The original Memorial Sloan-Kettering Cancer Center (MSKCC) risk group categorisation identified five high-risk factors in cytokine-treated patients:
- Karnofsky performance status (KPS) <80;
- serum calcium >10 (corrected for albumin);
- haemoglobin below normal;
- absence of prior nephrectomy; and
- lactate dehydrogenase (LDH) >1.5 x upper limit of normal.1
The median survivals were 20, 10 and four months for good- (no risk factors), intermediate- (one to two risk factors) and poor-risk patients (three or more risk factors), respectively. In another analysis at MSKCC, similar adverse prognostic factors emerged, although absence of nephrectomy was replaced by less than one year from initial diagnosis to start of therapy.2
The MSKCC criteria were validated in a separate data set at the Cleveland Clinic, and in addition to four of the five MSKCC criteria (excluding KPS), prior radiotherapy and presence of hepatic, lung and retroperitoneal nodal metastases were found to be prognostic factors.3 Using the number of metastatic sites as a surrogate for individual sites (zero to one versus two to three), MSKCC risk groups were expanded to accommodate these two additional prognostic factors. Similar prognostic factors were also subsequently demonstrated to be important for cytokine-pre-treated patients in an analysis from MSKCC.4 Update of Current Systemic Therapy for Renal Cell Carcinoma
Sunitinib Malate
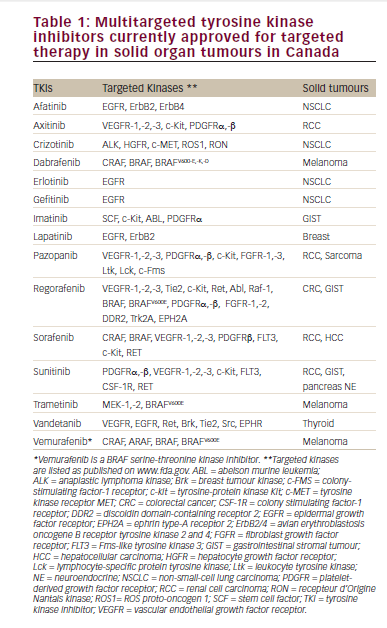
Sunitinib, a multitargeted tyrosine kinase inhibitor (TKI), was compared with IFN-α in the front-line setting in a trial of 750 patients with clear-cell RCC (see Table 1).5,6 Sunitinib yielded a significantly longer median progressionfree survival (PFS) (11 versus 5.1 months) and response rate (RR) (39 versus 8%). Almost all responses were partial responses, and all risk groups appeared to benefit, although the poor-risk subset included only a small cohort of patients. In a recent update, the median overall survivial (OS) imporved with sunitinib (26.4 versus 21.8 months; p=0.051).6 On censoring patients who crossed over from IFN to sunitinib, the difference in survival attained statistical significance (26.4 versus 20 months; p=0.0362). When considering only those patients who did not receive any post-study therapy, sunitinib again significantly outperformed IFN, with OS of 28.1 and 14.1 months, respectively (p=0.0033). Patients in the sunitinib group had higher rates of grade 3 diarrhoea (5 versus 0%), vomiting (4 versus 1%), hypertension (8 versus 1%) and the hand–foot syndrome (5 versus 0%). Grade 3 decline in left ventricular ejection fraction seldom occurred (2 versus 1%) and was reversible. Modest myelosuppression was observed, with grade 3–4 neutropenia, thrombocytopenia and anaemia in 12, 8 and 4%, respectively. Thyroid function abnormalities appear to be common, and routine monitoring is warranted.7 Based on these data, IFN-α has been supplanted by sunitinib as the reference standard for advanced clear-cell RCC. Sunitinib has also been demonstrated in separate phase II trials to have significant activity following prior therapy with cytokines or bevacizumab.8–10
Bevacizumab
The AVastin for Renal Cell Cancer (AVOREN) trial randomised 649 previously untreated patients with a significant clear-cell component (>50%) to front-line IFN-α 2a plus placebo or IFN-α 2a plus bevacizumab, a humanised monoclonal antibody to vascular endothelial growth factor (see Table 1).11 The addition of bevacizumab to IFN-α 2a significantly increased median PFS (10.2 versus 5.4 months, hazard ratio [HR] 0.63; p<0.0001) and investigatorassessed objective response rate (30.6 versus 12.4%; p<0.0001). The median PFS for combination bevacizumab and IFN-α versus the IFN-α group was 12.9 versus 7.6 months in the favourable prognosis group, 10.2 versus 4.5 months in the intermediate prognosis group and 2.2 versus 2.1 months in the poor prognosis group, suggesting that good- and intermediate-risk patients appear to garner most of the benefit. A trend towards improved OS was observed with the addition of bevacizumab to IFN-α 2a (p=0.0670), although this analysis was confounded because physicians were encouraged to offer bevacizumab to patients on the placebo arm after the interim PFS analysis and due to the increasing availability of TKIs. Grade 3–4 adverse events with bevacizumab included four gastrointestinal perforations (1%) and 10 thromboembolic events (3%). Seven patients (2%) with hypertension discontinued treatment due to this event and 16 patients (5%) discontinued due to proteinuria. Toxic deaths were reported in eight patients (2%) who received bevacizumab and in seven (2%) of those who did not receive the drug.
The similar but not placebo-controlled Cancer and Leukemia Group-B trial was recently reported and confirms improved outcomes with bevacizumab plus IFN-α compared with IFN-α alone.12 The median time to progression was 8.5 months in patients receiving bevacizumab plus IFN versus 5.2 months with IFN monotherapy (HR 0.71; p<0.0001). Bevacizumab plus IFN had a higher objective RR (25.5 versus 13.1%; p<0.0001) and exhibited expected but manageable toxicities. Given the PFS reported in a previous trial of bevacizumab alone of 8.5 months in the front-line setting, the value of adding IFN-α 2a is unclear.13 Following cytokines, the RR with a similar dose of single-agent bevacizumab (10mg/kg every two weeks) was 10%, with a median time to progression of 4.8 months.14 The combination of bevacizumab and IFN-α 2a is a legitimate option for the front-line therapy of advanced good- and intermediate-risk clear-cell RCC.
Temsirolimus
In a randomised phase II trial of three different doses of the mammalian target of rapamycin (mTOR) inhibitor temsirolimus in the setting of prior cytokine therapy, in most patients an objective RR of 7% and a minor response rate of 26% was observed. Median time to tumour progression was 5.8 months and median survival was 15.0 months. Intermediate- and poor-risk patients appeared to selectively benefit compared with historical controls.15
Temsirolimus, IFN-α 2a and combination temsirolimus plus IFN-α 2a were compared in a subsequent 626-patient phase III trial that selected patients with poor-risk RCC (see Table 1).16 At least three of the following six poor prognostic factors were required:
- serum LDH >1.5 times the upper limit of the normal;
- haemoglobin level below the lower limit of normal;
- corrected serum calcium level >10mg/decilitre;
- less than one year from initial diagnosis to randomisation;
- KPS of 60 or 70; and
- metastases in multiple organs.
These poor-risk criteria are slightly modified from the original MSKCC criteria, and essentially allowed the accrual of mostly poor- and some intermediate-risk patients according to the original MSKCC criteria. Median survival was 7.3 months in the IFN group, 10.9 months in the temsirolimus group and 8.4 months in the combination group. Compared with interferon alone, this translates into a 49% improvement in median survival in patients in the single-agent temsirolimus arm, while the combination of temsirolimus plus IFN did not improve OS. The objective RRs of 4.8, 8.6 and 8.1% among patients receiving IFN, temsirolimus and combination therapy, respectively, did not differ significantly. On exploratory subset analysis, the effect of temsirolimus on OS was greater for patients under 65 years of age with serum LDH >1.5 times the upper limit of normal and non-clear-cell RCC.
The most common grade 3–4 adverse effects of temsirolimus were asthaenia (11%), rash (4%), anaemia (20%), nausea (2%), dyspnoea (9%), diarrhoea (1%), peripheral oedema (2%), hyperlipidaemia (3%) and hyperglycaemia (11%). Based on these data derived from a large phase III trial, temsirolimus should be considered the preferred agent for poor-risk RCC, and perhaps the preferred agent for non-clear-cell RCC, although this needs further evaluation. Sorafenib
The Treatment Approaches in Renal Cancer Global Evaluation Trial (TARGET) enrolled 903 patients with only good- and intermediate-risk clear-cell RCC and compared sorafenib, a multitargeted TKI, with placebo following prior cytokine therapy (see Table 1).17 The median PFS was 5.5 months with sorafenib and 2.8 months with placebo (HR 0.44; p<0.01). Responses were seen in 10% of patients receiving sorafenib and in 2% of those receiving placebo (p<0.001). The survival improvement of 13.5% for sorafenib was not significant (median 17.8 versus 15.2 months, HR 0.88; p=0.146). However, secondary analysis censoring cross-over data showed a significant OS benefit for sorafenib (HR 0.78; p=0.0287), since patients received benefit from sorafenib upon cross-over.18 Severe grade 3–4 diarrhoea, rash, fatigue, hypertension and hand–foot skin reactions occurred in a small proportion (1–6%) of patients with sorafenib.
A smaller randomised phase II trial in the front-line setting did not demonstrate improved outcomes with sorafenib compared with IFN-α.19 Sorafenib is an active agent, but is increasingly reserved for salvage therapy following other agents given the lack of robust front-line data (see Table 2).
In a recently reported phase II trial, 27 evaluable patients with clear-cell RCC refractory to either bevacizumab (n=11) or sunitinib (n=16) were treated with sorafenib.20 No objective responses have been observed, although nine patients (33%) had >5% decrease in tumour burden, with a median PFS of 3.7 months. Interestingly, there was no association between tumour shrinkage and response to prior therapy. Toxicities were significant, with grade 3 toxicities experienced by 67% of patients. Other retrospective reports support the presence of only partial cross-resistance between available novel anti-angiogenic agents.21–23 Hence, a rationale can generally be made for continued targeting of angiogenesis with a sequence of different agents (see Table 2). Everolimus
A phase III trial of a novel oral mammalian target of rapamycin (mTOR) kinase inhibitor, everolimus (RAD-001), versus placebo was completed with a 2:1 randomisation.24 These data were presented at the American Society of Clinical Oncology 2008 annual meeting. Four hundred and ten patients were enrolled; patients who had received sunitinib only, sorafenib only or both constituted approximately 45, 30 and 25%, respectively. Additionally, 50% of patients had received prior IFN, and approximately 50% had received interleukin (IL)-2, chemotherapy or bevacizumab, but none had received prior temsirolimus. Almost all patients had undergone prior nephrectomy. Treatment with everolimus was associated with a significant improvement in PFS compared with placebo, the primary end-point of the trial (4.0 versus 1.9 months, HR 0.30, 95% CI 0.22–0.40; p<0.001). The sixmonth PFS was 26% versus 2%, although the Response Evaluation Criteria in Solid Tumors (RECIST) response rate with everolimus was low at only 1%. Additionally, all risk groups appeared to benefit. Everolimus exhibited a favourable toxicity profile, with a low incidence of myelosuppression and a low incidence of grade 3–4 non-haematological toxicities (12% hyperglycaemia, 4% hypophosphataemia, 3% hypercholesteroleamia, stomatitis, pneumonitis, infections and fatigue). Based on these data, everolimus should be considered a standard for patients with progressive clear-cell RCC following prior TKIs, and formal approval by regulatory agencies is awaited.
High-dose Interleukin-2
High-dose (HD) IL-2 has been employed for metastatic RCC based on a small fraction (~7%) of patients who demonstrate durable complete remission (CR) and apparent cures.25 Clinical trials have demonstrated improved objective RRs and response duration when HD IL-2 was compared with other administration schedules of IL-2 and IFN.26,27 However, no randomised trial has demonstrated an overall survival advantage. Additionally, the commonly observed life-threatening toxicities of HD IL-2 severely limit its application. The best outcomes have been associated with good- and intermediate-risk clear-cell RCC, certain histological features (clear-cell and alveolar features; absence of papillary and granular features), carbonic anhydrase IX (G250 antigen) expression and certain gene expression profiles.28–31
The Cytokine Working Group recently launched the HD IL-2 Select trial to prospectively determine whether predictive factors (baseline immune function, immunohistochemical markers and gene expression patterns) can identify patients more likely to respond to HD IL-2 than a historical, unselected patient population.
Role of Cytoreductive Nephrectomy
Randomised trials have demonstrated a significant survival advantage in metastatic RCC with cytoreductive nephrectomy followed by IFN-α.32,33 Retrospective reviews also suggest that this approach may improve outcomes in patients receiving HD IL-2.34 The vast majority of patients accrued on trials with novel TKIs had undergone prior nephrectomy. However, the role of prior nephrectomy in improving outcomes in the era of novel highly active agents remains to be defined.
The paradigm of pre-surgical systemic therapy followed by cytoreductive nephrectomy may be employed to develop individualised therapy, elucidate mechanisms of resistance and develop reliable prognostic and predictive biomarkers. Early data suggest that administration of the novel antiangiogenic agents prior to surgery is not associated with increased perioperative mortality or morbidity.35 The MD Anderson Cancer Center reported that bevacizumab discontinued at least four weeks before and sorafenib/sunitinib discontinued at least 24 hours before surgery appear feasible. In an ongoing prospective clinical trial at MD Anderson, patients are randomised to upfront surgery followed by sunitinib two weeks after the operation or sunitinib for one to four weeks prior to nephrectomy performed 24 hours after discontinuation of sunitinib, and then followed by resumption of sunitinib. Another randomised trial in France is comparing front-line sunitinib with versus without initial cytoreductive nephrectomy in order to elucidate the value of this procedure with sunitinib therapy. Therapy of Non-clear-cell Renal Cell Carcinoma
Based on the large randomised trial comparing temsirolimus with IFN demonstrating improved OS and a more profound benefit for the subset with non-clear-cell RCC, temsirolimus may be considered an option for the front-line therapy of non-clear-cell RCC with poor-risk features.16 In another report, 41 patients with papillary and 12 with chromophobe histologies who received sorafenib or sunitinib were analysed.36 RR, PFS and OS for the entire cohort were 10%, 8.6 months and 19.6 months, respectively. Three of 12 patients (25%) with chromophobe RCC achieved a response, while two of 41 (4.8%) papillary RCC patients responded. However, these outcomes are clearly suboptimal and a clinical trial is an excellent alternative, notably to evaluate Met inhibitors for papillary RCC (see Table 3).
Ongoing Clinical Trials for Advanced Renal Cell Carcinoma
Generally, patients progressing after prior TKIs, bevacizumab or temsirolimus are candidates for clinical trials (see Tables 2 and 3). Notwithstanding the improved outcomes seen with the above agents, previously untreated patients are also legitimate candidates for clinical trials since current therapy is palliative and not curative. The thrust of ongoing clinical research is to continue to explore novel agents, devise optimal combination and sequencing strategies, re-visit the role of cytoreductive nephrectomy, evaluate these agents in the adjuvant setting and discern factors predictive of response.
The BEST trial is planned to compare the following four arms: bevacizumab alone; bevacizumab plus temsirolimus; bevacizumab plus sorafenib; and sorafenib plus temsirolimus. Another trial will compare sunitinib with sunitinib plus enzastaurin, a novel orally administered inhibitor of protein kinase C (PKC)-β and Akt. Toxicities have rendered the combination of sunitinib with IFN, temsirolimus or bevacizumab difficult. The ongoing renal EFFECT trial compares two schedules of sunitinib (50mg daily for four of every six weeks and 37.5mg daily continuous).
Novel multitargeted TKIs including pazopanib and axitinib have demonstrated promising activity accompanied by favourable toxicity profiles.37–39 Pazopanib and axitinib are novel multitargeted TKIs that appear to have significant activity in clear-cell RCC.40–42 A placebocontrolled phase III trial of pazopanib in patients with clear-cell RCC who are untreated or have either failed or are intolerant to cytokines has completed accrual and results are pending. A recently initiated phase III trial will compare sunitinib with pazopanib. Another front-line trial is comparing the combination of bevacizumab with IFN or temsirolimus. A trial will compare salvage everolimus with everolimus plus bevacizumab, while another ongoing randomised trial will compare first-line everolimus plus bevacizumab versus interferon plus bevacizumab. Other salvage trials following prior sunitinib are planned or ongoing: temsirolimus versus sorafenib in patients who have failed sunitinib, and axitinib versus sorafenib in patients who have failed one or more of the following: sunitinib, bevacizumab + IFN, temsirolimus or cytokines. Ongoing trials are also evaluating sunitinib or sorafenib as adjuvant therapy for high-risklocalised RCC following nephrectomy (ASSURE, SORCE, S-TRAC). Biomarkers predictive of outcomes and response (e.g. baseline serum vascular endothelial growth factor [VEGF], soluble VEGF receptors, circulating endothelial cells) are being developed and may make it possible to individualise therapy and confer a higher therapeutic index.43
Vaccine Therapy
Immunotherapy with cytokines and vaccines continues to be evaluated, both alone and in combination with other agents. A total of 183 patients with metastatic RCC were treated with dendritic cell vaccination in 15 phase I/II clinical trials.44 Seventy-seven patients (38%) had a clinical response, with four complete and eight partial responses, and 61 had disease stabilisation, whereas four had a mixed response. TroVax is a 5T4 tumour-associated antigen delivered by poxvirus (modified vaccinia ankara [MVA]).45 MVA is recognised and processed by the immune system, which stimulates a systemic anti-5T4 immune response and generation of anti-5T4-specific antibodies and/or CT8+ cytotoxic T cells. The body’s own immune system then targets cancer cells expressing 5T4. The 5T4 antigen is defined by a monoclonal antibody raised against 72kDa oncofoetal glycoproteins isolated from syncytial trophoblast microvillous membranes that are expressed on >70% of carcinomas of the kidney, breast, gastrointestinal tract and ovaries, and is highly expressed in renal cancer. It appears to be tumour-specific, with only low-level sporadic staining observed in the gut and pituitary. Tumour cells transfected with the complementary DNA (cDNA) encoding 5T4 display increased motility, as 5T4 may induce metastatic properties in a tumour.
The phase III industry-sponsored TroVax Renal Immunotherapy Survival Trial (TRIST) will include 700 untreated patients with locally advanced or metastatic measurable clear-cell RCC (see Table 2). Patients are stratified by MSKCC criteria, geography and standard of care, with a primary end-point of OS and secondary end-points of progression at six months and quality of life. Patients are randomised 1:1 between standard agents alone (subcutaneous [sc[ IL-2, sc IFN-α, sunitinib) or in combination with TroVax.
Vaccines are also emerging as active agents in the setting of micrometastases as adjuvant therapy.46 Vaccination with autologous heat-shock protein (HSP)-peptide (Oncophage®) complexes produced from each patient’s tumour has been investigated in early-stage highrisk RCC compared with no therapy in a phase III trial.47 Of the 728 patients entered, data review indicated that 124 (17%) were ineligible as they had disease present at baseline. When the 604 eligible patients were analysed, recurrence-free survival (RFS) was not statistically different between the arms. An ad hoc hypothesis-generating analysis demonstrated a significant improvement in RFS of approximately 45% (HR 0.55; p<0.01) and a trend towards improved survival in a subset of intermediate-risk patients (n=362). A randomised placebocontrolled phase III trial (ARISER) is evaluating WX-G250, a monoclonal antibody against CA-IX, for the adjuvant therapy of resected localised high-risk clear-cell RCC.
Conclusion
The burgeoning list of active agents for the therapy of RCC presents a unique set of challenges. However, the rational selection of currently approved agents for the therapy of patients with RCC in different settings is feasible by implementing evidence-based medicine. Incorporation of risk factor models will enable clinicians to appropriately select patients for specific agents. It is also critical to remember that the vast majority of patients are not cured with currently approved agents, and continued support of clinical trial programmes is essential (see Tables 2 and 3). The future for the therapy of RCC appears promising, with the emergence of novel active agents and with a plethora of other exciting agents waiting to be evaluated.
Disclosures
Author disclosures are as follows:
- Thomas E Hutson receives research support from Bayer/Onyx, Pfizer and GlaxoSmithKline; is an Advisory Board Member/Consultant for Bayer/Onyx, Pfizer Dendreon and sanofi-aventis; and is on the Speakers’ Bureau for Bayer/Onyx, Pfizer, Amgen, sanofi-aventis, Novartis and Genentech.
- Guru Sonpavde receives research support from Eli Lilly, Pfizer, BMS, Cytogen and AstraZeneca and is on the Speakers’ Bureau for Pfizer, Wyeth, Novartis and sanofi-aventis.
- Cora N Sternberg receives research support from Eli Lilly, sanofiaventis, Pharmion, GPC Biotech and Bayer/Onyx.