A DEcho evaluation of the parameters of systolic and diastolic function at baseline is necessary to stratify the individual risk for developing cardiotoxicity in these patients and as a basis of comparison for future monitoring.
It is recommended to perform a serial monitoring of cardiac function in patients receiving anthracyclines and/or trastuzumab in the adjuvant setting at baseline, 3, 6 and 9 months during treatment, and then at 12 and 18 months.58,76
Patients who develop LVD with LVEF <50 % or a decline of >10 % should discontinue anti-cancer therapy and must be treated at first-line with an ACE-In and a beta-blocker.77,78 A re-evaluation should be performed after 3 weeks, assessing the potential risks and benefits in terms of continuation of treatment. Patients who experience cardiotoxicity while receiving trastuzumab generally recover their cardiac function with discontinuation at 1.5 months, with well tolerated re-challenge.1,16 ACEIn have been tested in a randomised trial of patients after high-dose chemotherapy. An early and intensive treatment of these patients, even asymptomatic, is necessary, especially if the tumour might have a long-term survival. The earlier the therapy, the better is the therapeutic response.79 Treatment with monoclonal antibodies can be recovered in asymptomatic patients between 40 to 50 %.58,60 Patients with symptomatic HF and/or persistent LVEF <40 % should stop chemotherapy and treated according to guidelines. If fluid retention is present, patients should initially be treated with intravenous diuretics and nitrates. ACE-In should be administered, followed by beta blockers after 3 to 4 weeks, initiated at a low dose. The goal is to reach the highest tolerated dose. Renal function, serum potassium concentration, BP and signs of HF should be carefully monitored during titration of ACE-In and beta blockers. Aldosterone receptor antagonists are indicated in addition in NYHA III or IV patients.60
Conclusion
Taken together, these data suggest that a careful cardiovascular examination and management could not only prevent adverse events from antineoplastic drugs, but could also be used to exclude their use in individuals at high risk. The choice of the therapeutic approach and the prognosis depends on the delicate balance between the need for an optimised anti-cancer therapy and an improved management of cardiovascular complications, in order to avoid that the gain in survival offered by new therapies is offset by the emergence of new complications related to the therapy itself.