In 2008, primary kidney malignancies accounted for a predicted 4% of all cancer diagnoses in the US. There were an estimated 54,390 new diagnoses of renal cell carcinoma (RCC), which constitute approximately 90% of all malignant kidney tumors (approximately 85% clear-cell histology), and 13,010 kidney cancer deaths.1 The Surveillance, Epidemiology and End Results (SEER) data from 2005 reported five-year survival rates of 89% for localized resected disease, 61% for regionally advanced disease, and 9.5% for metastatic disease. The age-adjusted SEER incidence rates have nearly doubled from 7.1 per 100,000 population in 1975 to 13.4 per 100,000 in 2005. The economic burden of RCC in the US in 2004 was an estimated $4.4 billion in healthcare costs and lost productivity.2 Surgery remains the curative standard of treatment for localized disease; however, up to 50% of patients will relapse.3 Approximately 30% of new diagnoses are metastatic and the median overall survival (OS) for metastatic disease is reported as 10–12 months.4 Due to high relapse rates and limited survival with metastatic disease there has been considerable focus on developing systemic therapies to improve these outcomes.
Adjuvant Treatment
Several adjuvant treatment modalities have been investigated. Radiotherapy post-radical nephrectomy is not beneficial in controlling local recurrence or development of metastatic disease5 or improving survival rates.6 Attention also focused on adjuvant cytokine therapy in high-risk patients. Adjuvant interferon-alfa (IFN-α) did not demonstrate a median OS advantage over observation alone in the phase III setting (OS 5.1 versus 7.4 years; p=0.09).7 Similarly, adjuvant high-dose interleukin-2 (IL-2) did not confer a relapse rate advantage at two years over observation alone (76 versus 65%; p=0.73).8 Other adjuvant treatments assessed for their efficacy have included medroxyprogesterone acetate (MPA), bacille Calmette-Guérin (BCG), or autologous tumor vaccine (ATV). Neither MPA9 nor BCG10 demonstrated any survival advantage over observation. A small progression-free survival (PFS) benefit was seen for ATV over observation (77 versus 68%; p=0.02), but OS was not reported.11
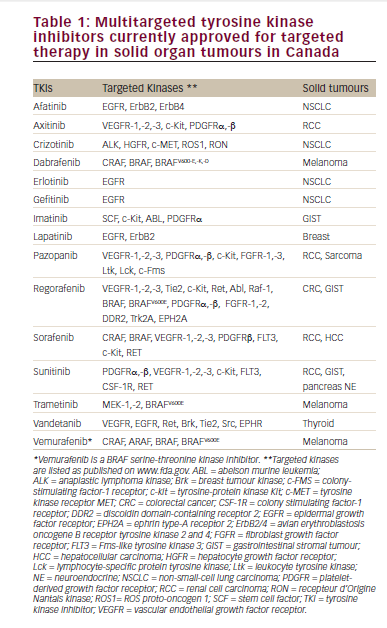
There has been significant recent interest in targeted agents on the basis of their efficacy in advanced disease. The two multikinase inhibitors sunitinib and sorafenib are currently being investigated in the ongoing phase III trials STAR (sunitinib versus placebo), SORCE (sorafenib versus placebo), and ASSURE (sorafenib versus sunitinib versus placebo). Their role in neoadjuvant therapy is not yet clear. Current literature does not demonstrate any definitive benefit for adjuvant therapy after curative resection, but ongoing trials with newer targeted agents continue.
Advanced Disease
Cytokine Therapy
Observed spontaneous remissions in RCC led to the identification of the potential importance of the host immune response and the development of cytokine therapies. IFN-α provided a small but significant benefit in response and survival in metastatic RCC, summarized in the Cochrane analysis of 6,880 patients in 58 studies.12 An IFN-α dose of 8–18 megaunits (MU) three weekly in five studies was compared with non immunotherapy controls. The pooled overall response rate (ORR) was 12.5 versus 1.5% for controls (pooled odds ratio 7.6, 95% confidence interval [CI] 3.0–19.2), with a reduced one-year mortality (hazard ratio [HR] 0.56, 95% CI 0.40–0.77). IFN-α conferred a median survival advantage of 3.8 months over the control subjects (11.4 versus 7.6 months). Further pooled retrospective analyses of 463 patients from six prospective trials suggested a similar benefit from first-line IFN-α , reporting a median OS of 13 months, a median time to progression (TTP) of 4.7 months, and a five-year survival rate of 10%.13 These trials established IFN-α as a suitable control for future studies. The minimum effective dose is 20–40MU weekly. Higher doses result in greater toxicity without higher response rates.14 The toxicities include a flu-like syndrome of fatigue, fever, and myalgia, as well as nausea and depression. Historical attempts to combine IFN-α with other agents have not led to the development of clinically useful regimens.15
High-dose bolus IL-2 regimens can provide durable responses in advanced RCC. Reported response rates vary from 14 to 23%16,17 and median duration of partial response (PR) from 11 to 19 months,18,19 with approximately 6% achieving durable complete radiological responses lasting up to 148 months.19 However, this is achieved at the expense of significant toxicity, including hypotension, arrhythmias, vascular leak syndrome, and 4% treatment-related mortality.18 Efforts to combine IL-2 with other therapies including IFN-α and fluorouracil in order to reduce toxicity have not been successful. A pooled analysis of these regimens compared with non-IL-2-containing controls has confirmed that they convey higher toxicity yet no significant OS benefit.20 Despite the 5–7% durable complete response (CR) rate of high-dose IL-2, excess toxicity restricts its use to patients of good performance status and limits its availability. One retrospective study has reported that high expression levels of carbonic anhydrase IX (CAIX) correlate with the response rate.21 Currently, the high dose IL-2 SELECT trial is recruiting to validate the predictive value of CAIX in a prospective setting. Targeted Agents
The von Hippel Lindau (VHL) gene is a tumor suppressor gene found to have mutated in 70–80% of sporadic clear-cell RCC. In normal oxygen tensions, wild-type VHL gene product binds and degrades hypoxiainducible factors (HIFs) 1α and 2α. In hypoxic conditions or absent VHL gene product, HIF-1α accumulates and leads to the production of various growth factors, including transforming growth factor-α (TGF-α), platelet-derived growth factor (PDGF), and vascular endothelial growth factor (VEGF). Investigators have focused on targeting this pathway, which is important for cellular proliferation and angiogenesis in RCC.
Bevacizumab
Bevacizumab is a humanized monoclonal antibody directed against the VEGF receptor (VEGFR). Its activity was first demonstrated in metastatic RCC in a phase II study that reported a TTP (primary end-point) advantage for bevacizumab at 10mg/kg every two weeks over placebo of 4.8 versus 2.5 months (p<0.001).22 Bevacizumab was well-tolerated, with asymptomatic proteinuria and hypertension being the most frequently detected toxicities. The double-blind randomized AVOREN phase III trial compared the combination of IFN-α (9MU three times weekly) plus bevacizumab with IFN-α plus placebo (see Table 1).23 A total of 649 untreated nephrectomized metastatic RCC patients were recruited, with a primary end-point of OS. Reported median OS was not statistically significantly different; however, analysis of OS may have been confounded by patients receiving second-line therapies that becaimproved with the addition of bevacizumab to IFN-α (10.2 versus 5.4 months; p=0.0001), and this pre-specified analysis was accepted for regulatory submission. The PFS benefit was seen in all risk groups, and was independent of the IFN-α dose. The Cancer and Leukemia Group B (CALGB) 90206 phase III study randomized between IFN-α plus bevacizumab and IFN-α alone and reported a similar PFS benefit (8.5 versus 5.2 months; p<0.0001) and a significant improvement in ORR (25.5 versus 13.1%; p<0.0001) (see Table 1).24 Toxicities of the combination in both phase III studies were manageable: grade 3 hypertension, anemia, fatigue, and proteinuria, occurring more frequently in the combination arm. The evidence for this combination is persuasive; however, its use has been limited by the simultaneous availability of effective oral therapies.
Sunitinib
Sunitinib malate is an oral oxinodole tyrosine kinase inhibitor (TKI) of the VEGF2, PDGF, c-KIT, and FLT-3 receptors.25 It is orally administered at a dose of 50mg once daily for four of every six weeks.26 Phase II studies reported response rates of 40–43%, with an additional 22–27% having stable disease (SD) for more than three months.27,28 Median TTP was 8.7 months (95% CI 5.5–10.7 months)27 and median PFS 8.3 months (95% CI 7.8–14.5 months).28 The most frequently observed toxicities were fatigue and diarrhea.
A total of 750 clear-cell RCC patients were recruited into a randomized multicentre phase III trial comparing first-line sunitinib with IFN-α (see Table 129 The primary end-point was PFS with secondary end-points including OS and ORR. Patients were of good or intermediate performance status by the Memorial Sloan-Kettering Cancer Center (MSKCC) risk criteria30 and over 90% had undergone prior cytoreductive nephrectomy. Sunitinib afforded a PFS advantage over IFN-α (11 versus 5 months, HR 0.42, 95% CI 0.32–0.54; p<0.001) and improved ORR (31 versus 6%; p<0.001). A subsequent OS analysis reported a nonsignificant trend to superiority for sunitinib by intention-to-treat analysis (26.4 versus 21.8 months; p=0.051). However, over 50% of the patients randomized to IFN-α crossed over to sunitinib or other anti-VEGF therapies. Analysis restricted to patients who did not receive secondline therapy demonstrated a substantial and statistically significant OS benefit for sunitinib (28.1 versus 14.1 months; p=0.0033).31 Patients on sunitinib reported a significantly better quality of life (p<0.001). This landmark study led to sunitinib being widely adopted as standard firstline treatment for advanced RCC in 2007.Sorafenib
Sorafenib tosylate similarly inhibits VEGF2 and 3, c-KIT, PDGF, and FLT-3 receptors, but also inhibits the serine/threonine kinases B-RAF and C-RAF of the RAF/ MEK/ ERK pathway.32 Promising activity and safety data from phase I/II studies in metastatic RCC led to a phase II discontinuation study randomizing between either continued oral sorafenib after an initial 12 weeks of treatment or placebo.33 The authors demonstrated a significant PFS advantage with continued sorafenib of 50 versus 18% at 24 weeks (p=0.0077) as the primary endpoint. The sorafenib toxicity profile of fatigue, rash, and hand–foot syndrome was acceptable. A total of 903 patients of low and intermediate MSKCC risk were recruited to the phase III Treatment Approach in Renal Cancer Global Evaluation Trial (TARGET) study comparing 400mg twice daily sorafenib with placebo as second-line treatment (see Table 1).34 More than 80% had received prior cytokine therapy. The sorafenib cohort had a superior PFS of 5.5 versus 2.8 months (p<0.01) and greater PR rate (10 versus 2%; p <0.001).
Subsequent analysis of OS again demonstrated the confounding influence of cross-over.35 OS was not significantly different by intentionto- treat analysis, but censored analysis did demonstrate an OS benefit for sorafenib (HR 0.78, 95% CI 0.62–0.97; p=0.0287). Serious toxicities related to sorafenib included grade 3/4 hypertension (4 versus <1%; p=0.001) and cardiac ischemia or infarction (3 versus <1%; p=0.01).
First-line sorafenib was compared with IFN-α in a recently published randomized phase II trial.36 The primary end-point of PFS was not significantly different (5.7 versus 5.6 months; p=0.50), but sorafenib provided better quality of life, fewer toxicities, and greater tumor response rates. Dose escalation of sorafenib from 400mg twice daily to 600mg twice daily on initial progression resulted in 41.9% response rate,and was well-tolerated. The reason for the relative ineffectiveness of sorafenib as first-line treatment compared with sunitinib is unclear, but is postulated to be due to weaker inhibition of VEGFRs. The concept of achieving greater efficacy from sorafenib by dose escalation is the subject of ongoing trials.
Temsirolimus
The mammalian target of rapamycin (mTOR) pathway has generated significant interest in several tumor types, most notably in RCC. It is downstream from several growth factor receptors, and when activated increases HIF-1α gene expression. Temsirolimus, an intravenous mTOR inhibitor, demonstrated activity in the phase II setting. A clinical trial of 111 patients, the majority of whom had received prior therapy, reported a 7% ORR and a 51% SD rate.37 Grade 3–4 hyperglycemia (17%), hypophosphatemia (13%), anemia (9%), and hypertriglyceridemia (6%) were the most frequent side effects. A phase III study recruited 626 systemic therapy-naïve patients with poor-risk features (see Table 1).38 Patients were randomized to one of three groups: IFN-α 9–18MU three times weekly alone, temsirolimus 25mg weekly alone, or temsirolimus 15mg weekly plus IFN-α 6MU three times weekly. Analysis of the efficacy of temsirolimus alone versus IFN-α alone showed improved median OS (10.9 versus 7.3 months, HR for death 0.73, 95% CI 0.58–0.92; p=0.008) and better PFS (3.8 versus 1.9 months investigator assessment and 5.5 versus 3.8 months by independent assessment; p<0.001) with temsirolimus. Comparison between the combination arm and IFN-α alone revealed no significant difference in median OS (8.4 versus 7.3 months; p=0.70), and the ORR did not differ significantly between all three groups. The toxicity profile of temsirolimus alone was more acceptable than IFN-α alone, but the combination arm resulted in significantly greater toxicities. Therefore, single-agent temsirolimus was established as standard first-line treatment in poor-risk patients.
Everolimus
Everolimus (RAD-001) is an orally bioavailable mTOR inhibitor. Clinical activity was initially demonstrated in an open-label trial of 25 metastatic RCC patients given 10mg daily continuous dosing.39 The PR rate was 32% and a further 51% had SD for greater than three months.me available while the trial was in progress. Median PFS was A phase III study of everolimus versus best supportive care (BSC) recruited 410 patients who had progressed following VEGFR TKI therapy (sunitinib or sorafenib) and many of whom had received other additional lines of treatment (see Table 1).40 The primary end-point was PFS, which favored everolimus (4 versus 1.9 months; p < 0. 0001). It was well-tolerated, with stomatitis, anemia, and rash the most commonly reported toxicities. Everolimus is currently the only treatment showing benefit in a randomized phase III setting following progression on VEGFR TKI therapy.
Other Promising Agents
A number of other agents show clinical efficacy in advanced RCC. Phase II activity has been reported for the anti-VEGFR therapies axitinib,41 pazopanib,42 and cediranib,43 with VEGF-Trap and IMC-1121B also currently undergoing a clinical trial investigation. Other small molecules investigated in a mixture of phase II/III randomized and non-randomized studies include the epidermal growth factor receptor (EGFR) inhibitor erolitinib,44 the EGFR/Her2 inhibitor lapatinib,45 the proteasome inhibitor bortezomib,46 and the anti-TNF-α antibody infliximab.47 Although some have shown activity, other agents remain to demonstrate clinical benefits. A final analysis of the phase III study of lapatinib versus hormonal treatment suggested equivalent efficacy in cytokine-refractory disease, but the choice of control in this study remains debatable for the general clinic population.48 This rapid expansion in novel agents holds great promise, but also presents a considerable challenge in identifying the most effective agents, combinations, and sequences.Metastatic Non-clear-cell Renal Cell Carcinoma
Non-clear-cell RCC accounts for 15% of RCCs and comprises predominantly papillary and chromophobe tumors. In particular, metastatic papillary-cell RCC often responds poorly to therapy and carries a poorer prognosis than clear-cell RCC. Non-clear-cell RCC patients were excluded from many of the main registration studies. Trials have reported activity of sorafenib, sunitinib, and temsirolimus in metastatic papillary tumors and of sorafenib and sunitinib in chromophobe tumors.33,49–52 Therefore, it is reasonable to consider using these agents for metastatic non-clear-cell RCC. Inhibition of c-MET has also proved promising in papillary cell carcinoma, and is currently being further evaluated.53
Scheduling—A Sequential or Combination Approach?
Multidrug schedules aim to overcome resistance and optimize patient outcomes. It is currently unclear whether sequences or combinations of active agents are preferable for the optimal management of advanced RCC. A retrospective analysis of 29 patients who received sorafenib followed by sunitinib and 20 patients who received the reverse sequence has been published.54 Responses were seen in both groups and a non-significant trend toward benefit assessed by median duration of SD and median OS was seen for those who received sorafenib first. A number of other reports validate the potential efficacy of this sequential approach. Responses have also been reported for sunitinib following bevacizumab,55 sorafenib following bevacizumab,56 and sorafenib following bevacizumab or sunitinib.57 Similarly, sunitinib, sorafenib, and axitinib have been reported to have second-line clinical benefit in patients who have received a variety of prior antiangiogenic therapies.41,58
Combination regimens aim to maximize response and overcome or avoid resistance, yet maintain an acceptable toxicity profile. They are widely utilized in the delivery of cytotoxic chemotherapy but, to date, targeted therapies have rarely been combined. Combination regimens employ either ‘horizontal’ or ‘vertical’ blockade. Horizontal blockade inhibits critical targets that lie in different pathways, such as VEGF and PDGF or VEGF and EGFR. Vertical blockade uses combinations that inhibit sequential steps in a single pathway, for example HIF and VEGF or VEGF and VEGFR. Data for novel targeted agents in combination regimens in RCC have been described but are limited. Clinical benefit is seen for the combination of bevacizumab with IFN-α over IFN-α but not for temsirolimus with IFN-α over IFN-α (although temsirolimus monotherapy was superior to IFN-α ). Other combination treatments have been assessed. Phase I and II trials have evaluated and seen activity for combinations of temsirolimus with sunitinib,59 bevacizumab with sunitinib,60 sorafenib with bevacizumab,61 sorafenib with everolimus,62 and bevacizumab with everolimus.63 To date, toxicity has been most notable for the sunitinib-based combinations.
Other novel targets and combinations have been investigated. One such example is inhibition of VEGF by bevacizumab combined with inhibition of EGFR by the EGFR TKI erlotinib.44 Erlotinib 150mg daily plus bevacizumab 10mg/kg intravenous (IV) twice weekly was investigated in a randomized phase II trial of 104 patients against bevacizumab plus placebo and resulted in no significant difference in median PFS (9.9 versus 8.5 months, HR 0.86, 95% CI 0.50–1.49; p=0.58), while OS data were still immature. Numerous other combination regimens are undergoing investigation in the phase II setting, such as sorafenib with infliximab, and in the phase III setting, such as the Eastern Cooperative Oncology Group (ECOG) E2804 BeST trial, in which patients are randomized to bevacizumab alone, bevacizumab plus sorafenib, bevacizumab plus temsirolimus, or sorafenib plus temsirolimus. Other sequencing questions are being evaluated in trials addressing optimal second-line therapy in patientswho are resistant or refractory to first-line anti-VEGF or mTOR therapy. These data of novel sequences and combinations have provoked further research, but are not yet sufficient to inform routine practice.
An Optimal Approach?
The described pivotal phase III trials have resulted in bevacizumab, sunitinib, sorafenib, and temsirolimus being granted regulatory approval by the US Food and Drug Administration (FDA) and the European Medicines Agency (EMEA) in advanced RCC. It is likely that everolimus will be granted a license in 2009. Stratification of the available data according to risk factors, histology, and line of therapy has allowed the drafting of treatment guidelines, such as those published by the US National Comprehensive Cancer Network (NCCN),64,65 the European Association of Urology (EAU), the European Organization for Research and Treatment of Cancer (EORTC), and the International Society of Geriatric Oncology (SIOG).66
Adjuvant therapy after curative nephrectomy remains unproven in the phase III setting, and current recommendations are for either protocolized surveillance or entry into clinical trials. The results of ongoing adjuvant trials with targeted agents are keenly awaited. Although cytokine-based therapy was the cornerstone of treatment for metastatic RCC for many years, it has now been superseded in routine therapy by specific, targeted multikinase and mTOR inhibitors. While IL-2 remains the only treatment to provide durable CRs, its cost in terms of high-dependency support, toxicity, and mortality precludes its widespread use. Evidence-based recommendations for first-line therapy of advanced RCC consist of sunitinib or bevacizumab plus IFN-α combination for good- and intermediate-risk patients and temsirolimus or sunitinib for poor-risk patients (see Table 2). Data for both sunitinib and bevacizumab plus IFN-α are persuasive, although many centers prefer the ease of administration afforded by sunitinib. High-dose IL-2 or clinical trials are alternatives for good- and intermediate-risk patients of good performance status.
Second-line therapy recommendations depend on previous treatment exposure (see Table 2). Cytokine-refractory patients have been shown to benefit from sorafenib, while patients who have failed anti-VEGF treatment could receive second-line everolimus once licensed. Suitable first-line mTOR-inhibitor-failed patients could enter clinical trials or otherwise receive BSC. Sunitinib, bevacizumab, and temsirolimus are rational alternate approaches for cytokine refractory patients, but lack supportive phase III evidence.
The last five years have seen a genuine paradigm shift in the treatment of RCC. Current data allow consensus over an optimal approach based on patient risk categories, performance status, and administration preference. RCC clinicians now face the responsibility of designing clinical trials that will best determine the most effective agents, sequences, or combinations for patients with RCC and to investigate how molecular biology and functional imaging can be utilized to optimize predicted therapeutic benefit. ■